Corrosion Processes on Iron Electrode in Presence of Heterocyclic Amines
Vadim Nikolaevich Statsyuk1, Sauyk Ait3, Lidiya Alekseevna Fogel1, Murat Zhurinovich Zhurinov1 and Aleksei Aleksandrovich Abrashov2
1D.V. Sokolsky Institute of Fuel, Catalysis and Electrochemistry, Almaty, Republic of Kazakhstan.
2School of Chemical Engineering, Kazakh-British Technical University, Almaty, Republic of Kazakhstan.
3D.Mendeleev University of Chemical Technology of Russia, Moscow, Russia.
Corresponding Author E-mail: sauik-ait@mail.ru
DOI : http://dx.doi.org/10.13005/ojc/330620
A relationship between the adsorption capacity of 2,2/ -dipyridyl, 4,4/ -dipyridyl heterocyclic amines, 1,10-phenanthrolin and Fe (dipy)32+ complex on mercury with the corrosive activity of these compounds on the iron electrode by cyclic voltammetry was established. The use of this method made it possible to establish a regularity between the process of the iron electrode ionization and the appearance of a current maximum due to the electroreduction of corrosion products on the cathode volt-ampere curves. It was found that 4,4/ -dipyridyl, isomer of 2,2/ -dipyridyl, has a stronger interaction of π-electrons with the surface of mercury and a flat arrangement on the surface of the electrode, which contributes to its more corrosive activity. It should be noted that 4,4/ -dipyridyl is able to form polymer low solubility compounds with metal ions, whereas 2,2/-dipyridyl and 1,10-phenanthroline form chelate soluble complexes with Fe (II). Unlike molecules of 2,2/ -dipyridyl, which inhibit corrosion processes, complex compounds Fe (dipy)32+, being corrosion products, lead to increased corrosion.
KEYWORDS:Anodic Dissolution; Cathodic Maximum; Cyclic Voltammetric Curves; Initial Potential; Potential Sweep Rate
Download this article as:| Copy the following to cite this article: Statsyuk V. N, Ait S, Fogel L. A, Zhurinov M. Z, Abrashov A. A. Corrosion Processes on Iron Electrode in Presence of Heterocyclic Amines. Orient J Chem 2017;33(6). |
| Copy the following to cite this URL: Statsyuk V. N, Ait S, Fogel L. A, Zhurinov M. Z, Abrashov A. A. Corrosion Processes on Iron Electrode in Presence of Heterocyclic Amines. Orient J Chem 2017;33(6). Available from: http://www.orientjchem.org/?p=40392 |
Introduction
For conducting corrosion tests it is necessary to have information about the course of both cathode and anode processes. The most complete information can be obtained by using cyclic voltammetry allowing to establish the relationship between ionization processes and cathodic processes of their recovery.
Based on the fact that many organic surfactants lead to inhibition of corrosion processes, it was expedient to use the proposed voltammetric method for evaluating the inhibitory ability of a number of heterocyclic compounds.
Gatos [1], who studied the adsorption of amines on mercury, found that the surface activity of amines on mercury changes in the same way as the inhibitory effect of these compounds in acid corrosion. The greatest inhibitory effect is possessed by nitrogen-containing amine compounds. The inhibitory effect of these compounds depends on the size and structure of organic molecules, the nature of their adsorption on the metal surface, and the protective properties of the formed or adsorbed films. The potential scale used by Andropov [2] makes it possible to establish a clear quantitative dependence between the surface activity of a number of compounds on mercury and their inhibitory properties in acid corrosion. The established regular dependence makes it possible to determine the inhibitory effect of the compounds on the basis of their electro capillary curves. According to Hakerman [3,4], the inhibitory properties of organic compounds are determined by the electron density at the atom, which is a special reaction center. With the increase of the electron density near the center adsorption (chemisorption) between the inhibitor and the metal are enhanced. It has been established [3,4] that the protective properties of pyridine derivatives increase with increasing electron density at the nitrogen atom in the sequence of pyridine ˂ 3-picoline ˂ 2-picoline ˂ 4-picoline. Along with the enhancement of the electron density at the nitrogen atom, aromatic substances have the ability to be adsorbed due to π-electrons, being oriented flat to the surface of the mercury electrode [5].
