Increased of Cellulase Obtained from Bacillus Subtilis ITBCCB148 with Chemical Modification Using P-Nitrophenolcarbonate-Polyethylenglycol (NPC-PEG)
Yandri, Putri Amalia, Tati Suhartati, Mulyono and Sutopo Hadi
Department of Chemistry, University of Lampung, Bandar Lampung, 35145 Indonesia.
Corresponding Author E-mail: yandri.as@fmipa.unila.ac.id
DOI : http://dx.doi.org/10.13005/ojc/330547
This research aims to increase the stabilty of cellulase obtained from local bacteria isolate Bacillus subtilis ITBCCB148 by chemical modification using p-nitrophenolcarbonate polyethylenglycol (NPC-PEG). The characterization of the native and modified enzymes were performed by determination of optimum pH and temperature as well as thermal stability. The result showed that the native enzyme has optimum temperature and pH of 55oC and 6, respectively. The thermal stability test of native enzyme for 60 minutes at temperature of 55oC gave values of thermal inactivation rate constant (ki) 0.035/min., half-life (t1/2) 19.80 minutes and denaturation energy ΔGi)= 100.908 kJ/mol. The modified enzymes with modification degree of 63, 70 and 81% have the same values of optimum temperature and pH as native enzyme, but their stability data have increased very significantly. Their ki values were 0.014; 0.012 and 0.011, respectively. Their t1/2 values were 49.5; 57.75 and 63 minutes, respectively; and their ΔGi values were 103.407; 103.828 and 104.065 kJ/mole, respectively. Thus, the chemical modification using NPC-PEG has shown to increase the enzyme stability of modified enzyme 2.5 – 3.2 times compared to the native enzyme, although no change of optimum temperature and pH was observed.
KEYWORDS:B. Subtilis ITBCCB148; Celullase; Chemical Modification; NPC-PEG
Download this article as:| Copy the following to cite this article: Yandri Y, Amalia P, Suhartati T, Mulyono M, Hadi S. Increased of Cellulase Obtained from Bacillus Subtilis ITBCCB 148 with Chemical Modification Using P-Nitrophenolcarbonate-Polyethylenglycol (NPC-PEG). Orient J Chem 2017;33(5). |
| Copy the following to cite this URL: Yandri Y, Amalia P, Suhartati T, Mulyono M, Hadi S. Increased of Cellulase Obtained from Bacillus Subtilis ITBCCB 148 with Chemical Modification Using P-Nitrophenolcarbonate-Polyethylenglycol (NPC-PEG). Orient J Chem 2017;33(5). Available from: http://www.orientjchem.org/?p=37463 |
Introduction
The research on thermostable enzymes nowadays is so interesting due to their used in industrial developments especially in industries of detergent, production of glucose syrup from starch, synthesis of organic compound, paper and pulp, animal feed, the treatment of industrial wastes1. The thermostable enzyme work optimum at temperature at 60-125°1. The research about this enzyme has been a hot topic as it is considered as the best way to study the protein stability.
Cellulase is an induction enzyme produced by microorganism in cellulose medium2. This enzyme works gradually in hydrolizing cellulose to glucose3. Cellulase is widely used mainly in the enzymatic conversion of cellulose to glucose. In order to be applied in the industrial process, cellulase must comply with some requirements, especially it can stand at extreme pH and temperature. Thus, enzyme with high stability must be obtained as in general is not stable at high pH and temperature4.
Enzymes with high stability and activity at extreme condition may be obtained by direct isolation from natural organisms which are found at this condition or by chemical modification to enzyme obtained from organism found in not extreme condition5. Mozhaev and Martinek6 stated that the enzyme stabilization of enzyme from mesofilic organism by chemical modification is a prefered way to obtain a stable enzyme.
In our previous researches, chemical modification to cellulase with low molecular weight modifiers, i.e. glyoxylic acid and dimethyladipimidate have been performed7,8..The results showed that all modified enzymes have increased their stability compared to the native enzyme. In this paper, to increase the stability of cellulase obtained from local bacteria isolate B. subtilis ITBCCB148, the chemical modificatin using high molecular weight modifier, p-nitrophenolcarbonate polyethylenglycol (NPC-PEG) was carried out.
Experimetanl
Materials
All materials used in this research were pro analysis reagent and used without purification. Local bacteria isolate B. subtilis ITBCCB148 was obtained from Microbiology Laboratory, Chemical Engineering Department, Bandung Institute of Technology, Bandung, Indonesia.
Research Phases
Research phases performed were production, isolation, purification and characterization of the purified (native) enzyme similar to the procedures previously reported9.
Cellulase Activity Test and Protein Content Determination
The cellulase activity test was performed based on the Mandels method using dinitrosalycilic acid10 and the protein content determination was performed based on the method by Lowry et al. 11.
