Effect of Electrolytic Bath Temperature on Magnetic and Structural Properties of Electrodeposited NiFeW Nano Crystalline Thin Films
Department of Physics, Government Arts College, Udumalpet-642126, Tamil Nadu, India.
Corresponding Author E-mails: kanagarajbu @gmail.com
DOI : http://dx.doi.org/10.13005/ojc/330624
Nickel iron films are vitally used in new generation recording heads, designing of micro electro mechanical devices and magnetic random access memory. The addition of tungsten (W) can enhance magnetic and mechanical properties of NiFe thin films. Electrodeposited Ni-Fe-W films were prepared at different temperature (30, 50, 70 and 90˚C) and they were subjected to morphological, structural, magnetic and mechanical characterization analysis. Nickel content was maximum as 61.41 wt% at 90˚C temperature. The tungsten content increased when electrolytic bath temperature was increased. Ni-Fe-W films were bright and uniformly coated on the surface. Also the deposits of Ni- Fe- W films were in nano scale and the average crystalline size was around 22 nm. Thin films prepared at high temperature exhibited a high saturation magnetization and low coercivity.The micro hardness of Ni-Fe-W was 221 VHN at 90˚C
KEYWORDS:Electrodeposition; Electrolytic Bath; Crystalline Size; VSM; Ni-Fe; X-ray Diffraction; VHN; SEM
Download this article as:| Copy the following to cite this article: Kanakaraj M, Ramalingam H. B. Effect of Electrolytic Bath Temperature on Magnetic and Structural Properties of Electrodeposited NiFeW Nano Crystalline Thin Films. Orient J Chem 2017;33(6). |
| Copy the following to cite this URL: Kanakaraj M, Ramalingam H. B. Effect of Electrolytic Bath Temperature on Magnetic and Structural Properties of Electrodeposited NiFeW Nano Crystalline Thin Films. Orient J Chem 2017;33(6). Available from: http://www.orientjchem.org/?p=39813 |
Introduction
The chemical, mechanical and thermal electrochemical interaction of materials is initiated from materials surface [1]. Therefore, surface is the most important engineering part of a material. The role of technologies which modifying surface in the production and manufacturing process is predictable [2-4].Also the surface technology has vital role in production process. Electrodeposition is an electrochemical process and an old method which applied for surface structure modification [5,6]. Nickel and its alloys have important advantages such as good wear and corrosion resistance .So deposition of nickel are needed to improve wear and corrosion resistance in the industry, repairing eroded metals, to improve magnetic properties, to change the dimensions of small metal pieces and preparing organic coating. NiFeW, NiCoW, and NiW are most commonly used magnetic thin film materials in MEMS and NEMS [7-10]. The electroplating of NiFeW thin film is used to obtain enhanced electrical conductivity, good soft magnetic characters, special optical properties and improved corrosion resistance [11-14]. Electrodeposition method is widely used in MEMS,NEMS, communication, optical and sensors industries [15]. In this study, the effects of different temperatures on characters of NiFeW alloy thin films are analysed. This paper explains the preparation and characterization of electroplated Ni-Fe-W thin films.
Experimental Part
The electroplated NiFeW alloy films were prepared for different temperature 30, 50, 70 and 90 o C. The time duration of deposition process was 15 minutes. In this investigation, copper and stainless steel substrates acted as cathode and anode respectively with 1.5 cm x 7.5 cm. dimension [16-19]. The NiFeW thin films were prepared from electrolytic solution which contain Ferrous Sulphate (10 g/l), Nickel sulphate (30 g/l), Sodium tungstate (15 g/l), Ammonium Sulphate (40 g/l) , Boric acid(10 g/l) and Saccharin(10 g/l) [20-21].The pH value of electrolytic solution was fixed as 6.0 by mixing ammonia solution and electroplating process was carried out with current density 3 mA/cm2 . The copper or cathode was carefully removed from the bath after 15 minutes and dried for few minutes [22-23]. The surface nature of Ni- Fe- W films was characterized by Scanning Electron Microscope .The elemental content of deposits of films was examined by Energy-dispersive X-ray Spectroscopy and crystal structure of deposits was analyzed by X-ray diffraction study. The micro hardness of films was measured by Vickers Hardness Test. The important magnetic properties of deposits are saturation magnetization and coercivity .Analysis of thin films with Vibrating Sample Magnetometer gives magnetic properties.