It was of interest to establish a relationship between the adsorption capacity of heterocyclic amines: 2,2/ -dipyridyl, 4,4/ -dipyridyl, 1.10-phenanthroline and Fe (dipy)32+ on mercury with the corrosive activity of these compounds on an iron electrode using the voltammetric method.
Materials and Methods
Cyclic current-voltage curves were obtained at the potentiostat-galvanostat AUTOLAB (Netherlands) in a sealed cell, thermo stated at 250C with a LOIPLT-105 (Russia) thermostat. The working electrode was a disc, compact iron electrode (Ст.3) with a visible surface of 0.03 cm2. Rotation of this electrode was carried out on the AUTOLAB RDE-2 (Netherlands). To update the working electrode surface using abrasive paper grade MIRKA 2000 (Finland) with the electrode rotating at 2500 rpm followed by washing with distilled water. Before reproduction the current-voltage curves, the electrode was kept in a solution of sodium sulfate at E = -1.1 V for 5 minutes. As an auxiliary electrode, a platinum wire with a visible surface exceeding the working electrode surface more than 100 times was used. The potentials presented in the article were measured with respect to silver chloride electrode firm Metrohm (Switzerland) with a potential of 198 mV relative hydrogen electrode. The potential sweep rate was 15 mV / s, with the exception of cyclic volt-ampere curves obtained at different potential sweep rates. Deaeration of the solution in the cell was carried out with an inert gas of argon “х.ч”. Distilled water was used to prepare the solutions. The reagents Na2SO4, 2,2/-dipyridyl and 4,4/-dipyridyl used in the work had the mark “х.ч” and “ос.ч “, 2,2 / -dipyridyl and 4,4 / -dipyridyl of the company ‘Reanal’ Hungary, 1,10-phenanthroline of the company “Chemapol” Czech Republic.
Results and Discussion
Figure 1 shows cyclic voltammetric curves on an iron electrode in solutions of 0.3 M Na2SO4 and 0.3 M Na2SO4 + 5.7·10-4 M 2,2/ -dipyridyl at different potential sweep rates, which varied from 5 to 300 mV / s. Cyclic volt-ampere curves were taken as follows: first, a cathode part of the current-voltage curves was obtained from -0.35 to -1.1 V, after reaching the potential E = -1.1 V, the direction of the potential sweep was reversed and the anode part of the current-voltage curves was obtained, reaching a value of the initial potential E = -0.35 V. Comparing the current-voltage curves in Fig. 1 it should be noted that in the presence of 5.7·10-4 M 2,2/ -dipyridyl, the current value of the maximum (A) is significantly less than in the solution not containing 2,2/ -dipiridyl. In this case, it should be noted that the potential of the maximum (A) at the same potential sweep rate does not depend on the content of 2,2/ -dipyridyl in the solution.
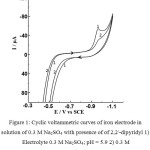 |
Figure 1: Cyclic voltammetric curves of iron electrode in solution of 0.3 M Na2SO4 with presence of of 2,2/-dipyridyl 1) Electrolyte 0.3 M Na2SO4; pH = 5.9 2) 0.3 M Na2SO4 + 5.7·10-4М 2,2/-dipyridyl Click here to View figure |
A relationship is established between the current value of the maximum (A) on the cathode curve and the ionization current on the anode curve at a constant value of the initial potential E = -0.35 V. Increase of the ionization current leads to increase of the cathode current of the maximum (A). In other words, increase in the degree of the iron electrode filling with ionization products leads to increase of the cathode current of the maximum (A), caused by the electroreduction of this products.
