Synthesis of Some New Selenonitrone Compounds
Batool Saleh Haddad
Department of Chemistry, College of Science, University of Basrah, Basrah, Iraq.
Corresponding Author E-mail: batoolsalih16@yahoo.com
DOI : http://dx.doi.org/10.13005/ojc/330615
The carbonyl selenonitrile and dicarbonyl diselane compounds were prepared by using the inorganic reagent such as potassium selenocyanate. New nitrone compounds were synthesized from the condensation of carbonyl group in the selenocarbonyl compounds with substituted N-phenylhydroxylamine and N-benzyl hydroxylamine. The resulting products were identified by physical properties such as melting point (M.p.), retardation factor (Rf) and color. Also compounds showed the expected data in identification techniques such as FTIR, 1H NMR,13 C NMR, mass spectroscopy and Elemental analysis (CHN). The results proved the validity of the expected chemical structures of synthesized compounds.
KEYWORDS:Nitrones; Potassium selenonitryl; Carbonyl selenonitrile
Download this article as:| Copy the following to cite this article: Haddad B. S. Synthesis of Some New Selenonitrone Compounds. Orient J Chem 2017;33(6). |
| Copy the following to cite this URL: Haddad B. S. Synthesis of Some New Selenonitrone Compounds. Orient J Chem 2017;33(6). Available from: http://www.orientjchem.org/?p=40870 |
Introduction
Interest in nitrone is due to their successful application, can be used as building blocks in the synthesis of various natural and biologically active compounds.Nitrone has been extensively investigated, because of their utility as versatile synthetic intermediates. Interest in nitrone also due to their successful application, can be used as building blocks in the synthesis of various natural and biologically active compounds. The synthesis of nitrone containing sulfur and nitrogen have been extensively studied [1, 2], while the synthesis of nitrone containing selenonitrile and diselenide analogues have not been appreciably investigated [3, 4]. This is mainly due to the easily decomposed of nitrone and organoselenium compounds in light and moisture. In this research [5, 6], several new carbonyl compounds containing selenonitrile group and diselenide [7] group were prepared. Diphenyl diselenide [(PhSe)2] is being carried out by the alkaline hydrolysis of substiuted phenylselenonitrile (the synthesis of phenylselenonitrile) is being carried out by the reaction between potassium cyanide and selenium metal [8]. For the preparation of nitrones, the most popular method is the condensation of aldehydes or ketones with N-monosubstituted hydroxylamines [9, 10, 11].Recently, organ-oselenium compound has gradually investigated because of their interesting reactivates and their pharmaceutical applications [12, 13]. The nitrone compounds are of marked interest because of their anti-tumor [14, 15], antibacterial [16] and of anti-cancer agents by intercalating DNA [17, 18, 19] with higher affinity and found it was easily metabolized with low toxicity [20]. The aim of the study : according to the literature, study has been done on nitrone compounds and organoselenium compounds separately. These two classes of compounds have been combined in this study.
Materials and Methods
FT.IR spectra were measured by an instrument called BRUKER and the range was between 375-4500 cm-1. 1H NMR spectra were measured by an instrument called BRUKER 300 MHz. The samples were recorded in DMSO solutions containing TMS as internal standard. Chemical shifts for all 1H and 13C NMR spectra were reported in δ units downfield from internal reference Me4Si. Elemental analyses (C, H and N) were obtained and Mass spectra were measured by instruments called MP- H- Vario Elemental analyzer and Agilent- O.T-LABRO, respectively.
All the carbonyl selenonitrile compounds being carried out by the reaction of the corresponding benzoyl chloride with potassium selenocyanate. Nitrone compounds were prepared from the condensation of carbonyl selenonitrile with N-phenyl hydroxylamine and N-benzyl hydroxyl amine compounds.