Results and Discussion
Composition of Ni Fe W films
Energy-dispersive X-ray analyser (EDAX) gives elemental composition of deposits. Table 1 shows weight percentage of Fe, Ni and W at different electrolytic bath temperature by EDAX analysis. From results, films prepared at 90˚C have high content of tungsten. The highest ferrous content of 22.67 wt% was found at electrolytic temperature of 30˚C. While electrolytic temperature increases, the content of nickel and tungsten also increase. The higher nickel content of 61.41 wt% was found at 90˚C temperature.
Table 1: EDAX analysis of thin films
|
S. No |
Temperature |
Fe Wt% |
Ni Wt% |
W Wt% |
|
1. |
30˚C |
22.67 |
48.52 |
28.81 |
|
2 |
50˚C |
18.12 |
52.73 |
29.15 |
|
3 |
70˚C |
11.38 |
57.29 |
31.33 |
|
4 |
90˚C |
05.33 |
61.41 |
33.26 |
Morphological Study of Nifew films
The surface structure of Ni- Fe- W thin films with temperature 30, 50, 70 and 90˚C were analysed by Scanning Electron Microscope (SEM) images and they are shown in Fig 1. The thin films are bright and uniformly coated on the surface. They are crack free by appearance.
 |
Figure 1: Ni FeW films – SEM images at (a) 30˚C (b) 50˚C (c) 70˚C (d) 90˚C Click here to View figure |
Structural Analysis of Ni Fe W films
Crystal structure of Ni- Fe- W alloy films was analyzed by powder X-ray diffraction (XRD). Fig 2 shows diffraction patterns of Ni- Fe- W films prepared with different temperature. The occurrence of sharp peaks in X-ray diffraction pattern exhibit crystalline nature of deposits. The XRD patterns of all samples deposited at 30, 50, 70, and 90˚C are being indicative of the (111), (200) and (220).Also XRD result reveal the existence of FCC type crystal. The particle sizes of Ni- Fe- W deposits are 27.15 nm, 22.18nm, 20.72nm and 17.28 for the bath temperature 30, 50, 70, and 90 ˚C respectively. So it is concluded that crystal size of thin film deposits decreases by increasing electrolytic temperature. Also deposits of thin films reveals nano scale and average crystalline size is around 22 nm. When bath temperature increases, strain value increases from 1.333 x 10-3 to 2.095 x 10-3 dislocation density increases from 13.57 x 1014 / m2 to 33.49 x 1014 / m2.
 |
Figure 2: NiFeW films – XRD patterns at (a) 300C (b) 500C (c) 700C (d) 900C Click here to View figure |
The crystal size, strain value and dislocation density of Ni-Fe-W alloy films are shown in table 2. The crystalline size of deposits decreases due to onset orientation of crystals while bath temperature increases. Figure 3 shows that crystal size reduces with increasing bath temperature.
Table 2: Structural characteristics of Ni-Fe-W alloy thin films
|
S.No |
Bath Temperature (˚C) |
2θ (deg) |
d (A0) |
Particle Size(D) (nm) |
Strain (10-3) |
Dislocation Density (1014 / m2) |
|
1 |
30 |
50.83 |
1.783 |
27.15 |
1.333 |
13.57 |
|
2 |
50 |
50.76 |
1.792 |
22.18 |
1.632 |
20.33 |
|
3 |
70 |
51.13 |
1.818 |
20.72 |
1.747 |
23.29 |
|
4 |
90 |
51.02 |
1.825 |
17.28 |
2.095 |
33.49 |
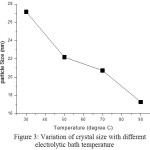 |
Figure 3: Variation of crystal size with different electrolytic bath temperature Click here to View figure |
Mechanical Properties of Ni Fe W films
Micro hardness measurement of deposits was done by Vickers hardness tester. The hardness values of thin films prepared for temperature 30, 50, 70 and 90 o C are 114,139,187,221 VHN respectively. So hardness test shows that the micro hardness increases by increasing electrolytic bath temperature due to lower stress associated with thin films. Fig 4 shows the variation of hardness with increasing bath temperature.