The presence in the solution of 0.3 M Na2SO4 + 5.7·10-4 M 2,2/ -dipyridyl leads to a decrease of the current maximum (A). This is due to the adsorption of 2,2/ -dipyridyl on the iron electrode surface, which prevents its ionization. The decrease in the degree of filling of the iron electrode surface by the products of its ionization due to the adsorption of 2,2/ -dipyridyl leads to decrease of the current value of the maximum (A) on the cathode volt-ampere curves.
The change of the current value of the maximum (A) from the potential sweep rates (V) in the coordinates lgI – lgV (Figure 2) is shown for solutions of 0.3 M Na2SO4 (curve 1), 0.3 M Na2SO4 + 1.9·10-4 M Fe (dipy)32+ (curve 2), 0.3 M Na2SO4 + 5.7·10-4 M 2,2/ -dipyridyl (curve 3), 0.3 M Na2SO4 + 5.7·10-4 M 1.10 / -phenanthroline (curve 4).
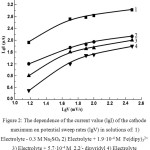 |
Figure 2: The dependence of the current value (lgI) of the cathode maximum on potential sweep rates (lgV) in solutions of: 1) Click here to View figure |
Electrolyte – 0.3 M Na2SO4 2) Electrolyte + 1.9·10-4 М Fe(dipy)32+ 3) Electrolyte + 5.7·10-4 М 2,2/- dipyridyl 4) Electrolyte + 5.7·10-4 М 1,10- phenanthrolin
From Fig. 2 it follows that the lgI – lgV dependence for all the solutions has a similar character. So, at a potential sweep rate (V) from 15 to 50 mV / s, the value lgI / lgv ~ 1.3, while at V ˃ 50 mV / s this value is equal to ~ 0.3. It is established that at a potential sweep rate of V ˃ 300 mV / s, the current value of the maximum (A) remains practically constant.
The observed events are apparently related to the lifetime of the particles formed. At low potential sweep rates, the particles formed during the ionization of the iron electrode succeed in breaking down. As the sweep speed of the potential increases, these particles do not have time to undergo destruction. Under these conditions, a sharp increase of the current value of the cathode maximum (A) is observed. A further increase of the potential sweep rate leads to the fact that the proportion of particles that have not undergone destruction increases more and more, and finally, when practically all the particles that are formed do not undergo destruction, the current value of the maximum reaches a constant value.
The destruction of the formed compounds on the surface of the iron electrode at active dissolution potentials is facilitated not only by a decrease in the potential sweep rates, but also by an increase in the rotation speed of the disk electrode. Thus, at a rotational speed of the disk electrode w = 80 rpm, the current maximum at the cathode part of the cyclic volt-ampere curves is not observed. This effect is associated with an increase in the mass exchange between the particles on the surface of the electrode and the ions of the electrolyte (0.3 M Na2SO4).
Increasing the concentration of 2,2/ -dipiridyl from 1.4·10-3 to 2·10-5 M in 0.3 M Na2SO4 solution at a constant potential sweep rate of 15 mV / s results in a reduction of the maximum current (A) (Figure 3). A similar effect occurs in the presence of 1,10-phenanthroline in 0.3 M Na2SO4 solution in a concentration ranging from 1.4·10-3 to 2·10-5 M. The presence of 2,2/ -dipyridyl and 1,10-phenanthroline in 0.3 M Na2SO4 solution leads only to a decrease in the current value of the maximum (A) without a significant change in its potential. The observed decrease of the current maximum (A) without changing its potential and decrease the ionization current led to the conclusion that the observed effect is caused by shielding the surface of the iron electrode by heterocyclic amines.