The general procedure of preparation carbonyl selenonitrile [21, 22]: To a stirred Solution of (0.8) KCN in absolute ethanol (50 ml), (1 gm.; 125 mole) of selenium metal was added. The reaction was in red color. After 3 hrs. of reflux, (206 ml 0.025 mole) of substituted benzoyl chloride was added and refluxed for 2 hr. The reaction was followed by TLC. The result was filtered, washed, dried and recrystallized from absolute ethanol.
The general procedure of preparation diorgano diselenide [23]: To a stirred solution of (0.28 gm; 1.02 mole) in 25ml absolute ethanol of carbonyl selenonytrile, a solution of (0.16 gm ,2.47 mole ) in 15 ml absolute ethanol sodium hydroxide was added. The mixture was stirred for 50 min at 40 o C. After one hour of reflux, the reaction was followed by TLC, the mixture reaction cooled to R .T. and filtered. The filtrate was acidified with 10% HCL. A red solid compound was obtained, washed with a small amount of benzene and dried. Recrystalization from a mixture of methanol and dichloro methane (1:4), gave a red solid compounds.
The General Procedure of synthesis nitrone [24-26]: In a 250 ml one-necked round bottomed flask (0.02 mole) of selenocarbonyl compounds were placed and a solution of 15gm of anhydrous CaCl2 with added 30 ml of absolute ethanol was added. The solution was stirred and warmed to 50 0C and a solution of (0.02mol) of [(N-phenyl hydroxylamine) in 30 ml of absolute ethanol was added, to synthesis the compounds ( N1, N2, N3 and N4) while the compound N5 was synthesized by added N-benzyl hydroxylamine] [25]. 3 drops of benzene sulfunic acid was added to the mixture. After the addition was completed the reaction mixture was kept with stirring in dark, overnight at room temperature. The reaction was followed by TLC, the mixture was cooled and then the precipitated was filtered by suction filtration and the product was recrystallized by abs. ethanol. All the synthesized compounds were characterized by FTIR, 1H NMR, 13C NMR, mass spectroscopy and Elemental analysis (CHN) and all the results were in agreement with the theoretical calculation. For example the spectra are represented in the figures (1 – 4) for compound (N4). Chemical structure and some physical properties of nitrones are represented in table (1).
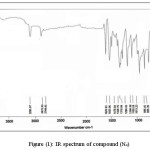 |
Figure 1: IR spectrum of compound (N4) Click here to View figure |
Click here to View figure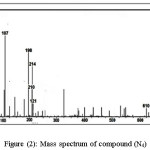 |
Figure 2: Mass spectrum of compound (N4) Click here to view figure |
 |
Figure 3: 1H NMR spectrum of compound (N4) Click here to View figure |
 |
Figure 4: 13C NMR spectrum of compound (N4) Click here to View figure |
 |
Table 1: Chemical structure and some physical properties of nitrones Click here to View table |
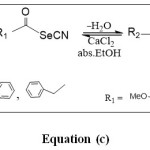
The carbonyl selenonitril compounds was prepared by the reaction of potassium cyanide with benzoylchloride as explain in equation (a).
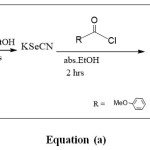 |
Formula Click here to View formula |
Dicarbonyl dislenide can be prepared by the alkaline hydrolysis of carbonyl selenonitrile compounds, as explain in equation (b).
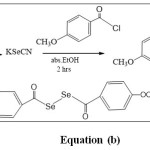 |
Formula Click here to View formula |
N-phenylhydroxylamine were synthesized by the reduced of nitro aromatic compounds with zinc dust and using water as a solvent while N-benzylhydroxylamine was prepared by the reaction between hydroxyl amine hydrochloride and benzyl chloride, in the presence of sodium carbonate.
