Biodegradable Polymer Nanoparticles: Therapeutic Applications and Challenges
Department of Chemistry, GLA University, Mathura, India.
Corresponding Author E-mail: dr.rama76@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/380612
Article Received on : 03 Nov 2022
Article Accepted on : 04 Dec 2022
Article Published : 21 Dec 2022
Reviewed by: Dr. S.K. Pandey
Second Review by: Dr. Faheem Khan
Final Approval by: Dr. Vandana Magarde
The greatest medication encapsulation and distribution options have received substantial research on biodegradable natural polymers. For their potential to act as an effective vehicle for site-specific medication delivery in the body, biodegradable nanoparticles (NPs) are attracting more interest. They provide enhanced biocompatibility, and practical release patterns for a variety of medicines to be used in a number of applications. This article has explored the various applications of these particles, including cancer therapy, implantable device, and antioxidant delivery. However, there is still potential to investigate more biodegradable polymers for cutting-edge biological applications.
KEYWORDS:antioxidant delivery; Biodegradable nanoparticles; challenges; cancer therapy; Implantable device
Download this article as:| Copy the following to cite this article: Sharma R. Biodegradable Polymer Nanoparticles: Therapeutic Applications and Challenges. Orient J Chem 2022;38(6). |
| Copy the following to cite this URL: Sharma R. Biodegradable Polymer Nanoparticles: Therapeutic Applications and Challenges. Orient J Chem 2022;38(6). Available from: https://bit.ly/3Wfr0MZ |
Introduction
Innovative carriers for the organization of drug atoms, biodegradable nanoparticles (NPs) are a new type of nanotechnology. The arrangement of drug atoms is done using innovative transporters called biodegradable nanoparticles (NPs). Due to their distinctive qualities, biodegradable nanoparticles have recently grown in prominence, including targeted medicine delivery, enhanced bioavailability, and improved therapeutic viability to administer the medication at a steady rate1. With the right design, nanoparticles have a huge potential to increase the viability of active anticancer agents by focusing more intensely on the correct place for a successful treatment with fewer adverse effects. A few natural boundaries should be considered to facilitate a basic conveyance, starting at the level of the framework and moving down to the level of the organ and the cell2. Recently, a significant amount of research on nanoparticles has been conducted with a number of uses in the therapeutic field. Nanoparticles are designed to transfer medications in an improved and maintained way. The development of long-flowing nanoparticles with high catalytic productivity has been inspired by recent breakthroughs in nanotechnology. This article focuses on characteristics like biocompatibility and biodegradability, which are regarded as essential requirements for nanoparticles to be used successfully as a transporter atom in drug delivery systems. Understanding the physical properties of nanoparticles, notably their particle size and dispersion, zeta potential, and surface science, is crucial for the development of efficient drug delivery systems. In general, nanoparticle size and surface properties enable enhanced intracellular gathering in tumor cells through detached targeting elements and quick advancement of nanoengineering and help towards achieving dynamic targeting delivery by co-functionalization of nanoparticles utilizing appropriate targeting ligands3. Due to their ease of incorporation and the fact that they have no effect on cells or tissues, biodegradable nanoparticles have generated a great deal of interest in pharmaceuticals and quality delivery systems. This article provides an overview of how biodegradable nanoparticles are used in quality treatment and boost the effectiveness of the attributes that are being targeted.
Biodegradable Polymer Nanoparticles and Their advantages
The same as their non-biodegradable counterparts, biodegradable nanoparticles can be organized according to the way they are made and how they are used, either by encapsulating the elements of interest in nano capsules or by merging them into nanospheres. Dendrimers serve as an example of nanospheres, and micelles, liposomes are other examples of good nano capsules.
The use of biodegradable nanoparticles supports the overall benefits of nanomedicine, such like controlled and moderated load delivery and targeted conveyance, which boost restorative effects and diminished outcomes, particularly for some cytotoxic medications. Biodegradable nanoparticles have the additional benefit of lowering cytotoxicity in the body. To further enhance the drug release profile and focus on proficiency, other surface changes should also be allowed.
