Supercritical Fluid Extraction as A Technique to Obtain Essential Oil from Rosmarinus Officinalis L.
Golchehreh Khalili1, Ali Mazloomifar2, Kambiz Larijani1, Mohammad Saber Tehrani1 and Parviz Aberoomand Azar1
1Department of Chemistry, Science and Research Branch, Islamic Azad University, Tehran, Iran.
2Department of Chemistry, Yadegar-e- Imam Branch, Islamic Azad University, Tehran, Iran.
Corresponding Author E-mail: mazloomifar@yahoo.com
DOI : http://dx.doi.org/10.13005/ojc/330549
The aim of this study was the comparison of essential oil of one herb from lamiaceae family namely Rosemary, via supercritical fluid extraction (SFE) and hydrodistillation (HD). The essential oils were analyzed by gas chromatography – quadrupole mass spectrometry (GC-MS). The compounds identified in higher relative abundance were, α-pinene (HD: 30.42%, SFE: 0.47%), 1,8-cineole (HD: 15.46%, SFE: 25.05%), Verbenone (HD: 13.09%, SFE: 5.17%), Camphor (HD: 8.97%, SFE: 15.69%), Borneol (HD: 7.16%, SFE: 9.15%) and β- Caryophyllene (SFE: 6.85%).
KEYWORDS:Rosemary; Supercritical fluid Extraction; Hydrodistillation; Essential Oil
Download this article as:| Copy the following to cite this article: Khalili G, Mazloomifar A, Larijani K, Tehrani M. S, Azar P. A. Supercritical Fluid Extraction as A Technique to Obtain Essential Oil from Rosmarinus Officinalis L. Orient J Chem 2017;33(5). |
| Copy the following to cite this URL: Khalili G, Mazloomifar A, Larijani K, Tehrani M. S, Azar P. A. Supercritical Fluid Extraction as A Technique to Obtain Essential Oil from Rosmarinus Officinalis L. Orient J Chem 2017;33(5). Available from: http://www.orientjchem.org/?p=38803 |
Introduction
Rosemary (Rosmarinus officinalis L.) is a Long-lasting evergreen herb that belongs to the Lamiaceae family1. Rosemary is native to the Mediterranean region. It possesses needle-like leaves which contain a powerful fragrance2. It is used as a food flavoring agent, and well known medicinally for its powerful antitumor, antibacterial, antifungal and antioxidant activity3-7. Rosemary essential oil composition and the ratio between different oil constituents plays a role in its bioactivity8. Rosemary oil composition depends on various environmental and genetic factors, as well as on postharvest processing factors9. Supercritical fluid extraction showed extremely higher selectivity compared to the conventional techniques tested. Supercritical fluid extraction is an innovative, clean and environmental friendly technology with particular interest for the extraction of essential oil from plants and herbs. Furthermore, supercritical extracts were often recognized of superior quality when compared with those produced by hydrodistillation10-11. The advances that supercritical technology is reaching in the past years are related to the continuous increase of activities connected to scientific research and technological development, which focus on inserting novel processes in the food, pharmaceutical, chemical and cosmetics industries11.In this paper, we present a comparative study of the ability of two different techniques to extract the essential oils from Rosmarinus leaves to find the most advantageous in term of essential oil quantity and quality.
Materials and Methods
HPLC grade n-hexane and analytical grade anhydrous sodium sulfate were purchased from Merck (Darmstadt, Germany). Dichloromethane was purchased from Aldrich. Carbon dioxide (99.8% purity) contained in a cylinder was purchased from Sabalan Co. (Tehran, Iran). The Rosemary (Rosmarinus officinalis L.) leaves used was collected from the north of Tehran, Iran in August 2016.
GC-MS Analysis
The Analyses were performed using a GC-MS system (Hewlett-Packard 6890 gas chromatograph coupled to a 5973A mass spectrometer), equipped with split/splitless injector in the splitless mode and at 250°C during the chromatographic run. The volatile compounds were separated in a capillary column (30 m × 0.25 mm, 0.25 µm film thickness, HP-5MS) using N2 (99.99%) as a carrier gas at a 1 mL/min flow rate. The oven temperature was varied as follows: 60°C (5 min), then warmed to 250°C at 5°C/min, then held 10 min at 250°C. The mass detector conditions were: transfer line temperature of 220°C, ion source temperature of 220ºC and ionization mode with electron impact at 70 eV. The Kovats retention index (RI) values were calculated using a homologous series of n-alkanes (C8 – C25) and values were compared with values reported in the literature and available Adams data12. All mass spectra were also compared with the data system library (Wiley 275). Identification of volatile compounds was achieved by matching mass spectra and retention indices (RI) with those obtained from Wiley library as well as from literature data.
Hydrodistillation (HD)
Rosemary leaves were shade dried at room temperature for 72 hours. The dried rosemary leaves were loaded to hydrodistillation using a Clevenger-type apparatus, according to European Pharmacopeia, and extracted with 1 L of water for 240 min (until no more essential oil was reused).