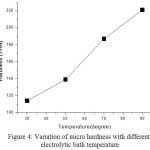 |
Figure 4: Variation of micro hardness with different electrolytic bath temperature Click here to View figure |
Magnetic Properties of Ni Fe W films
The important magnetic characters coercivity, saturation magnetization, retentivity and Squareness of NiFeW alloy films were obtained by VSM and they are given in table 3.The hysteresis curves for thin film NiFeW with various temperature are shown in figure 5.The elemental composition of deposits, applied potential, additive materials, temperature and electrolytic bath are the deciding factors of thin film magnetic properties.
When electrolytic temperature is changed as 30˚C, 50˚C, 70˚C and 90˚C, the magnetization increases from 38.713 x 10-3 emu/cm2 to 152.824 x 10-3 emu/cm2 .Also when temperature is increased from 30˚C to 90˚C, the coercivity decreases from 220.634 G to 121.82 G. From result of vibrating sample magnetometer, it is concluded that films obtained at higher temperature reveals high value of magnetization and low value of coercivity. The enhanced value of saturation magnetization and reduced value coercivity promote magnetic properties of alloy coating.
Table 3: Magnetic Properties of NiFeW deposits
|
S.No |
Bath Temperature (˚C) |
Coercivity Hs (G) |
Magnetization Ms (emu/cm2) 10-3 |
Retentivity Mr (emu/cm2) 10-3 |
Squareness S(Mr/ Ms) |
|
1 |
30 |
220.63 |
38.713 |
3.4703 |
0.089642 |
|
2 |
50 |
163.16 |
87.216 |
2.1315 |
0.024439 |
|
3 |
70 |
144.14 |
108.715 |
1.1760 |
0.010817 |
|
4 |
90 |
121.82 |
152.824 |
5.1027 |
0.033389 |
 |
Figure 5: Hysteresis curves for NiFeW film at electrolytic bath temperatures (a) 30˚C (b) 50˚C (c) 70˚C (d) 90˚C Click here to View figure |
The magnetization and other important magnetic property coercivity can be changed by decreasing the grain size of deposits .The film stress is reduced by increasing bath temperature. The Ni-Fe-W thin films prepared at 90˚C have lower coercivity and higher magnetization due to low stress formation during deposition. So it is concluded that soft magnetic nature of NiFeW thin films are enhanced by increasing bath temperature.
Conclusion
The NiFeW alloy thin films with different temperature were prepared by electrodeposition by maintaining current density 3 mA/cm2 and pH of solutions 6.0. The thin films are bright and uniformly coated on the surface. The XRD result reveal the existence of FCC type crystal .The crystalline size of deposits decreases due to online orientation of crystal while increasing bath temperature. The hardness increases with increasing bath temperature due to lower stress associated with thin films. When bath temperature is increased from 30˚C to 90˚C, the magnetization increases from 38.713 x 10-3 emu/cm2 to 152.824 x 10-3 emu/cm2 .Also when temperature is increased from 30˚C to 90˚C, the coercivity decreases from 220.634 G to 121.82 G. This is due to nano crystalline structure of deposits. The addition of tungsten (W) with NiFe alloy in electroplating can enhance its magnetic, mechanical and structural properties and this alloy films can be used in NEMS,MEMS and memory devices.
References
- Brenner; Electrodeposition of alloys Principle and practices, 1963, Vol. I and II, Academic Press, NewYork.
- Iwasaki, S.; Nakamura, Y.; An analysis for the magnetization mode for high density magnetic recording, Journal of Magnetism and Magnetic Materials, 1977, 200, 634-648.
- Osaka, T.; A soft magnetic CoNiFe film with high saturation magnetic flux density and low coercivity, Nature, 1998, 392, 796 – 798.
CrossRef - Nosang, V.; Park, D.Y.; Yoob, B.Y.; Paulo, T.A.; Development of electroplated magnetic materials for MEMS, Journal of Magnetism and Magnetic Materials, 2003, 265 , 189-198.
CrossRef - Ghorbani, M.; Dolati , A.; Afshar, A.; Electrodeposition of Ni–Fe Alloys in the Presence of Complexing Agents, Russ. J. Electrochem, 2002, 38, 1173-1177.