Polymers have demonstrated good biocompatibility and biosafety when creating biodegradable nanoparticles. In order to regulate drug discharge, it is also possible to modify the surface and size of the molecules4. Potent polymeric nanoparticles of 10-500 nm size can be employed to transport beneficial species to the surface by either implanting or encapsulating the species inside their polymeric gride4,5 . Based on the fundamental materials used to make the nanoparticles, polymeric nanomaterials can be separated into two major types: blended compounds like poly-D-L-lactide-co-glycolide (PLGA), polylactic acid (PLA), poly- ε-caprolactone (PCL); and common compounds like chitosan. This collection of polymers can go through a cycle of debasement to become tarnished into substances that can be safely handled inside the body. Numerous internal and external variables, including the size and shape as well as internal variables like pH and temperature, both have an impact on the payload delivering profile, all affect the rate of reaction of polymeric nanoparticles. Comparing engineered polymers to their conventional polymer counterparts, the overall benefit of a fairly lengthier drug discharge period is enjoyed6 .
Drugs, antibodies, and other biomolecules have been delivered at specific sites using biodegradable nanoparticles. Among the most popular biodegradable polymer matrices for the preparation of nanoparticle are:
Poly-lactic-co-glycolic acid (PLG)
PLG is one of the most widely utilised biodegradable polymers (Fig.1). Get hydrolyzed as it enters the body to create biodegradable metabolite monomers such lactic and glycolic acid. As lactic acid and glycolic acid is already present in body and participating in different pathways so there is almost no risk to use PLG for successful delivery of drugs. Most commonly, emulsification-dissemination, dissolvable dissipation, and nanoprecipitation procedures have been used to create PLG NPs7 . PLG nanoparticles have been utilised in the fabrication of protein- and peptide-based nanomedicines, and for in-vivo delivery systems6,7 .
 |
Figure 1: The PLGA’s structure. The suffixes x and y, respectively, indicate the quantity of lactic and glycolic acids 8. |
Polylactic acid (PLA)
In the body, the polymer PLA(Fig.2) breaks down into lactic acid monomers, making it biocompatible and biodegradable. As part of the Calvin cycle, the liver converts lactic acid into glucose, a typical transitory outcome of anaerobic breath. When it happens, the body starts using glucose as fuel. As a result, using PLA nanoparticles is safe and doesn’t cause any substantial harm. The majority of the methods used to create PLA nanoparticles9,10 include soluble uprooting, salting out, and soluble dispersion. The fundamental advantage of the salting out technique is that it releases pressure on protein encapsulants 7.
 |
Figure 2: Poly lactic acid’s chemical structure (PLA)8 |
Poly-ε-caprolactone (PCL)
In human body, the ester bond present in PCL (Fig.3) is degraded by hydrolysis and produce compound which is almost nontoxic. In this regard, analysts have begun to pay attention to PCL as a potential competitor for use in implanted devices and medicine delivery. Due of PCL’s slower rate of deterioration than polylactides, it is now a stronger applicant for long-lasting implantable devices. Most of the time, nanoprecipitation, dissolvable uprooting, and dissolvable dissipation have been used to arrange PCL nanoparticles7,11,12.
 |
Figure 3: Poly-ε-caprolactone (PCL) Chemical structure 8. |
Chitosan
The scavenger found ordinary biopolymer chitin which was incompletely N-deacetylated to produce chitosan (Fig.4), a changed normal sugar polymer. For the readiness of chitosan nanoparticles, four strategies have been announced- polyelectrolyte complex formation, ionotropic gelation, microemulsion, and emulsification solubilized dissemination7,13,14.
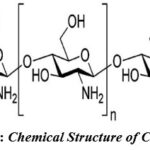 |
Figure 4: Chemical Structure of Chitosan 8. |
Gelatin
A common and secure element in food and medicine is gelatin. Gelatin NPs are useful for delivering drugs in a regulated manner. They are inexpensive, non-toxic, bioactive, and degradable. Gelatin (Fig.5) is ampholytic in nature containing cation and anion clusters as well as a hydrophilic cluster.
How many cross links there are between molecules cationic and anionic groups that gelatin exhibits affect its mechanical properties, including its ability to retain heat and work. Gelatin’s properties can be changed to create the specific kinds of NPs that are needed. Emulsion, desolvation, or a combination of these processes can be used to produce gelatin nanoparticles 7,15,16 .
 |
Figure 5: Gelatin Chemical Structure 8. |
Poly-alkyl-cyano-acrylates (PAC)
Chemical esterases present in the body degrade poly-alkyl cyanoacrylates (Fig.6), which are both biodegradable and biocompatible. They produce some toxic substances upon degradation that either energize or destabilize the focused sensory system. As a result, it is not permitted to use this polymer on humans. PAC nanoparticles are typically organized by polymerization reaction, and nanoprecipitation7,9.
