A Novel Synthesis of [4-(3-Methyl-1H-Indol-2-Yl)Phenyl]Phenylmethanone
Harindran Suhana and S. Rajeswari
and S. Rajeswari
Department of Chemistry, SRM University, Kattankulathur-603203, Tamil Nadu, India. Corresponding author Email: suhana.h@ktr.srmuniv.ac.in
DOI : http://dx.doi.org/10.13005/ojc/330668
We have developed a simple route for the synthesis of [4-(3-methyl-1H-indol-2-yl)phenyl](phenyl)methanone from easily available starting materials. The (4-propylphenyl)phenylmethanone was prepared by Friedel Craft’s benzoylation of n-propyl benzene with benzoyl chloride in dichloromethane using aluminium chloride as the Lewis acid catalyst. Side chain bromination with N-bromo succinimide in tetra chloro methane furnished the bromo derivative which on oxidation with bis-tetra butyl ammonium dichromate gave 1, 4–diacyl benzene. The target molecule was obtained through Fischer indole cyclisation of the diacyl benzene namely 1-(4-benzoylphenyl)propan-1-one via the formation of the hydrazone, followed by cyclization in presence of boron trifluoride ethyl etherate in acetic acid. The structure of the target molecule was elucidated by IR, H1, C13 NMR, Mass spectroscopy and elemental analysis. This method proves to be an efficient route for the synthesis of [4-(3-methyl-1H-indol-2-yl)phenyl](phenyl)methanone in high yields, thereby facilitating the generation of potential biologically active compounds.
KEYWORDS:Fischer indole synthesis; 1, 4-diacylbenzene; Friedel-Craft’s benzoylation; allylic bromination
Download this article as:| Copy the following to cite this article: Suhana H, Rajeswari S. A Novel Synthesis of [4-(3-Methyl-1H-Indol-2-Yl)Phenyl]Phenylmethanone. Orient J Chem 2017;33(6). |
| Copy the following to cite this URL: Suhana H, Rajeswari S. A Novel Synthesis of [4-(3-Methyl-1H-Indol-2-Yl)Phenyl]Phenylmethanone. Orient J Chem 2017;33(6). Available from: http://www.orientjchem.org/?p=40971 |
Introduction
Indole is one of the most important heterocyclic compounds found in nature. It consists of a six-membered benzene ring fused to five-membered nitrogen containing pyrrole ring1. The discovery of indomethacin, ethodolac and tenidap, as potent anti-inflammatory agents, has led to the exploration of the indole nucleus2. A significant number of derivatives were reported to possess potent wide spectrum of biological activity especially antibacterial3-4, antifungal5, anti inflammatory6-10 and analgesic11. Due to high electron density at position 3, indole undergoes electrophilic substitutions at position 2 and 3, respectively. A variety of synthetic methods for the indole ring system have been developed for the past hundred years12-15. The palladium-catalyzed annulation of 2-haloanilines with alkynes, in particular, is of great importance due to the easy availability of substrates16. Indole integrated with diphenylmethanone at position 3 plays a significant role in control of larvicidal mosquitoes that cause malarial fever17. Similar compound with diphenylmethanone substituent at position 2 was synthesized from o-ethynyl aniline and trifluoro sulfonyl benzophenone via two step reaction18. This reaction was catalyzed by palladium in variable oxidation state and high temperature had been used throughout the process. In this study, a novel 2-aryl indole has been synthesized with methyl substituent in position 3, using simple starting materials and novelty in the synthetic strategy.
Experimental
Material and Methods
All commercially available reagents and solvents were obtained from Sigma Aldrich Chemicals and SD Fine Chemicals. TLC was performed on pre-coated alumina sheets and visualized under UV light. . Chromatographic separations were carried out on silica gel 60 (Merck,Darmstadt, Germany). Melting points were obtained using open capillary tubes and uncorrected. Infra-red spectra were recorded on a Jasco Model 5300 FT-IR spectrometer in KBr over a range of 4000-200 cm-1. The 1H NMR and 13C NMR data were recorded with a Bruker 500MHz high resolution NMR Spectrophotometer and CDCl3 was used as a solvent. Chemical shifts are reported in ppm and tetramethylsilane was taken as internal standard. Mass spectra were recorded on JEOL SX 102/DA-6000 mass spectrometer/data system. Elemental analysis results were found to be in good agreement (±0.4%) with the calculated values.