The Modification of Native Enzyme with P-Nitrophenolcarbonate Polythylenglycol (NPC-PEG)12
10 mL of native cellulase in 0.1 M borate buffer pH 8 was added with NPC-PEG solid with variation of 5, 10 and and 15 mg. The reaction was mixed with magnetic stirrer for 2 hours at 4oC.
Determination of Modification Degree
Determination of modification degree was done based on the method used by Synder and Sobocinski13 and as follows: 0.1 mL of modified enzyme was dissolved into 0.9 mL borate buffer (pH 9.0) and then was added with 25 μl 0.3 M 2,4,6-trinitrobenzene sulfonic acid (TNBS). The mixture was then shaken and left it at room temperature for 30 min. The standard solution was made with the same composition but using the native enzyme, while the blank solution was made up of 1 mL borate buffer 0.1 M pH 9 and 25 μl 0.3 M TNBS. The absorbances were measured at the lmax. 420 nm.
Characterization of Enzyme Before and Aftervmodification
Determination of Optimum Temperature
The determination of optimum temperature was performed by varying the temperature at 45, 55, 60, 65, 70, 75 and 80°C.
Determination of Optimum pH
Determination of optimum pH was performed by varying pH at 5; 5.5; 6; 6.5; 7; 7.5; 8; 8.5; 9.
Determination of KM and Vmax.
The values of Michaelis-Menten (KM)and maximum reaction rate (Vmax.) of the enzymes were performed by varying the concentrations of substrate (amylum solution) at 0.25; 0.5; 0.75; 1.0; and 1.25%.
The Thermal Stability Test of Enzyme
The thermal stability of native and modified enzymes was performed by measuring the residual activity of the enzyme after being incubated for 0, 10, 20, 30, 40, 50, and 60 minutes at temperature of 55°C14.
Determination of Half-Life (t½), ki and ΔGi
Determination of ki value (thermal inactivation rate constant) of the enzyme was performed using the first order of inactivation kinetics equation (Eq. 1)15:
ln (Ei/E0) = – ki t (1)
The change of energy due to denaturation was performed using Equation 215.
ΔGi = – RT ln (ki h/kB T) (2)
Results and Discussion
Chemical modifications of native enzyme with NPC-PEG were performed with 3 concentration variations at 5, 10, and 15 mg (Table 1)
Table 1: Determination of modification degree using TNBS
|
Sampel |
Δ A420 nm |
Modification (%) |
|
Native Enzyme |
0.5991 |
0 |
|
Modification with NPC-PEG 5 mg |
0.5867 |
63 |
|
Modification with NPC-PEG 10 mg |
0.5853 |
70 |
|
Modifikasi dengan NPC-PEG 15 mg |
0.5834 |
81 |
The determination of modification degree was based on the comparison of lysine residue before and after modification. Based on Table 1, the result of modification with NPC-PEG produced the modification degree of 63, 70 and 81%, respectively. The lysine residue on the native enzyme which was not modified with TNBS will form yellow complex, while on the modified enzymes, the unmodified lysine residu react with TNBS to form pale yellow complex. The more lysine residues modified, the less intense of the yellow complex observed.
Determination of Optimum Temperature
All modified enzymes with NPC-PEG have optimum temperature of 55°C, the same value as the native enzyme. This finding can be expained that due to bulky molecular size of PEG, it was unable to enter the area heading to the site active of the enzymem as a result the three dimentional structure of the enzyme did not change so the activation energy also did not change.
This condition caused the optimum temperature did not change. The result is inline with assumption that the enzyme modification is only targeted occurred to NH2 group on the side chance of lysine on the surface of the enzyme. Kazan et al.15 and Francis et al.16 reported that chemical modification did not always cause the optimum temperature of the modified enzyme. Although no change on optimum temperature, however, the activity of the modified enzymes at higher temperature was better than that of the native enzyme, especially the modified enzymes at higher modification degree (70 and 81%). This is because the modification caused the enzyme rigidity increased, so the modified enzymes were more stand against temperature.
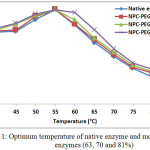 |
Figure 1: Optimum temperature of native enzyme and modified enzymes (63, 70 and 81%)
|
Determination of Optimum pH
There was no different observed for optimum pH for native and modified enzymes, i.e. at pH 6 (Fig. 2). The native and modified enzymes were stable at pH range of 5-8. Fig. 2 also showed that although optimum pH of modified enzymes did not change, however the modified enzymes have better activity compared to the native enzyme especially at pH range of 6.5 and 8.5.
