Vibrational Frequencies of Oxygen Difluoride Using Lie Algebraic Model
J. Vijayasekhar1*, N. Srinivas2 and K. Anil Kumar1
Department of Mathematics, GITAM (Deemed to be University), Hyderabad, India.
Corresponding Author E-mail: vijayjaliparthi@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/380126
Article Received on : 09-Dec-2021
Article Accepted on :
Article Published : 11 Feb 2022
Reviewed by: Dr. Mohammad Vakili
Second Review by: Dr. katanguru laxmi
Final Approval by: Dr. Ioana Stanciu
In one-dimensional Lie algebraic framework, we calculated the vibrational frequencies of oxygen difluoride (F2O) molecule in fundamental mode, higher overtones and their combinational bands.
KEYWORDS:Lie algebraic model; Oxygen difluoride; Vibrational frequencies
Download this article as:| Copy the following to cite this article: Vijayasekhar J, Srinivas N, Kumar A. K. Vibrational Frequencies of Oxygen Difluoride Using Lie Algebraic Model. Orient J Chem 2022;38(1). |
| Copy the following to cite this URL: Vijayasekhar J, Srinivas N, Kumar A. K. Vibrational Frequencies of Oxygen Difluoride Using Lie Algebraic Model. Orient J Chem 2022;38(1). Available from: https://bit.ly/3gFF1QY |
Introduction
Analysis of vibrational specta of small-size molecules is among the most challenging aspects of current studies in molecular physics. The emergence of novel experimental techniques to produce vibrational spectra of molecules in higher overtones, requires reliable theoretical models for their interpretation. In this paper we are concerned with the vibrational spectra of oxygen difluoride using Lie algebraic approach. In this model, vibrational Hamiltonian matrix describes the vibrational energies of the molecule. In 1991, one-dimesional Lie algebraic method applied for the study of vibrational frequencies of small size molecules by Iachello and co-researchers 1, 2 and then this method was subsequently improved in vibrational spectra of medium-size molecules 3-10.
Lie algebraic method 11, 12
The general Hamiltonian for n vibrational modes of oxygen difluoride (F-O and F-O) is
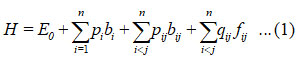
According to equation (1), b1 and b2 are invariant operators for uncoupled and coupled bonds respectively and are known as
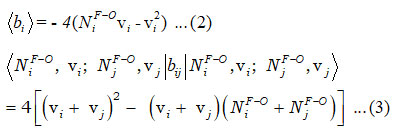
and the Majorana operator ƒij is used to express the local mode interactions in pairs and contains non-diagonal and diagonal matrix elements,
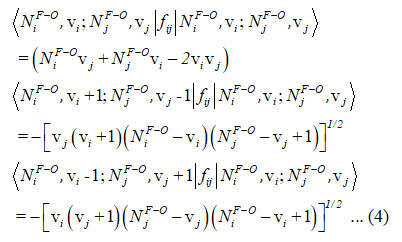
Hamiltonian for two stretching vibrations of oxygen difluoride molecule with C2v symmetry point group can be written as
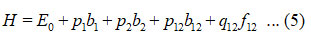
(cm– In equation (5), p1 , p2 , p12 and q12 1), are algebraic parameters (cm-1), estimated from the spectroscopic data. Two bonds (F-O) are equivalent in the oxygen difluoride; so, we consider as p1 = p2 = p, N1F‒O = N2F‒O = NF‒O in equations (2), (3) and (4).