 |
Figure 6: Chemical structure of Poly-alkyl-cyano-acrylates8. |
Immunology
Since different nanotechnologies have developed over time, they can now be used in a variety of fields, primarily for the drug delivery and diagnostics17–21. The biocompatibility requirements for using nanomaterials (NMs) successfully in medicine are far exceeded by the physicochemical variability of NMs. As an example, growing concerns about tissue collection and cytotoxicity result from the fact that the great majority of inorganic or carbon-based compounds are not biodegradable and ineffectively dissolvable22. High concentrations of inorganic nanoparticles (NPs) alter physiological processes that cause injury, but smaller doses could not exhibit immediate toxicity but could hide long-term events that would manifest later. Since NPs have the ability to trigger potentially explosive reactions that need to be appropriately examined before any biological case, NP communication with the resistive framework (IS) needs special attention23 . The physical properties of the NPs determine whether or not the IS’s cells and atoms will recognize them. In particular, the distinctive surface science frequently identifies NP insusceptible similarities and plays a significant impact in how humans interact with the climate24 . NM coatings that can evade the IS and delay their release once administered, such as in the circulatory system, are the subject of ongoing research25 . To improve drug delivery frameworks, NM surfaces can be modified with polymers like poly (ethylene glycol) or poly(2-oxazoline). and useful nano-instruments is perhaps the most well-known technique. Many researchers are actively striving to trigger a resistant response by tailoring inorganic or natural NPs for explicit adjuvant mobility in antibody conveyance, although there haven’t been many new materials developed to stop IS 26 . Numerous NMs reveal amazing drug/quality conveyance performances or immunomodulatory activities27 . Some of them already contribute significantly, particularly in the delivery of Covid 2 (SARS-CoV-2) antibody, while others have potential uses for interfering with odd resistant responses such as the COVID-19-triggered cytokine storm28.
The abundance of evidence emphasizes the need for a thorough investigation of nanomaterials induced invulnerable reactions in order to separate their safe dangerous components and design safe particles for therapeutic applications. This article concentrates on a few models biodegradable NPs and their untapped potential.
A few formulations are gradually incorporating nanotechnology’s advantages29,30. Particularly, biodegradable NPs reveal extraordinary flexibility of action in the medical field. The possibility of designing safe, functional nano-devices will support the development of very effective quality or drug delivery systems. The use of degradable NPs with tailored resistance responses could also advance antibody adjuvant technology. In any case, the astonishingly rapid development of NM’s improvement will require constant immunological research and painstakingly controlled IS calibration. The immunizer intervened uncontrolled safe reactions to PEGylated NPs previously used in nanomedicines propose that NP surface science and its alterations to expand solvency and postponed flexibility be further studied.
Applications
In Cancer Therapy
In developed nations, cancer is a leading cause of death. Chemotherapy, radiation, and medical procedures are currently the standard treatment modalities for the management of cancer. Chemotherapeutic professionals destroy healthy cells during chemotherapy, which reduces the useful vitality and causes high poisonousness. Chemotherapy is at present widely used. Therefore, delivering the drug selectively to malignant cells in order to extend their life and lessen their toxicity is the main goal of nanomedicine in the treatment of oncological disorders. For instance, Park et al. have shown that the most serious side effect of using this medicine is the development of cardiomyopathies, which is greatly reduced when doxorubicin, a powerful antineoplastic agent, is incorporated in pegylated PLGA-NPs31. Polymeric NPs have been found to transmit several Chemotherapy medications, such as doxorubicin, camptothecin, etc in a variety of malignant growths. It should be mentioned that many forms of cancers are not susceptible to the remarkable supporting intercessions due to variations in the cell tools, which, in turn, accounts for the multidrug resistance32 .
Poly(lactide-co-glycolide)-d-tocopheryl polyethylene glycol 1000 succinate nanoparticles (NPs) were successfully employed in heart disease-safe cells to mimic doxorubicin and metformin as P-glycoprotein inhibitors33 . Drug delivery methods used by NPs in the treatment of neoplastic diseases can include dynamic focusing or uninvolved focusing, with the latter method being strictly regulated by the former34 . Detached targeting involves delivering the medication by utilizing the tumor’s characteristic vascularization, which permits atoms to split via convection or unbiased dispersion in the interstitial space and in living cells. A common interaction identified in many tumors influencing individuals is the improved penetration and maintenance impact (EPR-impact), which is specifically misused. When conditions such as hypoxia or a fiery state are present, the endothelium lining of veins becomes more porous, enabling particles that gather in the tissue spaces to enter35 . The NPs are adequately functionalized to allow active targeting by blocking certain antibody, protein, or peptides from being present on the exterior. Finally, the receptor type that is continuously overexpressed in tumour cells was taken into consideration while choosing the ligand36 .