CrossRef - Yan ,X.H.; Sun ,J.Q.; Wang, Y.W; Yang, J.F.; A Fe-promoted Ni-P amorphous alloy catalyst (Ni-Fe-P) for liquid phase hydrogenation of m- and p chloro nitro benzene, Journal of Molecular Catalysis A: Chemical, 2006, 252,17–22.
CrossRef - Celine, R.; Patrick, F.; Electrodeposition of thin films and nanowires Ni–Fe alloys, study of their magnetic susceptibility, J, Mater Sci., 2011, 46, 6046-6053.
CrossRef - Fernandez ,G.V; Grundy, P.J.; Vopson, M.M.; Control and analysis of grain Size in sputtered NiFe thin films, J. Phys, Condens. Matter, 2013, 1(1),6-9.
CrossRef - Thangaraj ,N .; Tamilarasan,K.; Sasikumar,D.; Effect of Phosphorous Acid on the Ferrous Tungsten Phosphorous Magnetic Thin Film, International Journal of ChemTech Research, 2014, 6, 384-390.
- Myung, N.; A Study on the Electrodeposition of NiFe Alloy Thin Films Using Chronocoulometry and Electrochemical Quartz Crystal Microgravimetry, Bull. Korean Chem. Soc., 2001, 22, 994-998.
- Meng, X.F.; Li. D.H.; Shen, X.Q.; Liu, W.; Preparation and magnetic properties of nano-Ni coated cenosphere Composites, Applied Surface Science, 2010, 256, 3753–3756.
CrossRef - Wang, S.L.; Studies of electroless plating of Ni-Fe-P alloys and the influences of some deposition parameters on the properties of the deposits, Surface and Coatings Technology, 2004, 186, 372–376.
CrossRef - Hamid, Z.A.; Electrodeposition of Cobalt- Tungsten Alloys from Acidic Bath Containing Cationic Surfactants, Materials Letters,2003, 57, 2558.
CrossRef - Kannan, R.; Kanagaraj, R.; Ganesan, S.; Influence of Tri Sodium Citrate Bath Concentration on the electrodeposition of NiFeWS Thin Films, Journal of Ovonic Research, 2013,9, 45-54.
- Baonkeup, K.; Bongyoung, Y.; Electrodeposition of low-stress NiFe thin films from a highly acidic electrolyte, Surf. Coat. Technol, 2010, 205, 740 – 744
CrossRef - Kannan, R.; Ganesan, S.; Selvakumari ,T.M.; Synthesis and characterization of nano crystalline NiFeWS thin films in diammonium citrate bath, Digest journal of nanomaterials and biostructures, 2012,7, 1039-1050.
- Balachandran, R.; Yow, H.K.; Ong, B.H.; Tan, K.B.; Anuar ,K.; Wong ,H.Y.; Surface morphology and electrical properties of pulse electrodeposition of NiFe films on copper substrates in ultrasonic field, Int. J. Electrochem. Sci., 2011, 3564-3579.
- Esther, P.; Joseph Kennady,C.; Effect of sodium tungstate on the properties of Electrodeposited nanocrystalline Ni-Fe-W films, Journal of Non Oxide Glasses., 2010, 1, 35-44.
- Sulztanu, N.; Fbrinza, J.; Electrodeposited Ni-Fe-S films with high resistivity for Magnetic recording devices, J. Optoelectron Adv Mat., 2004, 6, 641- 645.
- Sam, S.; Manavalan, G.; Guittoum ,A.; Gabouze, N.; Djebbar, S.; Electrodeposition of NiFe films on Si(100) substrate, Surf. Sci., 2007, 601, 4270 – 4273.
CrossRef - Kannan, R.; Ganesan, S.; Selvakumari, T.M.; Structural and Magnetic properties of electrodeposited NiFeWS thin films, Optoelectronics and advanced materials-Rapid Communication, 2012, 3-4, 383-388.
- An, Z.G.; Zhang, J.J.; Pan, S.L.; Fabrication of glass/Ni-Fe-P ternary alloy core/shell composite hollow Microspheres through a modified electroless plating process, Applied Surface Science, 2008, 255, 2219–2224.
CrossRef - Emerson, R.N.; Kennady, C.J.;.Ganesan, S.; Effect of Organic additives on the Magnetic properties of Electrodeposition of CoNiP Hard Magnetic Films, Thin solid films, 2007,515, 3391-3396.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.