Nitrones were synthesized by the condensation of substituted caronyl selenonitrile with N-phenylhydroxylamine and N-benzylhydroxylamine as explain in equation (c) and scheme (1).
 |
Formula Click here to View formula |
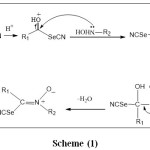 |
Scheme 1: The mechanism of synthesis nitrone compounds |
Results
The resulting data were obtained for the synthesized compounds from analysis data as following
N-(1, 1-(4-methoxyphenyl) selenonitrile methylene) aniline oxide (N1)
Yield: 55 %. M.p. 100-99 o C. FTIR ῡ /cm-1: 1600 (C=N), 1394 (C=C), 1377 (C-N), 588 (C-Se), 2101 (C N), 1199 (N-O), 2988 (C-H aliph), 3099 (C-H arom.). 1H NMR [300MHz, (DMSO), δ (ppm)]; 6.6 (4H-ph), m 7.9 (5H-ph), 3.6 (CH3). 13 C NMR [300MHz, (DMSO), δ (ppm)]; 50.6(CH3), 123.4, 123.6, 129.0, 129.7, 129.9, 149.0 (ph-N), 130.5, 130.6, 130.8,131, 13I.7, 160.4 (ph-O), 165 (C=N), 110 (C N). For C15H12N2OSe. (Found) 54.11 % C, 3.50 % H and 8. 55 % N. MS: m/z: 330.9 (M+).
N-(1, 1-(N, N-dimethylamino phenyl) selenonitrile methylene) aniline oxide (N2)
Yield: 57 %. M.p. 80-82 o C. FTIR ῡ /cm-1: 1605 (C=N), 1390 (C=C), 1380 (C-N), 589 (C-Se), 2110 (C N), 1188 (N-O), 2998 (C-H aliph), 3100 (C-H arom.). 1H NMR [300MHz, (DMSO), δ (ppm)]; 6.4 (4H-ph), m 7.7 (5H-ph), 2.9 (CH3). 13 C NMR [300MHz, (DMSO), δ (ppm)]; 50.6, 50.9 (CH3-N), 121.4, 121.6, 122.9, 123.3, 126.9, 147.0 (ph-N), 129.5, 129.9, 130.4, 130.5, 13I.8, 161.0 (ph-O), 162 (C=N), 115 (C N). For C16H15N3OSe. (Found) 55.30 % C, 4.30 % H and 12.30 % N. MS: m/z: 344(M+).
N-(1, 1-(N, N-dimethylamino phenyl) selenonitrile methylene)-4-methoxy aniline oxide (N3).
Yield: 70 %. M.p. 83-82 o C. FTIR ῡ /cm-1: 1590 (C=N), 1375 (C=C), 1385 (C-N), 590 (C-Se), 2111 (C N), 1180 (N-O), 2988(C-H aliph), 3110 (C-H arom.). 1H NMR [300MHz, (DMSO), δ (ppm)]; 6.7 (4H-ph-O), 7.9 (4H-ph-N), 3.9 (CH3-N), 2.7 (CH3-O). 13 C NMR [300MHz, (DMSO), δ (ppm)]; 49.8, 50.1 (CH3-N), 50.4 (CH3-O), 122.6, 122.8, 122.9, 124.3, 124.6, 166.9 (ph-O), 127.1, 127.9, 131.4, 131.1, 133.7, 147.7 (ph-N), 162.5 (C=N), 113 (C N). For C17H17N3O2Se. (Found) 54.57 % C, 4.90 % H and 11.22 % N. MS: m/z: 373.9(M+).