Epigallocatechin-3-gallate (EGCG) transporter polymeric nanoparticles were examined by Sanna et al. in models of prostate cancer cells37. They employed three distinct nanoparticles, and the dynamic fixing was demonstrated. They dispersed non-functionalized forms of two distinct NPs that were correctly tagged with tiny particles made to bind to the PSMA, which, in particular, is overexpressed in prostate cancer. PSMA is a prostate-explicit layer antigen. They used pseudo-mimetic dipeptide (DCL-NPs) and aspartate-glutamine (AG-NPs) to functionalize PLGA-PEG NPs (Fig. 7). They were able to demonstrate that the EGCG-containing nanoparticle framework had an impact on proliferation in vitro and the ability to prevent the growth of tumors in mice models. Additionally, a crucial increase in the opponent of cancer action was noticed at the site of in vivo activity for the functionalized NPs38 .
 |
Figure 7: Schematic representation of the designed targeted EGCG NPs37. |
One of the main goals of nanomedicine is the use of polymeric NPs to treat cancer since they could be tested on substances that are generally avoided during chemotherapy because they are so dangerous but could really be helpful in this situation.
The addition of polyethylene glycol (PEG) chains to the outside of the nanoparticles (NPs) results in reduced reticuloendothelial framework (RES) uptake and increased length in the flow. With this alteration, there is a new probability that the supportive specialist will achieve its goal without needing to modify the medication’s substance. A method of treatment for ovarian tumours included testing of the chemotherapy medication DOX. By adding a functionalized end to the PEG chains, the extracellular section of the FSHR receptor-specific MAb was transferred to the surface of the NPs. The PEG wire updated several aspects, such as the stream’s increased duration and the association’s likelihood of focusing on moieties outside of NPs.
By using biocompatible polymers instead of iron oxide on NPs’ surfaces, it may be possible to create biodegradable drug delivery systems that are useful for both in vivo and in vitro applications. To focus in vitro development on ovarian malignant, integrated polymeric nanocomposites were created by individually combining the anticancer medications (DOX, PTX). After 40 days, the developed plan resulted in a decrease in ovarian tumor size from 2920 to 143 mm in immunodeficient female Balb/c mice39.
A composite material for NPs is made of polyurethane because of its great synthetic flexibility. By choosing the optimum amount of folic acid (FA) to functionalize drug-stacked nanogels, it is possible to decide the materials’ ultimate properties while maintaining the stability of the carrier and preserving the ligand-restricting partiality. Different masking patterns for ovarian cancer cells have been noticed when the actin fibre was revised. Furthermore, the ultimate fate of NPs, in which they ultimately decay and deliver their contents, was also observed37.
Implantable Devices
The functionality of implanted devices composed of biodegradable materials is constantly being improved. Biodegradable nanoparticles can be included in implantable devices like muscle obsession devices in addition to being employed in nano drug delivery systems (such as meniscal fix tacks, fixation pins and plates for break obsession, impedance screws, and stitch secures) and biodegradable stents for percutaneous coronary intervention40,41. Some of these devices have been successfully supported and may be purchased on the market42 . The embedded devices are given the ability to gradually degrade, owing to the biodegradable nanoparticles during the beneficial reconstruction process, the host tissues gradually replace the implant43 .
Significant advancements have been made, especially in the area of interventional cardiology. In addition to limiting neointimal hyperplasia and reducing restenosis following revascularization, new medication-eluting stents have been developed to also reduce stent apoplexy, a problem that was reported at higher recurrence with the original stents44 . By neutralizing the acidic environment with the help of oligo(lactide)-joined magnesium hydroxide nanoparticles, Lih et al. have created a new method to stop corrosive-induced incendiary reactions related to biodegradable PLGA. They demonstrated in pig models that modifying Mg(OH)2 nanoparticles added to disintegrating coatings on drug-eluting blood artery stents could effectively reduce the stimulating reaction and in-stent intimal thickening45 . According to their research, biodegradable nanoparticle alterations could improve the clinical effectiveness and applicability of biodegradable devices used in a variety of biological specialties. Biodegradable stents were developed to take the place of exposed metal stents because of the significant risk of in-stent restenosis when utilizing metal materials. Amazing testing is still required to obtain the perfect balance of the polymer, medication, and corruption rate in order to avoid using excessive or continuous force and to keep the integrity of the circulatory system after the implantation of stents46 .