General procedure
Phényl(4-propylphényl)méthanone (2b)
This was prepared by the procedure20 employed earlier by the Friedel-Crafts benzoylation of n-propyl benzene with benzoyl chloride. The product was obtained as an orange syrupy liquid. Yield: 75%
[4-(1-bromopropyl)phenyl](phenyl)methanone (3b)
This was prepared by the procedure20 employed earlier using N-bromosuccinimide in tetra
chloro methane. Yield: 85%
1-[4-(benzoyl)phenyl]propan-1-one(4b)
This was prepared by using bis tetrabutylammonium dichromate as the oxidizing agent by the method20 employed earlier.
Yellow solid; Yield:99% m.p.86-880 C; IR (KBr): νmax 1678, 1664 cm-1; 1H NMR (CDCl3, ppm): 1.2 (t,3H,CH3), 3.1 (q, 2H,CH2), 7.4-8.1 (m, 9H,Aromatic-H); 13C NMR: 8.2, 32.3, 127.9, 128.5, 130.2, 133.1, 139.5, 141.2, 196.1, 200.4,Mass spectrum: m/z (%) = 238.281(M+, 55.6) 209.2 (100), 181.2 (10.2), 105.1 (69.1), 77.1 (61.1) ; Anal Calcd. for (C16H14O2): C 80.65 H 5.92; Found C 80.65 H 5.72; HRMS: (C16H14O2) M+ Calcd: 238.28116; Found: 238.28120
Synthesis of [4-(3-methyl-1H-indol-2-yl)phenyl](phenyl)methanone (5b)
To a solution of 1–[4-(benzoyl)phenyl]propan-1-one (4.76 g, 20 mmol) in ethanol (50 mL) phenyl hydrazine (2.2 mL, 20 mmol) was added. The solution was refluxed for 3 hrs. The ethanol was distilled off and the resulting hydrazone was obtained as a dark brown solid. This was dissolved in acetic acid (50 mL) and BF3-etherate (1 mL) was added drop wise. The solution was refluxed on a mantle for 3 hr. Finally the reaction mixture was cooled and poured over crushed ice. The precipitated bright yellow compound was filtered, washed with water and dried over anhydrous calcium chloride.
Yellow powder; Yield: (95%); mp:198-200 oC ; IR(KBr): νmax1650 (C=O), 3343 (NH)cm-1,
1H NMR (500MHz; DMSO-d6): 2.46 (s, 3H, CH3), 7.00-8.07 (m, 13H, Aromatic-H), 11.34 (s, 1H, indole-NH);13CNMR(125 MHz): 10.6,109.6,111.8,119.4,119.4,122.9,127.6,129.1,129.9,130.1,130.8,132.9,133.1,137.8,195.7.
Mass m/z (%): 311.30 (M+, 100%), 310.27 (37.2), 209.20(46.1), 130.17(25.8), 105.1(65.6) 77.14(64); Analysis Calcd. for C22H17NO: C 84.86,H 5.50, N 4.50 Found: C 84.51, H 5.51, N 4.62.
Phényl(4-ethylphényl)méthanone 2a
This was prepared by the earlier reported procedure by the Friedel-Craft’s reaction of ethyl benzene with benzoyl chloride.Yield:78%
Synthesis of [4-(1-bromoethyl)phenyl](phenyl)methanone 3a
The compound was prepared by bromination of phényl(4-ethylphényl)méthanone 2a using N-bromosuccinimide in carbon tetra chloride using the procedure previously reported in our laboratory. The product was obtained as an orange syrupy liquid.Yield:82%
Preparation of (4-benzoylphenyl)ethanone (4a)
The (4-benzoylphenyl)ethanone was prepared from [4-(1-
bromoethyl)phenyl](phenyl)methanone 3a by the reported procedure19 using bis tetra butylammonium dichromate as the oxidizing agent. The melting point and spectral data were in close agreement with that reported earlier. Light yellow solid, Yield: 74%; m.p:80-84oC; IR(KBr):νmax1689,1656cm-1,1H NMR(CDCl3/TMS,ppm): 2.56(s, 3H, CH3), 7.2-8(m, 9H, Ar)
Synthesis of [4-(1H-indol-2-yl)phenyl](phenyl)methanone (5a)
To a solution of 1-(4-benzoylphenyl)ethanone(5.6g, 25mmol) in ethanol (50mL) was added phenylhydrazine (3mL, 25mmol) and concentrated hydrochloric acid (0.5mL). The reaction mixture was refluxed for 3 h. Then ethanol was distilled off and the resulting hydrazone was obtained as a dark brown solid. This was dissolved in xylene (50mL) and anhydrous ZnCl2 (6.8g, 50 mmol) was added. The reaction mixture was refluxed in an oil bath for 3 h and the progress of the reaction monitored by TLC. Then xylene was distilled off and the pot residue obtained was digested with water on a water bath for 0.5 h. A yellow glassy solid which separated out, was filtered and dried. It was purified by chromatography over silica gel using hexane / ethyl acetate (1:1) as the eluent .