N, N’-(diselanediyl bis (4-methoxyphenyl methan-1-yl-1-ylidene)) bis (aniline oxide). (N4)
Yield: 61%. M.p. 90-92 o C. FTIR ῡ /cm-1: 1620 (C=N), 13710 (C=C), 1328 (C-N), 587 (C-Se), 1186 (N-O), 2872 (C-H aliph), 31285 (C-H arom.) .1H NMR [300MHz, (DMSO), δ (ppm)]; 3.0 (CH3), 7.3 (ph-N) 6.6 (ph-O). 13C NMR [300MHz, (DMSO), δ (ppm)]; 161.0, 160.5 (C=N), 30.3, 31.2 (CH3), 121.8, 121.9, 122.3, 122.9, 123.3, 123.9, 123.6, 124.5, 124.6, 124.7.166.3, 166.9 (ph-O), 124.8, 124.6, 124.9, 125.4, 125.9, 127.3, 127.9, 128.6, 129.0, 129.2, 147.0, 146.5 (ph-N),For C28H24N2O4Se2. (Found) 55.30 % C, 3.86 % H and 4.44 % N. MS: m/z: 610 (M+).
N, N’-(diselanediyl bis ((4-methoxyphenyl) methan-1-yl-1-ylidene)) bis(1-(3- methoxyphenyl) methanamine oxide) (N5)
Yield: 69%. M.p. 90-92 o C. FTIR ῡ /cm-1: 1589 (C=N), 1378 (C=C), 1378 (C-N), 581 (C-Se), 2976 1187 (N-O), (C-H aliph), 3108 (C-H arom.).1H NMR [300MHz, (DMSO), δ (ppm)]; 1.7 (CH3), 7.6 (ph-N) 7.3 (ph-O). 13C NMR [300MHz, (DMSO), δ (ppm)]; 161.0, 161.5 (C=N), 34.9, 35.1, 35.4, 35.9 (CH3), 60.4, 60.7 (CH2)121.5, 122.4, 122.9, 123.9, 124.3, 124.9, 125.6,125.5, 127.6, 128.7, 162.5, 163.9 (ph-O), 1298, 130.6, 130.9, 131.4, 1329, 133.3, 133.9, 134.0, 134.6, 134.8, 135.0, 146.5 (ph-N),For C32H28N2O6Se2. (Found) 55.38 % C, 4.20 % H and 4.12 % N. MS: m/z: 693.8 (M+).
Discussion
Five new nitrone compounds were synthesized by using the condensation method between carbonyl group (carbonyl selenonitryl and dicarbonyl diselenide) and N-(phenyl and benzyl)hydroxylamine group. Various substituted carbonyl selenonitrile compounds were synthesized by the reaction of potassium selenocyanate as inorganic reagent with benzoyl chloride. This method was easier than other methods to synthesis organoselenium compounds. The chemical structures of new selenonitrone compounds were characterized by identification methods.
All IR spectra of nitrone compounds showed the disappearance of (C=O) of carbonyl selenonitrile band in the region (1700) cm-1 and the appearance of (C=N), (N-O) and (C-N) bands in the regions (1589-1607) cm-1, (1180-1199) cm-1and (1377-1388) cm-1 respectively. The IR spectra of compounds (N4, N5) showed the disappearance of (C≡N) band due to the alkali hydrolysis of (C≡N) group to synthesis diselenide group, while the other synthesized nitrone compounds containing (C≡N) band in the region (2101-2111) cm-1. Mass spectroscopy gives the molecular ion and other fragments which indicated the structure of synthesized nitrone compounds. The nitrone compounds have been diagnosed by elemental analysis, and all the results were in agreement with the theoretical. The yield of products depends on the type of substituted groups. The presence of the both electron donating and drawing groups in the nitrone compound (N3) increases the yield of products more than the donating group as in compound (N1) or drawing group as in compound(N2). The diselane nitrone containing four donating groups as in compounds (N5) is more yield than the diselenide nitrone containing two donating groups as in compound (N4). Anhydrous CaCl2 was using as a drying agent to removed the water from the product and shifted the reaction to the right director.
Acknowledgment
I’m gratefully and thankful to Prof. Dr. Graham. J. Bodwell, college of science, University of Memorial NL-Canada for his help to complete this research in his lab.
References
- Al-Jassem, A. M; Majeed, N. N, J. Chemistry and Materials Research. 2014, 6, 100-108.