Supercritical fluid Extraction (SFE)
A Suprex MPS/225 system (Pittsburgh, PA) in the SFE mode was used for all the extractions. The extraction vessel was a 10 mL stainless steel vessel. Supercritical fluid extractions were conducted at pressure 30 MPa and temperature 40°C for a duration of 20 min, static, followed by 35 min, dynamic. These optimized conditions were obtained from literature13. A Dura flow manual variable restrictor (Suprex) was used in the SFE system to collect the extracted essential oil. In order to prevent sample plugging, the restrict point was warmed electrically. The supercritical CO2 flow rate through the Duraflow restrictor was approximately 0.3-0.4 mL/min (compressed). Plant powder (3.0 g) was well mixed with 2 mm diameter glass beads, and was then charged into the 10 ml extraction vessel. The essential oil was extracted from the plant using supercritical CO2. The extracted essential oil was collected in dichloromethane in a 5.0 mL volumetric flask. The final volume of the extract was adjusted to 5.0 ml with dichloromethane at the end of the extraction. Four milliliters of solution were poured into a 20 mL beaker. The solvent was evaporated by Bubbling argon gas through the solution. Then the weight of essential oil was measured and the extraction yield was calculated.
Result and Discussion
The identified constituents in the extracted essential oils from both methods are present in Table 1. For hydrodistillation and supercritical fluid extraction 29 and 16 compounds, respectively were characterized, according to 99.91% and 91.75% of the essential oils. In the hydrodistillation technique, the essential oil consists of oxygenated compounds 56.33% and 42.39% monoterpene hydrocarbons with, α-pinene (30.42%), 1,8-cineole (15.46%), Verbenone (13.09%), Camphor (8.97%) and Borneol (7.16%) as the major components in the oil.
Table 1: Chemical composition of the essential oil isolated from Iranian Rosmarinus officinalis via hydrodistillation (HD) and supercritical fluid extraction (SFE)
| Compounds | Formula | RI | HD(%) | SFE(%) |
| α-Pinene | C10H16 | 939 | 30.42 | 0.47 |
| Camphene | C10H16 | 954 | 5.29 | 0.31 |
| β-Pinene | C10H16 | 979 | 1.66 | 0.12 |
| 3-Octanone | C8H16O | 984 | 2.48 | – |
| β-Myrcene | C10H16 | 991 | 2.37 | 0.11 |
| α-Terpinene | C10H16 | 1017 | 0.50 | – |
| p-Cymene | C10H14 | 1026 | 0.93 | 0.15 |
| 1,8-Cineole | C10H18O | 1031 | 15.46 | 25.05 |
| γ-Terpinene | C10H16 | 1060 | 0.60 | – |
| Terpinolene | C10H16 | 1089 | 0.62 | 0.23 |
| Linalool | C10H18O | 1097 | 2.08 | 1.67 |
| Chrysanthenone | C10H14O | 1128 | 0.32 | – |
| Unknown | – | – | – | 0.15 |
| Camphor | C10H16O | 1146 | 8.97 | 15.69 |
| Pinocarvone | C10H14O | 1165 | 0.11 | – |
| Borneol | C10H18O | 1169 | 7.16 | 9.15 |
| Terpinen-4-ol | C10H18O | 1177 | 0.60 | 1.79 |
| α-Terpineol | C10H18O | 1189 | 1.72 | – |
| Verbenone | C10H14O | 1205 | 13.09 | 5.17 |
| Geraniol | C10H18O | 1253 | 0.55 | 1.32 |
| Unknown | – | – | – | 1.45 |
| Unknown | – | – | – | 1.13 |
| Bornyl acetate | C12H20O2 | 1289 | 2.07 | 1.29 |
| Thymol | C10H14O | 1290 | – | 1.71 |
| Carvacrol | C10H14O | 1299 | 0.21 | 2.07 |
| Unknown | – | – | – | 0.12 |
| Piperitenone | C10H14O | 1343 | – | 1.85 |
| α-Copaene | C15H24 | 1377 | 0.10 | – |
| Geranyl acetate | C12H20O2 | 1381 | 0.11 | 2.49 |
| β- Caryophyllene | C15H24 | 1419 | 0.36 | 6.85 |
| α-Humulene | C15H24 | 1455 | 0.12 | 3.49 |
| Germacrene D | C15H24 | 1485 | 0.10 | – |
| β-Selinene | C15H24 | 1490 | – | 1.86 |
| Valencene | C15H24 | 1496 | – | 0.81 |
| β-Bisabolene | C15H24 | 1506 | 0.11 | 1.35 |
| δ-Cadinene | C15H24 | 1523 | 0.40 | – |
| Caryophyllene oxide | C15H24O | 1583 | 1.40 | 2.87 |
| Globulol | C15H26O | 1585 | – | 1.71 |
| Caryophylla-4 (12), 8 (13)–dien-5-β-ol | C15H24O | 1641 | – | 2.17 |
| Unknown | – | – | – | 1.67 |
| Grouped components |
Table 1: (continued)
| Compounds | Formula | RI | HD(%) | SFE(%) |
| Hydrocarbon compounds | 43.58 | 15.75 | ||
| Oxygenated compounds | 56.33 | 76.00 | ||
| Monoterpene hydrocarbons | 42.39 | 1.39 | ||
| Oxygenated monoterpenes | 50.27 | 65.47 | ||
| Sesquiterpene hydrocarbons | 1.19 | 14.36 | ||
| Oxygenated sesquiterpenes | 1.40 | 6.75 | ||
| Others | 4.66 | 3.78 | ||
| Total identified (%) | 99.91 | 91.75 | ||
| Extraction time (min) | 240 | 35 | ||
| Oil yield (ml /100 g dry wt.) | 1.14 | 0.75 |
Comparatively, the main constituents of the oil in supercritical fluid extraction technique were α-pinene (0.47%), 1,8-cineole (20.05%), Verbenone (5.17%), Camphor (15.69%), Borneol (9.15%) and β- Caryophyllene (6.85%). Furthermore, the oil consists of 76% oxygenated compounds and 1.39% monoterpene hydrocarbons.