The Hamiltonian matrix for the first two local oscillators is
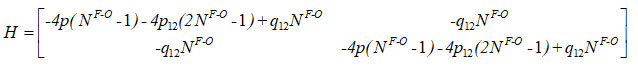
Results and Discussion
The energy equation (6) is used to find the initial value of the parameter p,
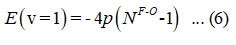
and the value for q12 determined from the relation,
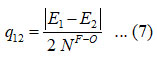
Where, E1, E2 are the oxygen difluoride, symmetric and antisymmetric vibrational frequencies, respectively. The oxygen difluoride vibrational frequencies were calculated using the Lie algebraic approach and the obtained results reported in (2) and table (3). The dimensionless Vibron number N1 for stretching and bending bonds of oxygen difluoride is determined using the following relation,
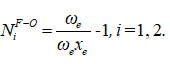
Where, ωe (= 1053.0138), ωe xe (=9.9194) are the vibrational harmonic and anharmonic (spectroscopic) constants, respectively for the bond F-O 13
Table 1: Algebraic parameters
| NF-O (stretching) |
106 |
| NO-F-O (bending) |
62 |
| p(stretching) |
-2.0940 |
| p(bending) |
-1.8893 |
| p12(stretching) |
0.0574 |
| p12(bending) |
0.4685 |
| q12(stretching) |
0.4575 |
| q12(bending) |
3.7177 |
Table 2: Fundamental vibrational frequencies (in cm-1)
|
Vibrational Mode |
Symmetry |
Observed [14] |
Calculated |
|
v1 (symmetric stretch) |
A1 |
928 |
928.0244 |
|
v2 (bend) |
A1 |
461 |
459.8062 |
|
v3 (antisymmetric stretch) |
B1 |
831 |
831.0344 |
Table 3: Higher overtone vibrational frequencies (in cm-1)
|
Overtone |
Vibrational mode |
||
|
symmetric stretch (A1) |
bend (A1) |
antisymmetric stretch (B1) |
|
|
1 |
2v1 (1847.133 ) |
2v2 (906.247) |
2v3 (1632.923) |
|
2 |
3v1 (2743.971) |
3v2 (1357.693) |
3v3 (2427.607) |
|
3 |
4v1 (3694.012) |
4v2 (1794.012) |
4v3 (3287.437) |
|
4 |
5v1 (4571.063) |
5v2 (2269.624) |
5v3 (4071.773) |
|
5 |
6v1(5513.453) |
6v2(2683.483) |
6v3(4870.003) |
|
6 |
7v1(6384.138) |
7v2(3172.896) |
7v3(5783.896) |
|
7 |
8v1(7353.148) |
8v2(3523.312) |
8v3(6561.324) |
|
8 |
9v1(8208.200) |
9v2(4004.915) |
9v3(7318.005) |
Table 4: Combinational bands (in cm-1)
|
Combinational band |
Calculated |
Combinational band |
Calculated |
|
v1 + v2 |
1389.933 |
2v1 + v2 |
2309.042 |
|
v1 + v3 |
1759.518 |
v2 + 2v2 |
1369.801 |
|
v2 + v3 |
1292.943 |
v2 + 2v3 |
2094.832 |
|
v1 + 2v1 |
2775.616 |
2v1 + v3 |
2678.626 |
|
v1 + 2v2 |
1836.374 |
v3 + 2v2 |
1739.384 |
|
v1 + 2v3 |
2561.406 |
v3 + 2v3 |
2464.416 |
Conclusion
In the table (2), the estimated fundamental vibrational frequencies were compared with the observed data. In table (3) and (4), vibrational frequencies upto the nineth harmonic and combinational bands upto third harmonic reported accordingly by the Lie algebraic method.
Conflict of Interest
There is no conflict of interest.
Funding Sources
There is no funding source.
References
- F. Iachello, S. Oss, R. Lemus, J. Mol. Spectrosc., 1991, 149 (1), 132-151. doi: 10.1016/00222852(91)90148-4
CrossRef - Iachello, F.; Oss S. J. Mol. Spectrosc. 1991, 146, 56-78. doi: 10.1016/0022-2852(91)90370-P
CrossRef - F. Iachello, S. Oss, J. Mol. Spectrosc., 1992, 153 (1–2), 225-239. doi: 10.1016/0022- 2852(92)90471-Y
CrossRef - A. Frank, R. Lemus, R. Bijker, F. Péilez Bernal, J.M. Arias, Ann. Physics, 1996, 252, 211-238.
CrossRef - Zhaochi Feng, Guang Xiong, Qihua Yang, Qin Xin & Can Li, Chin Sci Bull, 1999, 44, 1961–1964. doi:10.1007/BF02887118
CrossRef - V. Jaliparthi, M. Rao Balla, Spectrochim. Acta A, 2022, 264, 120289(1-8). doi:10.1016/j.saa.2021.120289
CrossRef - M. R. Balla, S. Venigalla, V. Jaliparthi, Acta Phys. Pol. A , 2021, 140 (2), 138-140. doi: 10.12693/APhysPolA.140.138
CrossRef - M. R. Balla, V. S. Jaliparthi, Polycyclic Aromatic Compounds , 2021. doi: 10.1080/10406638.2021.1901126.
CrossRef - M. R. Balla, V. S. Jaliparthi, Mol. Phys., 2021, 115 (5), e1828634. doi.org/10.1080/00268976.2020.1828634
CrossRef - J. Vijayasekhar, K. A. Kumar, N. Srinivas, Orient J Chem 2021, 37(6).
CrossRef - Iachello, F.; Levine, R. D. Oxford University Press, Oxford. 1995.
- Oss, S. Adv. Chem. Phys.1996, 93, 455-649.
- Irikura, K.K. J. Phys. Chem. Ref. Data., 2007, 36(2), 389-398.
CrossRef - Shimanouchi, T, Tables of Molecular Vibrational Frequencies Consolidated Volume I, National Bureau of Standards, 1972.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.