- Rosselin, M.; Choteau, F.; Zeamari, K.;Nash K.M., Das, A., Lauricella R., Lojou E., Tuccio B.,
CrossRef - Villamena F.A.and Durand G., Reactivities of Substituted alpha-Phenyl-N-tert-butyl Nitrones, J. Org. Chem. 2014, 79, 6615-6626.
- Keiko, N. A.; Funtikova, E.A.; Larina, L.I.; Sarapulova,G.I.; Mazhukin, D. G;T. V. RybalovaA, T.V.; Ya. Tikhonov, Y.A, J. Org. Chem. 2005, 41, 1421-1429.
- Nawaz, H..; Akhter, Z.; Yameen, S. ; Siddiqi, H. M; Mirza, B.;Rifat, A, J. Organomet. Chem. 2009, 694,2198- 2203.
CrossRef - Koketsu, M. ; Kanoh, K.: Ando, H.: Ishihara, H, J. Heteroatom Chem. 2006, 17, 88.
CrossRef - Torssell K.B.G., Nitrile Oxides, Nitrones and Nitronates in Organic Synthesis, Chapter 2, Wiley-VCH, Weinheim, Germany 2008.
- Sugie, S. ; Tanaka, T, J. Health science. 2000, 46, 422-425.
CrossRef - Wirth, T.; Organoselenium Chemistry: Modern Developments in Organic Synthesis; Springer: Berlin, 2000
CrossRef - Zink, O ; Nesvadba, P, J. Macromolecules. 2000, 33, 8106-8108.
CrossRef - Voinov, M. A. ; Grigor’ev, I. A, J. Organometallics. 2007, 26, 1607-1615.
CrossRef - Mobinikale, A ; Kalate, Z, J. Turk. chem. 2005, 29, 147.
- Hafez, A. M.; Taggi, A. E, J. Am. Chem. Soc. 2002, 124, 6626.
CrossRef - Lipsky, B.A.; Berendt, A.R.; Deery, H.G.; Embil, J.M.; Joseph, W.S.; Karchmer, A.W.; Lefrock, J.L.; Lew, D.P.; Mader, J.T.; Norden, C. Diagnosis and treatment of diabetic foot infections. Clin. Infect. Dis. 2004, 39, 885–910.
CrossRef - Koketsu, M; Ishihara, H, J. Org. Chem. 2003, 7, 175.
- Torssell K.B.G., Nitrile Oxides, Nitrones and Nitronates in Organic Synthesis, Chapter 2, Wiley-VCH, Weinheim, Germany, 2008.
- Chandrasekar, K.;.Malathi,R, J. Clin. Microbiol. 2000, 38, 3905.
- Wang, Y.; Larrik, J , Antioxidant Nitroxides and Nitrones as Therapeutic Agents, US Patent No.6852889, February 08, 2005.
- Sambasiva Rao, P.; C. Kurumurthy, C, J. Eur. Med. Chem. 2014, 80, 184-191.
CrossRef - Ramadan, A. A, J. Arch. Farm. Chem. Life Sci. 2006, 339, 242-249.
CrossRef - Floyd, R. A.; Kopke, R. D, J. Free Radic. Biol. Med. 2008, 45, 1361–1374.
CrossRef - Tarantelli, T.; Pecile, C, J. Org. Chem.1962, 52, 75-79
- Michael, J. P.; Brent, R. C, J. Org. Chem. , 1978, 43 (12), 2320–2323.
CrossRef - David, M. W.; Stuart,W. H, J. Org. Chem.1980, 45, 1924-1930.
CrossRef - Padungros, P, Practical Synthesis of Aromatic Dithiocarbamates. Synth Commun. 2014, 44, 2336- 2343.
CrossRef - Braslau, R.; Obryan, G, J. Synthesis. 2005, 79, 1496-1506.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.