Antioxidant Delivery
The cytotoxic effects of cells being exposed to engineered nanoparticles have been linked to oxidative pressure. The age of reactive oxygen species (ROS) and other natural processes might alter as a result of changes in the biological and physicochemical properties of nanoparticles after they enter the human body47 . Another type of nanoparticle, known as PVAX, has been developed by Kang’s team and was made from copolyoxalate that contained vanillyl alcohol (VA), a cell reinforcement that can be found in common spices. PVAX is broken down and releases VA, providing restraint and being hostile to apoptotic processes, when high quantities of H2O2 are present at the sites of ischemia/reperfusion (I/R). At the bottom of PVAX, VA and the H2O2-responsive peroxalate ester links are covalently united. PVAX has demonstrated effectiveness in treating a variety of I/R lesions, including rear appendage, liver, and heart I/R48,49.
Superoxide dismutase (SOD) and catalase (CAT) were successfully administered to the sore site by Andrabi et al.50 utilising biodegradable nanoparticles (nano-SOD/CAT) to protect mitochondria from oxidative stress, minimising accidental injury to the spinal cord51.
To track ROS and protect cells from apoptosis brought on by oxidative stress, Tapeinos et al.52 developed biodegradable PLGA microspheres enclosed in collagen type-I and MnO2 nanoparticles.
Conclusion
Creating new drug delivery systems can be facilitated by using NPs comprised of biocompatible and biodegradable polymers. These polymers’ biocompatibility, mechanical attributes, and low cytotoxicity make them suitable choices for drug delivery systems. Such NPs have always had the potential to function as an efficient medication delivery mechanism that can deliver a various medicinal substances and biomolecules to the targeted locations.
Challenges
A deeper comprehension of the various biological interaction mechanisms and particle engineering are still needed to maximize NPs as a delivery strategy. The hazardous effects of medications on healthy organs or body parts are lessened by manipulating the system over time. However, there is still potential to investigate more biodegradable polymers for cutting-edge biological applications. Due to its adaptable formulation, sustained release characteristics, nanoscale size, and biocompatibility with different cells and tissues in the body, biodegradable NPs, on the other hand, seem to be a potential drug delivery carrier.
Acknowledgements
The Head of the Chemistry Department at GLA University in Mathura, India, as well as the administration are to be thanked for allowing the author to use their facilities to produce this research.
Conflict of Interests
There are not any competing interests.
Funding Sources
References
- Petros, R. A.; DeSimone, J. M. Strategies in the Design of Nanoparticles for Therapeutic Applications. Nat. Rev. Drug Discov. 2010, 9 (8), 615–627.
- Alexis, F.; Pridgen, E.; Molnar, L. K.; Farokhzad, O. C. Factors Affecting the Clearance and Biodistribution of Polymeric Nanoparticles. Mol. Pharm. 2008, 5 (4), 505–515.
- Han, J.; Zhao, D.; Li, D.; Wang, X.; Jin, Z.; Zhao, K. Polymer-Based Nanomaterials and Applications for Vaccines and Drugs. Polymers (Basel). 2018, 10 (1), 31.
- Villemin, E.; Ong, Y. C.; Thomas, C. M.; Gasser, G. Polymer Encapsulation of Ruthenium Complexes for Biological and Medicinal Applications. Nat. Rev. Chem. 2019, 3 (4), 261–282.
- Panyam, J.; Labhasetwar, V. Biodegradable Nanoparticles for Drug and Gene Delivery to Cells and Tissue. Adv. Drug Deliv. Rev. 2003, 55 (3), 329–347.
- Carrasquillo, K. G.; Stanley, A. M.; Aponte-Carro, J. C.; De Jésus, P.; Costantino, H. R.; Bosques, C. J.; Griebenow, K. Non-Aqueous Encapsulation of Excipient-Stabilized Spray-Freeze Dried BSA into Poly (Lactide-Co-Glycolide) Microspheres Results in Release of Native Protein. J. Control. Release 2001, 76 (3), 199–208.