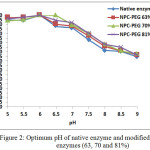 |
Figure 2: Optimum pH of native enzyme and modified enzymes (63, 70 and 81%) Click here to View figure |
Determination of KM and Vmax Values
Based on the Lineweaver-Burk equation, the Vmax. and KM values of native enzyme were 10.101μmol mL-1 min.-1 and 3.878 mg/mL substrate, respectively. While the values obtained for all modified enzymes were Vmax.: 8.547; 8.403 dan 9.615 μmol mL-1 min.-1, respectively; and KM:3.188; 3.327 and 4.096 mg/mL substrate, respectively. The determination result of KM values for native and all modified enzymes were not significantly different, this is predicted that no structural change occurred to the enzymes, mainly at the site active of the enzyme, so the enzyme afinity toward substrate also did not change.
The Vmax. values of all modified enzymes were decreased compared to the native enzymes, this is because the chemical modification with long chain NPC-PEG molecule on the surface of the enzyme made the enzyme was less flexible on aqueous solution14. PEG has also found to affect the enzyme proximity and orientation. Unmodified enzyme is easily able to interact with substrate, due to the present of long chain PEG molecule causing the condition around the enzyme is changed, so the VMax. is decreased. . Kazan et al.15 and Francis et al.16 reported that chemical modification did not always cause the values of KM and VMax. changed on the modified enzyme.
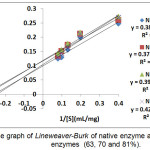 |
Figure 3: The graph of Lineweaver-Burk of native enzyme and modified enzymes (63, 70 and 81%). Click here to View figure |
Thermal Stability of the Enzyme
As can be seen from Fig. 4 that % residual acitivity of hative enzyme after being incubated at 55oC for 60 minutes was 8.071, while the modified enzymes (63, 70 and 81%) were 42.633; 48.415 and 48.616, respectively. The thermal stability of all modified enzymes with NPC-PEG were increased compared to the native enzyme (Fig. 4). All modified enzymes at different modification degree have half-life higher than that of native enzyme, thus the chemical modification has a good effect to the structure stability.
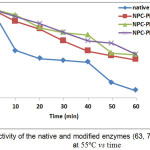 |
Figure 4: Activity of the native and modified enzymes (63, 70 and 81%) at 55ºC vs time Click here to View figure |
The mechanism of structure stability occurred to the cellulase due to chemical modification with activated PEG may be explained as follows: (1) the decrease of contact between the hydrophobic site and the solvent, since the wider of the hydrophobic site to contact with solvent will easily destabilize the enzyme structure; (2) the addition of hydrogen bond, as long chain of PEG may form hydrogen bond at the surface of the enzyme.
Modification of PEG to lysine residue exposed at the surface cause the hydropobic site at the enzyme surface is protected as a result its contact with solvent is decreased making the celluase structure is more desired
Janecek and Balaz17stated that the modification style with high molecular weight modifier such as PEG possibly made the modifiers to protect the hydrophobic site and prevent it to contact with water. PEG with molecular weight of 5000 is likely to follow this stabilization mechanism.
Gaetner and Puigserver18 reported that the stability of trypsin modified with activated PEG was due to the formation of many hydrogen bonds on the enzyme structure caused by the long chain of PEG. This fact supported the result obtained in this work, the higher the modification degree, the higher enzyme thermal stability of the modified enzymes, and it is predicted that the numbers of hydrogen bond form are increased with the increase the modification degree.
Half-Life (T1/2), Thermal Inactivation Constant (Ki), and Denaturation Energy Change (Δgi)
Rate of thermal inactivation constant (ki), Half-life (t1/2), and denaturation energy change (ΔGi) are presented in Table 2.
All modified enzymes have increased their half-lives between two to three times compared to the native enzyme. According to Stahl19, thermal stability of enzyme is determined by determining the half life of the enzyme. The half life increased of the enzyme modified by NPC-PEG reported here similar to the result reported by other12.
The half-lives of cellulase modified by NPC-PEG with modification degrees of 63, 70 and 81% have increased their half-lives 2.5; 2.9 and 3.2 times respectively (Table 2). The result reported by Hernaiz et al.12 which modified lipase obtained from Candida rugosa using activated PEG with modification degree of 60 and 68% have increased the half-lives to 2.1 and 5 times.