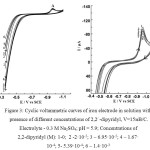 |
Figure 3: Cyclic voltammetric curves of iron electrode in solution with presence of different concentrations of 2,2/ -dipyridyl, V=15мВ/С. Click here to View figure |
Electrolyte – 0.3 M Na2SO4; pH = 5.9; Concentrations of 2,2-dipyridyl (M): 1-0; 2 -2·10-5; 3 – 6.95·10-5; 4 – 1.67·10-4; 5- 5.39·10-4; 6 – 1.4·10-3
In the works of Kolotyrkin et al. [5,6] it is noted that in the wide range of concentrations of sulfate ions and hydroxyl ions, the participation of these ions in two parallel reactions is excluded, while SO42- ions are not able to displace OH– ions. If the sulfate ions replaced the OH– ions, then a decrease in the total speed would be observed, and not an increase in it. Using cyclic voltammetric curves obtained at the iron electrode in sodium sulfate solutions of various concentrations from 0.3 to 1 M, it is established that the ionization current increases with increasing of Na2SO4 concentration, and an increase of the current maximum (A) is observed on the cathode curves without changing its potential E = -0.97 V. If OH– ions were displaced by sulfate ions, an opposite effect would be observed on the anode and cathode curves.
Dissolution of iron occurs according to the following reactions [5,6]:
Fe + H2O ↔ (FeOH ‾ )ads. + H+ (1)
(FeOH ‾ )ads. ↔ (FeOH)ads. + e (2)
(FeOH)ads. + SO42- → FeSO4 + OH ‾ + e (3)
In many respects similar data were obtained in [7]. In solutions close to neutral the anodic dissolution proceeds according to the reactions:
Fe + H2O ↔ (FeOH)ads. + H+ + e (4)
Fe(OH)ads. ↔ FeOH+ + e (5)
FeOH+ + H+ ↔ Fe2+ads. + H2O (6)
With an increase of pH˃5.5, the degree of electrode coating with the complex (FeOH)ads increases, and the formation of an intermediate complex Fe (OH)2 ads is possible
(FeOH)ads. + H2O ↔ Fe(OH)2 ads. + H+ +e (7)
[Fe(OH)2]ads. +Н+ ↔ FeOH+ + H2O (8)
In the presence of anions the intermediate complexes Fe (OH)2 ads come into reaction:
Fe(OH)2 ads. + 2А– FeA2 + 2 OH– (9)
In other words, the addition of 2,2/ -dipyridyl and 1,10-phenanthrolin into the solution in the concentration range under study does not lead to the destruction of FeOH‾ads hydroxycomplex. The formation of dipyridyl and phenanthrolin complexes becomes possible after the destruction of the hydroxyl complex FeOH‾ads, while the solution is colored red due to the reaction Fe2+ + 3 dipy ↔ Fe(dipy)32+ which is a qualitative reaction to iron (II). Similar processes are also characteristic of 1,10-phenanthrolin.
Figure 4 shows cyclic voltammetric curves on an iron electrode in a solution of 0.3 M Na2SO4 at different concentrations of 4,4/ -dipyridyl from 2·10-5 to 1.4·10-3 M. According to Fig. 4, in the presence of 4,4/ -dipyridyl at C ˃ 1.4·10-3 M, the current maximum (A) on cathode curves is practically not observed. Under the same conditions, at an initial potential E = – 0.35 V, the ionization of the iron electrode does not occur.
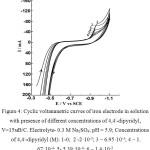 |
Figure 4: Cyclic voltammetric curves of iron electrode in solution with presence of different concentrations of 4,4/-dipyridyl, V=15мВ/С. Click here to View figure |
Electrolyte- 0.3 M Na2SO4; pH = 5.9; Concentrations of 4,4/-dipyridyl (M): 1-0; 2 -2·10-5; 3 – 6.95·10-5; 4 – 1.67·10-4; 5- 5.39·10-4; 6 – 1.4·10-3
Figure 5 shows cyclic voltammetric curves on an iron electrode in a solution of 0.3 M Na2SO4 in the presence of 2,2/ -dipyridyl, 1.10 -phenanthrolin and 4,4/ -dipyridyl at the same concentration 5.7·10-4 M. From comparison presented in Fig. 5 volt-ampere curves, it follows that the greatest inhibitory effect has a 4.4/ -dipiridil (Fig. 5, curve 4). The observed effect is evidently due to the higher adsorption of 4,4/ -dipyridyl than to 2,2/ -dipyridyl.