Yield: 50%; mp: 110 oC (decomposed); IR (KBr ): 1669 (>C=O), 3313 (NH) ) cm-1; 1H NMR(CDCl3/TMS): d 6.4-7.7 (m, 13H, Aromatic), 10.9 (s, 1H, indole-NH); Mass m/z (%): 298.19 (100) 221.26 (29.5), 193.03 (43.7), 105.02 (41.9), 77.1 (90.3), 55.06 (43.7).
Preparation of bis-tetrabutyl ammonium dichromate
The above oxidizing agent was prepared by the method reported19 in the literature, from tetra butyl ammonium bromide and potassium dichromate.
Results and Discussion
In this research work, the target molecule containing a diphenyl methanone moiety and methyl group at positions 2 and 3 respectively was synthesized. The product was obtained in good yield whereas the yield of the 2-substituted product namely [4-(1H-indol-2-yl)phenyl](phenyl)methanone which was prepared18 by the palladium catalysed coupling of diphenylmethanone triflate with 2-ethynyl aniline was only 39%. Moreover, the use of expensive and toxic starting materials was replaced with simple, inexpensive and less toxic starting materials.
To synthesise indoles with diphenylmethanone unit at the second position, 1-(4-benzoylphenyl)ethanone and 1-(4-benzoylphenyl)propan-1-one were required as starting materials. They were prepared by following the procedure20 employed earlier. Initially Fischer indolization of 1-(4-benzoylphenyl)ethanone with phenyl hydrazine in ethanol followed by refluxing the resulting hydrazone with anhydrous zinc chloride in xylene afforded [4-(1H-indol-2-yl)phenyl](phenyl)methanone 5a in 50% yield.
The product obtained was found to melt at 1100C and the structure has been confirmed by IR and NMR spectra. The IR spectrum showed peaks at 3313 cm-1 due to indole NH and at 1669 cm-1 due to carbonyl group. The 1H NMR spectrum showed a multiplet at ẟ 6.4-7.7 due to aromatic hydrogens and a broad singlet at d 10.9 due to indole NH. This compound has been reported18 by the palladium catalyzed coupling of 4-benzophenone triflate with 2-ethynylaniline.The structure was confirmed by its IR and NMR data which was in agreement with that reported in the literature.
Further, it was decided to indolise the corresponding 1-[4-(benzoyl)phenyl]propan-1-one to the corresponding indole by the Fischer indolisation of the hydrazone. 1-[4-(benzoyl)phenyl]propan-1-one was treated with phenyl hydrazine in ethanol and the resulting hydrazone was subjected to Fischer indolization using BF3– ethyl etherate in boiling acetic acid. The [4-(3-methyl-1H-indol-2-yl)phenyl](phenyl)methanone 5b was obtained in quantitative yield and the structure has been confirmed by IR, 1H NMR and 13C NMR spectra. The presence of a carbonyl group was confirmed from IR by the presence of a sharp peak at 1650cm-1.In the 1H NMR spectrum, peak at d 3.9 containing 3H singlet was due to indole methyl hydrogen, multiplet peak at d 6.7-8.4 for aromatic hydrogens and a highly deshielded singlet peak at d 8.5 was due to NH of indole. (Figure:1).The 13C NMR spectrum showed 15 peaks (for twenty two carbons-18 signals) at 10.65, 109.63, 111.78, 119.38, 119.42, 122.94, 127.59, 129.13, 129.86, 130.08, 130.83, 132.93, 133.13, 137.77, and195.72.(Figure:2).The mass spectrum showed a molecular ion peak at m/z = 311.30 (Figure:3).The scheme shown below represents the various steps involved in the synthesis.
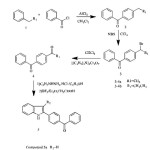 |
Scheme 1: Synthesis of diphenylmethanone integrated indoles Click here to View scheme |
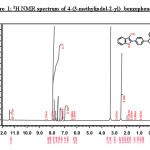 |
Figure 1: 1H NMR spectrum of 4-(3-methylindol-2-yl) benzophenone Click here to View figure |
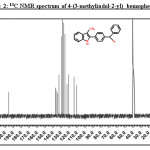 |
Figure 2: 13C NMR spectrum of 4-(3-methylindol-2-yl) benzophenone Click here to View figure |
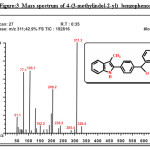 |
Figure 3: Mass spectrum of 4-(3-methylindol-2-yl) benzophenone Click here to View figure |
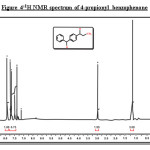 |
Figure 4: 1H NMR spectrum of 4-propionyl benzophenone Click here to View figure |
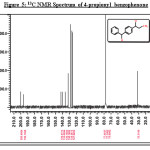 |
Figure 5: 13C NMR Spectrum of 4-propionyl benzophenone Click here to View figure |
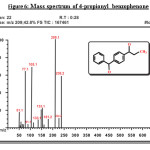 |
Figure 6: Mass spectrum of 4-propionyl benzophenone Click here to View figure |
Conclusion
In the present work, we have developed a simple but highly efficient method for the synthesis of a novel diphenylmethanone integrated indole, which is also supported by the IR and NMR spectral data. Further structural optimization of diphenylmethanone based indoles is well under way with the aim of evaluating their biological activity.