- Kumari, A.; Yadav, S. K.; Yadav, S. C. Biodegradable Polymeric Nanoparticles Based Drug Delivery Systems. Colloids surfaces B biointerfaces 2010, 75 (1), 1–18.
- Mahapatro, A.; Singh, D. K. Biodegradable Nanoparticles Are Excellent Vehicle for Site Directed In-Vivo Delivery of Drugs and Vaccines. J. Nanobiotechnology 2011, 9 (1), 1–11.
- Reis, C. P.; Neufeld, R. J.; Ribeiro, A. J.; Veiga, F. Nanoencapsulation I. Methods for Preparation of Drug-Loaded Polymeric Nanoparticles. Nanomedicine Nanotechnology, Biol. Med. 2006, 2 (1), 8–21.
- Fessi, H.; Puisieux, F.; Devissaguet, J. P.; Ammoury, N.; Benita, S. Nanocapsule Formation by Interfacial Polymer Deposition Following Solvent Displacement. Int. J. Pharm. 1989, 55 (1), R1–R4.
- Choi, C.; Chae, S. Y.; Nah, J.-W. Thermosensitive Poly (N-Isopropylacrylamide)-b-Poly ($\varepsilon$-Caprolactone) Nanoparticles for Efficient Drug Delivery System. Polymer (Guildf). 2006, 47 (13), 4571–4580.
- Kim, S. Y.; Lee, Y. M. Taxol-Loaded Block Copolymer Nanospheres Composed of Methoxy Poly (Ethylene Glycol) and Poly ($\varepsilon$-Caprolactone) as Novel Anticancer Drug Carriers. Biomaterials 2001, 22 (13), 1697–1704.
- Sinha, V. R.; Singla, A. K.; Wadhawan, S.; Kaushik, R.; Kumria, R.; Bansal, K.; Dhawan, S. Chitosan Microspheres as a Potential Carrier for Drugs. Int. J. Pharm. 2004, 274 (1–2), 1–33.
- Gan, Q.; Wang, T. Chitosan Nanoparticle as Protein Delivery Carrier—Systematic Examination of Fabrication Conditions for Efficient Loading and Release. Colloids Surfaces B Biointerfaces 2007, 59 (1), 24–34.
- Zillies, J. C.; Zwiorek, K.; Hoffmann, F.; Vollmar, A.; Anchordoquy, T. J.; Winter, G.; Coester, C. Formulation Development of Freeze-Dried Oligonucleotide-Loaded Gelatin Nanoparticles. Eur. J. Pharm. Biopharm. 2008, 70 (2), 514–521.
- Ofokansi, K.; Winter, G.; Fricker, G.; Coester, C. Matrix-Loaded Biodegradable Gelatin Nanoparticles as New Approach to Improve Drug Loading and Delivery. Eur. J. Pharm. Biopharm. 2010, 76 (1), 1–9.
- Abdullaeva, Z. Nanomaterials in Daily Life; Springer, 2017.
- Sharma, R.; Srivastava, N. Plant Mediated Silver Nanoparticles and Mode of Action in Cancer Therapy: A Review. Anti-Cancer Agents Med. Chem. (Formerly Curr. Med. Chem. Agents) 2021, 21 (14), 1793–1801.
- Goel, A.; Bhatia, A. K. Phytosynthesized Nanoparticles for Effective Cancer Treatment: A Review. Nanosci. \& Nanotechnology-Asia 2019, 9 (4), 437–443.
- Patel, S.; Singh, D.; Srivastava, S.; Singh, M.; Shah, K.; Chauahn, D. N.; Chauhan, N. S. Nanoparticles as a Platform for Antimicrobial Drug Delivery. Adv Pharmacol Pharm 2017, 5 (3), 31–43.
- Garg, P. Selective Preference of Antibody Mimetics over Antibody, as Binding Molecules, for Diagnostic and Therapeutic Applications in Cancer Therapy. Biointerface Res. Appl. Chem 2021, 11, 10765–10775.
- Perioli, L.; Pagano, C.; Ceccarini, M. R. Current Highlights about the Safety of Inorganic Nanomaterials in Healthcare. Curr. Med. Chem. 2019, 26 (12), 2147–2165.
- Gatto, F.; Bardi, G. Metallic Nanoparticles: General Research Approaches to Immunological Characterization. Nanomaterials 2018, 8 (10), 753.
- Gamucci, O.; Bertero, A.; Gagliardi, M.; Bardi, G. Biomedical Nanoparticles: Overview of Their Surface Immune-Compatibility. Coatings 2014, 4 (1), 139–159.