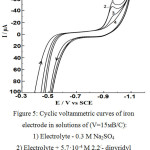 |
Figure 5: Cyclic voltammetric curves of iron electrode in solutions of (V=15мВ/С): Click here to View figure |
1) Electrolyte – 0.3 M Na2SO4
2) Electrolyte + 5.7·10-4 М 2,2/- dipyridyl
3) Electrolyte + 5.7·10-4 М 1,10- phenanthrolin
4) Electrolyte + 5.7·10-4 М 4,4/- dipyridyl
Adsorption studies of the above compounds on mercury were carried out in [6-10]. According to the experimental data [8-11] adsorption energy gained when moving from a solution / air boundary to boundary solution / mercury determines the interaction energy of organic molecules with the uncharged electrode surface. ( for 2,2/ -dipyridyl is 10.94 kJ / mol, and for 4,4/ -dipyridyl this value is 12.57 kJ / mol [8.9], for pyridine the value of ( is 3.81 kJ / mol [11]. It follows that the articulation of the dipyrine rings enhances their π-electron interaction with the surface of mercury, the greatest effect seen at 4,4/ -dipiridyl. With a positive charge of mercury, when the role of the π-electron interaction increases, 4,4/ -dipyridyl is more active at the solution / mercury boundary. The stronger π-electron interaction of 4,4/ -dipyridyl suggests a more flat orientation of this isomer.
In amplification π-electron interaction is also indicated by the Gibbs adsorption isotherm equation (Г by E) in the transition from 2,2/ -dipyridyl to 4,4/ -dipyridyl. This sequence reflects the change in the structure of these compounds. It should be borne in mind that such a strong effect of 4,4/ -dipyridyl, in contrast to 2,2/ -dipyridyl, is apparently due to the fact that 4,4/ -dipyridyl forms poorly soluble polynuclear compounds with iron ions in the form polymers [12], which are able to more effectively block the surface of the iron electrode. As a consequence of these processes, the ionization of the iron electrode, the degree of surface filling by the ionization products and the current value of maximum (A) on the cathode part of the current-voltage curves are sharply reduced.
In the presence of 1,10-phenanthrolin, the ionization of iron proceeds more strongly than in a 0.3 M Na2SO4 solution containing 2,2/ -dipyridyl. In this case, the value of the cathode current of the maximum (A) is less than in the presence of dipyridyl isomers.
Of certain interest is to establish the influence of complexes Fe (dipy)32+ which should be considered as the product of corrosion processes of the iron electrode in the presence of 2.2/ -dipyridyl. In Figure 6 shows cyclic voltammetric curves on an iron electrode in a 0.3 M Na2SO4 solution (curve 1) in the presence of 3.3.10-3 M Fe (dipy)32+ (curve 2) and 3.3.10-3 M 2,2/ -dipyridyl (curve 3). According to Fig. 6 (curve 3), the presence of 3.3.10-3 М Fe (dipy)32+ in the solution leads to an increase of the iron ionization and an increase of the current maximum (A) at E = -0.97 V.
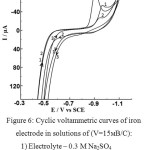 |
Figure 6: Cyclic voltammetric curves of iron electrode in solutions of (V=15мВ/С): |
1) Electrolyte – 0.3 M Na2SO4
2) Electrolyte – 3.3·10-3 М 2,2- dipyridyl
4) Electrolyte – 3.3·10-3 М Fe(dipy)32+
Trisdipyridyl iron complexes Fe (dipy)32+ are chelate thermodynamically stable complexes lgβ3 =17.45 [13]. In terms of their kinetic characteristics, these complexes are inert, the period of half-exchange of dipyridyl with water molecules in the coordination sphere of Fe (II) is 5 hours [14]. At low concentrations of Fe (dipy)32+ in solution the effect of this complex on ionization of iron and the cathode maximum (A) is much like effect 2.2/ dipyridyl at the same concentrations. However, in the case when the concentration of Fe (dipy)32+ reaches 3.3.10-3 M, the ionization of the iron electrode is sharply increased, and the current value of the maximum (A) does not decrease, as in the case of 2,2/ dipyridyl, but increases substantially. It is not possible to explain such effect by shielding the electrode surface with adsorbed Fe (dipy)32+. The observed effect can be explained by the enhancement of the oxidizing ability of Fe (dipy)32+ in the Fe / Fe (dipy)32+ system.