Acknowledgement
The authors gratefully acknowledge SRM University for the funds released under “The Faculty Selective Excellence Initiative” to carry out the work .We also thank University of Madras, Guindy Campus for providing the NMR and Mass Spectra.
References
- Ramesh Dhani, A.; Avinash, S.K.; M.V. Salenaagina, P.; SaicharanTeja, M.V.; Masthanaiah, P.; Raja Rathnam, Chandana silpa, V. J. Chem. Pharm. Res. 2011, 3(5), 519-523.
- Trilok Chandra, Neha Garg, Ashok Kumar. Int. J. Chem Tech Res. 2010, 2, 762-773.
- Siavosh, M.; Emerich, E.; Alfred, P.; Andreas, S.; Sigurd, M.; Ute, M. Eur. J. Med. Chem. 2006, 41, 176-191.
CrossRef - Siavosh, M.; Emerich, E.; Matthias, W.; Andreas, S.;Ute, M. Eur. J. Med. Chem. 2008,43(3), 633-656.
CrossRef - Velazquez, C.A.; Rao, P.N.; Citro, M.L.; Keefer, L.K.; Knaus, E.E. Bio-Org. Med.Chem. 2007,15(14),4767-74.
CrossRef - Reddy, M.V.; Billa, V. K.; Pallela, V.R.; Mallireddigari, M.R.; Boominathan, R.; Gabriel, J.L.; Reddy, E.P. Bioorg. Med. Chem. Lett. 2008, 16, 3907-16.
CrossRef - Sondhi, S.M.; Jain, S.; Rani, R.; Kumar, A. Ind. J. Chem. 2007, 46B, 1848-54.
- Mohamed, A.A.R.; Ragab, E.A.; Sabry, N.M.; S. El Shenawy. Bioorg. Med. Chem. 2007, 15(1), 3832-41.
- Khanna. S.; Madan. A.; Vangoori, R.; Banerjee, R.; Thaimattam, S.K.; Basha, J.S.; Ramesh, M.; Rao,S.; Pal, M. Bioorg. Med. Chem. 2006, 14(14), 4820–33.
CrossRef - Goksen, U.S.; Kelekci, N.G.; Goktas, O.; Koysal,Y.; Kilic, E.; Isik, S.; Aktay, G.; Ozalp, M. Bioorg. Med.Chem. 2007, 15, 5738-51.
CrossRef - Gribble, G.W. J. Chem. Soc. Perkin Trans. 1, 2000, 105, 1045–75.
CrossRef - Cacchi.S.; Fabrizi,G. Chem. Rev. 2005, 105, 2873– 2920.
CrossRef - Battistuzzi, G.; Cacchi ,S.; Fabrizi, G. Eur. J. Org. Chem. 2002, 2671–2681.
CrossRef - Zeni, G.; Larock, R.C. Chem. Rev. 2004, 104(5), 2285–2309.
CrossRef - Alonso,F.; Beletskaya, I.P.; Miguel,Y. Chem.Rev. 2004,104, 3079-3160.
CrossRef - Monguchi, Y.; S. Mori, S.; Aoyagi, S.; Tsutsui, A.; Maegawa, T.; Sajiki, H. Org Biomol. Chem. 2010, 14(8), 3338-3342.
CrossRef - Lakshmi Ranganatha, V.; Bushra Begum, A.; Prashanth,T.; Gurupadaswamy, H.D.; Madhu, S.K.; Shivakumar, S.; Khanum, S. A. Drug Invention Today.2013, 5(4), 275-280.
CrossRef - Arcadi, A.; Cacchi,S.; Marinelli,F. Tet.Lett.1989, 30(19), 2581–2584.
CrossRef - Landini,D.; Rolla,F. Chem. Ind. (London), 1979, 213.
- Suhana, H.; Srinivasan, P.C. Synth. Commun. 2003, 33(18), 3097-3102.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.