- Fam, S. Y.; Chee, C. F.; Yong, C. Y.; Ho, K. L.; Mariatulqabtiah, A. R.; Tan, W. S. Stealth Coating of Nanoparticles in Drug-Delivery Systems. Nanomaterials 2020, 10 (4), 787.
- Johnson, L.; Duschl, A.; Himly, M. Nanotechnology-Based Vaccines for Allergen-Specific Immunotherapy: Potentials and Challenges of Conventional and Novel Adjuvants under Research. Vaccines 2020, 8 (2), 237.
- Feng, X.; Xu, W.; Li, Z.; Song, W.; Ding, J.; Chen, X. Disease Immunotherapy: Immunomodulatory Nanosystems (Adv. Sci. 17/2019). Adv. Sci. 2019, 6 (17), 1970100.
- Pisani, A.; Pompa, P. P.; Bardi, G. Potential Applications of Nanomaterials to Quench the Cytokine Storm in Coronavirus Disease 19. Front. Bioeng. Biotechnol. 2020, 8, 906.
- Jena, A.; Behera, M.; Routray, C.; Kumar Biswal, S. Fabrication, Characterization and Antibacterial Study of Polyvinyl Alcohol/Cuprous Oxide Nanofluids and Polymer Nanocomposite Films. Orient. J. Chem. 2020, 36 (04), 713–719. https://doi.org/10.13005/ojc/360416.
- Balakrishnan, R.; Radhakrishnan, S. Investigation on Synthesis and Applications of Novel Polymeric Composites for the Removal of Heavy Metal Ion. Orient. J. Chem. 2020, 36 (04), 720–726. https://doi.org/10.13005/ojc/360417.
- Park, J.; Fong, P. M.; Lu, J.; Russell, K. S.; Booth, C. J.; Saltzman, W. M.; Fahmy, T. M. PEGylated PLGA Nanoparticles for the Improved Delivery of Doxorubicin. Nanomedicine Nanotechnology, Biol. Med. 2009, 5 (4), 410–418.
- Krishna, R.; Mayer, L. D. Multidrug Resistance (MDR) in Cancer: Mechanisms, Reversal Using Modulators of MDR and the Role of MDR Modulators in Influencing the Pharmacokinetics of Anticancer Drugs. Eur. J. Pharm. Sci. 2000, 11 (4), 265–283.
- Shafiei-Irannejad, V.; Samadi, N.; Salehi, R.; Yousefi, B.; Rahimi, M.; Akbarzadeh, A.; Zarghami, N. Reversion of Multidrug Resistance by Co-Encapsulation of Doxorubicin and Metformin in Poly (Lactide-Co-Glycolide)-d-$α$-Tocopheryl Polyethylene Glycol 1000 Succinate Nanoparticles. Pharm. Res. 2018, 35 (6), 1–13.
- Bae, Y. H. Drug Targeting and Tumor Heterogeneity. J. Control. release Off. J. Control. Release Soc. 2009, 133 (1), 2.
- Danhier, F.; Feron, O.; Préat, V. To Exploit the Tumor Microenvironment: Passive and Active Tumor Targeting of Nanocarriers for Anti-Cancer Drug Delivery. J. Control. release 2010, 148 (2), 135–146.
- Torchilin, V. Tumor Delivery of Macromolecular Drugs Based on the EPR Effect. Adv. Drug Deliv. Rev. 2011, 63 (3), 131–135.
- Sanna, V.; Singh, C. K.; Jashari, R.; Adhami, V. M.; Chamcheu, J. C.; Rady, I.; Sechi, M.; Mukhtar, H.; Siddiqui, I. A. Targeted Nanoparticles Encapsulating (-)-Epigallocatechin-3-Gallate for Prostate Cancer Prevention and Therapy. Sci. Rep. 2017, 7 (1), 1–15.
- Adjei, I. M.; Blanka, S. Modulation of the Tumor Microenvironment for Cancer Treatment: A Biomaterials Approach. J. Funct. Biomater. 2015, 6 (1), 81–103.
- Adams, G. P.; Schier, R.; McCall, A. M.; Simmons, H. H.; Horak, E. M.; Alpaugh, R. K.; Marks, J. D.; Weiner, L. M. High Affinity Restricts the Localization and Tumor Penetration of Single-Chain Fv Antibody Molecules. Cancer Res. 2001, 61 (12), 4750–4755.