Conclusion
Use of cyclic voltammetry of the iron electrode in solutions close to neutral pH 5.9, containing heterocyclic amines 2,2/ -dipyridyl, 4,4/ -dipyridyl and 1,10-phenanthrolin, as well Fe (dipy)32+ complexes in the investigated solutions allowed to establish the relationship between the ionization of the iron electrode and the appearance of a current maximum at the cathode part of the current-voltage curves.
It has been established that the enhancement of the surface activity of heterocyclic amines on a mercury electrode causes similar effect in the inhibition of corrosion processes at the iron electrode.
Dipyridyl isomers having stronger π-electron interaction with the surface of mercury during their adsorption on the iron electrode with a flat orientation of pyridine rings (4,4/ -dipyridyl) lead to increased anticorrosive ability. It is established that an increase in the concentration of Fe (dipy)32+ corrosion products in the investigated solutions causes an increase in the corrosion processes. It is found that increasing the concentration of corrosion product Fe (dipy)32+ in the investigated solutions causes increased corrosion processes.
The observed change in the current maximum on the cathode curves in the presence of corrosion inhibitors can be used to determine the optimum concentration of inhibitors that prevent corrosion processes.
Acknowledgments
The authors are grateful to the Ministry of Education and Science of the Republic of Kazakhstan for sponsoring this research on the topic: «Development of technology for obtaining new effective materials for anticorrosion coatings on metals and alloys of different nature».
References
- Gatos U.G. Nature. 1958, 181, 1060.
CrossRef - Andropov L.I.; Pogrebova I.S. Results of science and technology, a series of corrosion and corrosion protection. 1973, 2, 27-112 (in russ).
- Annand R.R.; Nard R.M.; Hakerman N.J. J. Electrochem. Sec. 1965, 112, 138.
CrossRef - Annand R.R.; Nard R.M.; Hakerman N.J. J. Electrochem. Sec. 1965, 112, 144.
CrossRef - Damaskin B.B.; Petriy O.A.; Betrakov V.V. M.: “Nauka”. 1968, 333 (in russ).
- Mambetkaziev E.A.; Zhdanov S.I.; Damaskin B.B. Electrochemistry. 1972, 8, 1650-1654 (in russ).
- Mambetkaziev E.A.; Shaldybaeva A.M.; Statsyuk V.N.; Zhdanov S.I. Electrochemistry. 1975, 10, 1750-1754 (in russ).
- Akhmetov N.K.; Kaganovich R.I.; Mambetkaziev E.A.; Damaskin B.B. Electrochemistry. 1977, 13, 280-284 (in russ).
- Akhmetov N.K.; Kaganovich R.I.; Damaskin B.B.; Mambetkaziev E.A. Electrochemistry. 1978, 14, 1761-1765 (in russ).
- Akhmetov N.K.; Kaganovich R.I.; Damaskin B.B. Herald of. MSU (ser.2 chem). 1979, 20, 442-445 (in russ).
- Damaskin B.B.; Survila A.A.; Vasina S.Ya.; Fedorova A.I. Electrochemistry. 1967, 3, 825-830 (in russ).
- Musgrave T.R.; Mattson C.E. Inorg. Chem. 1968, 1433.
CrossRef - Perrin D.D. Oxford: Pergamon Press. 1979, 807.
- Perrin D.D. M .: “Mir”. 1967, 96.

This work is licensed under a Creative Commons Attribution 4.0 International License.