Fig.1 shows the major compounds in Rosmarinus officinalis L. essential oil extracted by hydrodistillation and supercritical fluid extraction, it was noticed that a large amount of α-Pinene was present in the essential oil via hydrodistillation.
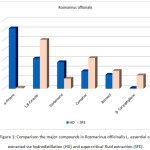 |
Figure 1: Comparison the major compounds in Rosmarinus officinalis L. essential oil extracted via hydrodistillation (HD) and supercritical fluid extraction (SFE). |
Additionally, higher levels of oxygenated compounds and lower levels of monoterpene hydrocarbons were found in the essential oil extracted by supercritical fluid extraction (Fig. 2). The selectivity of supercritical fluid extraction technique allowed to increase the quantities of oxygenated compounds.
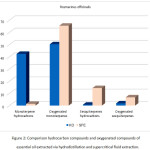 |
Figure 2: Comparison hydrocarbon compounds and oxygenated compounds of essential oil extracted via hydrodistillation and supercritical fluid extraction. Click here to View figure |
Conclusion
The supercritical fluid extraction of Rosmarinus officinalis L. was investigated, and the results, were compared with essential oil composition obtained via hydrodistillation. The SFE technique proposes well-known advantages over hydrodistillation. SFE needs shorter extraction times and less energy consumption. The possibility of influencing the composition of the oil, by altering the parameters of the extraction (pressure, temperature, modifier volume and dynamic extraction time) is more manageable in SFE. We obtained a higher selectivity in SFE than by the subsequent hydrodistillation technique. Besides, the use of polar co-solvent is not necessary and the subsequent processing for solvent elimination is not required.
References
- Bousbia, N.; Vian, M. A.; Ferhat, M. A.; Petitcolas, E.; Meklati, B. Y.; Chemat, F. Food Chem. 2009, 114, 355–362.
CrossRef - Jacotet-Navarro, M.; Rombaut, N.; Fabiano-Tixier, A. S.; Danguien, M.; Bily, A.; Chemat, F. Ultrason. Sonochem. 2015, 27, 102-105.
CrossRef
- Jarrar, N.; Abu-Hijleh, A.; Adwan, K. Asian Pac. J. Trop. Med. 2010, 3, 121-123.
CrossRef - Okoh, O. O.; Sadimenko, A. P.; Afolayan, A. J. Food Chem. 2010, 120, 308-312.
CrossRef - Wang, W.; Wu, N.; Zu, Y. G.; Fu, Y. J. Food Chem. 2008, 108, 1019-1022.
CrossRef - González-Vallinas, M.; Molina, S.; Vicente, G. Pharmacol. Res. 2013, 72, 61-68.
CrossRef - Hernández, M. D.; Sotomayor, J. A.; Hernández, Á.; Jordán, M. J. Chapter 77 – Rosemary (Rosmarinus officinalis L.) Oils,” in Essential Oils in Food Preservation, Flavor and Safety. 2016, 677-688.
- Lemos, M. F.; Lemos, M. F.; Pacheco, H. P.; Endringer, D. C.; Scherer, R. Ind. Crops Prod. 2015, 70, 41-47.
CrossRef - Li, G.; Cervelli, C.; Ruffoni, B.; Shachter, A.; Dudai, N. Ind. Crops Prod. 2016, 84, 381-390.
CrossRef - Fornari, T.; Vicente, G.; Vázquez, E.; García-Risco, M. R.; Reglero, G. J. Chromatogr. A. 2012, 1250, 34–48.
CrossRef - Zabot, G. L.; Meireles, M. A. A. J. Supercrit. Fluids. 2014, 94, 234-244.
CrossRef - R. P. Adams, R. P. Identification of essential oil components by gas chromatography/mass spectrometry, no. Ed. 4. Allured publishing co. 2007.
- Carvalho Jr.,R. N.; Moura, L. S.; Rosa, P.T.V.; M. A. A. Meireles, M. A. A. J. Supercrit. Fluids. 2005, 35, 197–204.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.