- Javid, A.; Ahmadian, S.; Saboury, A. A.; Kalantar, S. M.; Rezaei-Zarchi, S.; Shahzad, S. Biocompatible APTES–PEG Modified Magnetite Nanoparticles: Effective Carriers of Antineoplastic Agents to Ovarian Cancer. Appl. Biochem. Biotechnol. 2014, 173 (1), 36–54.
- Peñaloza, J. P.; Márquez-Miranda, V.; Cabaña-Brunod, M.; Reyes-Ram\’\irez, R.; Llancalahuen, F. M.; Vilos, C.; Maldonado-Biermann, F.; Velásquez, L. A.; Fuentes, J. A.; González-Nilo, F. D.; others. Intracellular Trafficking and Cellular Uptake Mechanism of PHBV Nanoparticles for Targeted Delivery in Epithelial Cell Lines. J. Nanobiotechnology 2017, 15 (1), 1–15.
- Li, C.; Guo, C.; Fitzpatrick, V.; Ibrahim, A.; Zwierstra, M. J.; Hanna, P.; Lechtig, A.; Nazarian, A.; Lin, S. J.; Kaplan, D. L. Design of Biodegradable, Implantable Devices towards Clinical Translation. Nat. Rev. Mater. 2020, 5 (1), 61–81.
- Tyler, B.; Gullotti, D.; Mangraviti, A.; Utsuki, T.; Brem, H. Polylactic Acid (PLA) Controlled Delivery Carriers for Biomedical Applications. Adv. Drug Deliv. Rev. 2016, 107, 163–175.
- Lee, D.-H.; de la Torre Hernandez, J. M. The Newest Generation of Drug-Eluting Stents and Beyond. Eur. Cardiol. Rev. 2018, 13 (1), 54.
- Lih, E.; Kum, C. H.; Park, W.; Chun, S. Y.; Cho, Y.; Joung, Y. K.; Park, K.-S.; Hong, Y. J.; Ahn, D. J.; Kim, B.-S.; others. Modified Magnesium Hydroxide Nanoparticles Inhibit the Inflammatory Response to Biodegradable Poly (Lactide-Co-Glycolide) Implants. ACS Nano 2018, 12 (7), 6917–6925.
- Di Mario, C.; Ferrante, G. Biodegradable Drug-Eluting Stents: Promises and Pitfalls. Lancet (London, England) 2008, 371 (9616), 873–874.
- Shvedova, A. A.; Kagan, V. E.; Fadeel, B. Close Encounters of the Small Kind: Adverse Effects of Man-Made Materials Interfacing with the Nano-Cosmos of Biological Systems. Annu. Rev. Pharmacol. Toxicol. 2010, 50, 63–88.
- Manke, A.; Wang, L.; Rojanasakul, Y. Mechanisms of Nanoparticle-Induced Oxidative Stress and Toxicity. Biomed Res. Int. 2013, 2013.
- Lee, D.; Bae, S.; Hong, D.; Lim, H.; Yoon, J. H.; Hwang, O.; Park, S.; Ke, Q.; Khang, G.; Kang, P. M. H2O2-Responsive Molecularly Engineered Polymer Nanoparticles as Ischemia/Reperfusion-Targeted Nanotherapeutic Agents. Sci. Rep. 2013, 3 (1), 1–8.
- Andrabi, S. S.; Yang, J.; Gao, Y.; Kuang, Y.; Labhasetwar, V. Nanoparticles with Antioxidant Enzymes Protect Injured Spinal Cord from Neuronal Cell Apoptosis by Attenuating Mitochondrial Dysfunction. J. Control. Release 2020, 317, 300–311.
- Bae, S.; Park, M.; Kang, C.; Dilmen, S.; Kang, T. H.; Kang, D. G.; Ke, Q.; Lee, S. U.; Lee, D.; Kang, P. M. Hydrogen Peroxide-Responsive Nanoparticle Reduces Myocardial Ischemia/Reperfusion Injury. J. Am. Heart Assoc. 2016, 5 (11), e003697.
- Tapeinos, C.; Larrañaga, A.; Sarasua, J.-R.; Pandit, A. Functionalised Collagen Spheres Reduce H2O2 Mediated Apoptosis by Scavenging Overexpressed ROS. Nanomedicine Nanotechnology, Biol. Med. 2018, 14 (7), 2397–2405.

This work is licensed under a Creative Commons Attribution 4.0 International License.