Table 2: The values of ki, t1/2, and ΔGi of all enzymes
|
Enzyme |
ki (min.-1) |
t1/2 (minute) |
ΔGi (kJ/mol) |
| Native |
0.035 |
19.80 |
100.908 |
| NPC-PEG 63% |
0.014 |
49.50 |
103.407 |
| NPC-PEG 70% |
0.012 |
57.75 |
103.828 |
| NPC-PEG 81% |
0.011 |
63.00 |
104.065 |
The rate of inactivation thermal constant (ki) values for all modified enzymes were 2.5-3.2 times compared to the native one (Table 2). These data indicated that the rates of enzyme denaturation to the modified enzymes have decreased quite significant. In this work, the observation was performed to look at closely on the effect of chemical modification to the ki values of the modified enzymes with NPC-PEG. The decrease of ki values are predicted due to the bond formation between PEG long chain with NH2 group on side chain lysine residue on the surface of the enzyme molecule. This condition made enzyme less flexible in aqueous solution because of the protein unfolding became less, as a result the enzyme stability increased14.
Table 2 also shows that the DGi of modified enzymes was increased significantly compared to the native one. The higher the modification degree, the higher DGi obtained, this indicated that the modified enzymes were more rigid and less flexible, as a result the energy to denature the enzyme was also higher.
Conclusions
The chemical modification of native cellulase with p-Nitrophenolcarbonate-Polyethylenglycol (NPC-PEG) has successfully increased the thermal stability of the cellulase where the stability was increased 2.5-3.2 times. The decrease of ki values, increase of half-life (t1/2) and DGi indicated that the modified enzymes were more stable than the native cellulase.
Acknowledgments
The authors would like to thank The Directorate of Research and Community Services, The Ministry of Resarch, Technology and Higher Education, Republic of Indonesia that provided funds for this project to be undertaken through the Competency Grant Research Scheme (Penelitian Hibah Kompetensi) 2016 with contract number 79/UN26/8/LPPM/2016, 13 April 2016.
References
- Vieille, C. and Zeikus, J.G. Tibtech., 14(6): 183-189 (1996).
CrossRef - Lee, S. M. and Koo, Y. J. Microbiol. Biotechnol. 11 : 229-233 (2001).
- Shiratori, H., Ikeno, H., Ayame, S., Kataoka, N., Miya, A., Hosono, K., Beppu, T. and Ueda, K. Appl. Environment. Microbiol. 72 (5) : 3702-3709(2006).
CrossRef - Goddete, D.W., Terri, C., Beth, F.L. , Maria, L., Jonathan, R.M., Christian, P., Robert, B.R., Shiow, S.Y., Wilson. C.R. J. Biotechnol., 28: 41-54 (1993).
- Wagen, E.S. Strategies for Increasing The Stability of Enzymes, in Enzyme Engineering. The New York Academy of Sciences, New York. 7: 1-19 (1984).
- Mozhaev, V.V. and K. Martinek. Enzyme Microb. Technol., 6 (2): 50-59 (1984).
CrossRef - Yandri, Rachmawati,R., Suhartati, T. and Hadi, S., J. Pure Appl.Microbiol., 8(5): 3675-3680 (2014).
- Yandri, Amalia, P., Suhartati, T. and Hadi, S. Biosci. Biotechnol. Res. Asia,12(3), 2089-2093 (2015).
CrossRef - Yandri, A.S., Suhartati, T. and Hadi, S. Eur. J. Sci. Res., 39: 64-74 (2010).
- Mandels M., Raymond A. and Charles R., Biotech & Bioeng. Symp. No 6. John Wiley & Sons Inc (1976).
- Lowry O.H., Rosebrough N.J., Farr A.L. and Randall R.J., J. Biol. Chem., 193-269 (1951).
- Hernaiz, M.J., Montero, J.M.S. and Sinisterra, J.V. Enzyme Microb. Tecnol. 24. 181-190 (1999).
CrossRef - Synder, S.L. and Sobocinski, P.Z. Anal,Biochem., 64: 284- 288 (1975).
CrossRef - Yang Z., Michael D., Robert A., Fang X.Y. and Alan J.R., Enzyme Microb. Technol., 18: 82-89 (1996).
CrossRef - Kazan D., Ertan H. and Erarslan A., Appl. Microbiol. Biotechnol., 48: 191-197 (1997).
CrossRef - Francis G.E., Delgado C. and Fisher D., PEG modified proteins in Stability of ProteinPharmaceuticals Part B (Ahern T J & Manning M C, Eds), Plenum Press, New York, 246-247 (1992).
- Janecek, S. and Balaz, S. Febs Lett., 304 (1), 1-3 (1992).
CrossRef - Gaertner, H. F. and Puigserver, A.J. Enzyme Microb. Technol., 14: 150-155 (1992).
CrossRef - Stahl S., Thermophilic Microorganisms: The Biological Background for Thermophily and Thermoresistance of Enzymes in Thermostability of Enzymes (Gupta, M.N.editor), Springer Verlag, New Delhi, 59-60 (1999).

This work is licensed under a Creative Commons Attribution 4.0 International License.









