Application of ZnO-Fe3O4 Nano Composite on the Phosphateremovalfrom Aqueous Solutions: Optimization and Statistical Modeling
Hamzeh Ali Jamali1, Cobra Hosseini2, Mehdi Shirzad Siboni2, Negar Pourvakhshoori 3, Mohsen Poursadeghiyan3 and Mehrdad Farrokhi3,2
1Environmental Health Department, school of Health, Qazvin University of Medical sciences, Qazvin, Iran.
2Health and Environment Research Center, school of Health, GuilanUniversity of Medical sciences, Rasht, Iran.
3Health in emergencies and Disasters Research Center, University of Social Welfare and Rehabilitation Sciences, Tehran, Iran.
Corresponding Author E-mail: mfarokhikhb@yahoo.com
DOI : http://dx.doi.org/10.13005/ojc/330419
Removal of nutrient salts from the receiving water is an important consideration to control eutrophication.Among various nano sized materials, ZnO and Fe3O4 have much attention because ZnO nanoparticles exhibit efficient photocatalytic removal efficiency through adsorption and decomposition of organic contaminants. In this study, commercial magnetite was immobilized with ZnO by chemical wet method and it was used as an adsorbent to remove phosphate from aqueous solution. The phosphate adsorption can be well modeled by the Response Surface Methodology. The removal efficiency of phosphate was optimized as the function of adsorbent dosage, pH, initial phosphate concentration, and contact time, using Response Surface methodology. Maximum adsorption of ZnO–Fe3O4 was obtained at pH of3.1, contact time of 53.3 min, adsorbent dosage of 1.35 g/L and initial phosphate concentration of 36.2 mg/L. An empirical model for phosphate removal was developed as quadratic equation. Results from experiments will agree to results from model. This agreement confirms accuracy and approach of obtained model
KEYWORDS:Nanocomposite; ZnO–Fe3O4; RSM; Optimization; statistical modeling; phosphate removal
Download this article as:| Copy the following to cite this article: Jamali H. A, Hosseini C, Siboni M. S, Pourvakhshoori N, Poursadeghiyan M, Farrokhi M. Application of ZnO-Fe3O4 Nano Composite on the Phosphateremovalfrom Aqueous Solutions: Optimization and Statistical Modeling. Orient J Chem 2017;33(4). |
| Copy the following to cite this URL: Jamali H. A, Hosseini C, Siboni M. S, Pourvakhshoori N, Poursadeghiyan M, Farrokhi M. Application of ZnO-Fe3O4 Nano Composite on the Phosphateremovalfrom Aqueous Solutions: Optimization and Statistical Modeling. Orient J Chem 2017;33(4). Available from: http://www.orientjchem.org/?p=35465 |
Introduction
Due to increasing population and decreasing water supplies, wastewater treatment is becoming necessary throughout the world to conserve water resources (1).
Phosphorus (P) exists in water in both particulate and dissolved forms. The usual forms of P in aqueous solutions are orthophosphates, polyphosphates, and organic phosphates Phosphate is an essential macronutrient that spurs the growth of photosynthetic algae and cyanobacteria and it is contained in the sewage of houses and factories and its concentration in lakes and seas rapidly increased because of the use of organic detergent(2).Superfluous phosphorus contained in wastewater from municipal, industrial and agricultural activities can cause eutrophication and hence deteriorate water quality(3,). Organic pollution in closed water bodies such as bays and lakes has been increasing by eutrophication, and removal of nutrient salts from the receiving water is an important consideration to control eutrophication (4).So, the removal of phosphates from Wastewater discharges to water bodies is an important component of eutrophication control strategies(5). As regulations and standards become increasingly more stringent, from 0.5 to 1.0 mg/L to even less than0.03 mg/L P, even further treatment of the effluent is required to meet effluent quality standards (6). Among the conventional phosphorus removal methods, sorption methods using the appropriate solid materials can be promising at lower phosphate concentrations, which pose a challenge to the use of the traditional flocculation methods (7). Many kinds of materials have the potential to remove phosphorus from water, and many materials including waste products have attracted widespread interest due to their potential ability for phosphorus remove, including nZVI (8),Clayey peat(9), Laterite and Red earth(6)Clinoptilolite(10),ACF-Nano HFO (11),)ZrO2 (12) sands (13), fly ash (14), slag (14), iron oxide tailings (15), Ca-based sorbents (16), alunite(17), RM, RM700, RMAH(16), Ca-dolomite, Raw-dolomite (16)and iron-based compounds(7,).Among various nano sized materials, ZnO and Fe3O4 have much attention because ZnO nanoparticles exhibit an efficient photo catalytic removal efficiency through adsorption and decomposition of organic contaminants (18).Today Most of the studies about phosphate adsorbents have focused on the on the nanoparticles due to the importance of selecting substrates with the highest P adsorption capacity to maximize phosphorus removal. In addition, super paramagnetic Fe3O4 nanoparticles possess a promising adsorption capacity for contaminants along with optimal magnetic properties, showing rapid separation of the adsorbent from solution via a magnetic field (19, 20).
Response Surface Methodology (RSM) is a statistical modeling technique employed for multiple regression analysis. In this method influences of individual factors as well as their complex interactive influences can be evaluated. One of the most important benefits of RSM is the reduction of the number of tests and experiments (21)
In recent year optimization of adsorption, biosorption and oxidation processes for different wastewater and synthetic wastewater using RSM have been studied, but only few researchers report about optimization of phosphate removal by nanoparticles using RSM. The most of reports have been focused on Kinetics and equilibrium studies (19, 20).
Hence, in this paper the adsorption of phosphate (PO43–) using zinc oxide immobilized on magnetite nanoparticles was considered in aqueous solutions and application of Response Surface Methodology (RSM) and Central Composite Design (CCD) for optimization of adsorption process have been investigated. Central composite design is one of the common methods of RSM model that is useful and applicable for design of experiments (22). In this method every parameter defined in different levels and in comparison to other experiments design methods have more ability to predict responses.
The main objectives of this study were characterization of ZnO-Fe3O4nano composite and optimization of phosphate adsorption process by it using Response Surface Methodology.
Materials and Methods
Materials
Potassium dihydrogen phosphate, Zinc chloride (99.5 %),sodium hydroxide, hydrogen peroxide, sodium chloride, sodium sulfate, sodium hydrogen carbonate, sodium carbonate, and hydrochloric acid, which were of analytical grade, were purchased from Merck, Germany and used without any purification. nanopowder (Fe3O4) was obtained from the Iranian nanomaterial Pioneer Company in Mashhad city from Iran. It has an approximately spherical shape, is nonporous, and has greater than 99.5 % purity. The specific surface area of the Fe3O4 particles was known as 81.98 m2/g by company. The average size of the spherical particles was 15–20 nm. The chemical structure of phosphate has been presented in Table 1. The initial pH of solution was adjusted by the addition of 0.1MNaOH or HCl and measured by pH meter (Metron, Switzerland). The experiments were carried out at room temperature (25±2°C).
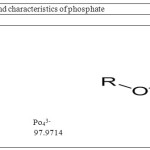 |
Table 1: The structure and characteristics of phosphate |
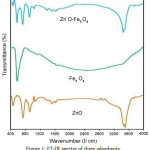 |
Figure 1: FT-IR spectra of three adsorbents
|
Immobilization of ZnO Nanoparticles on Fe3O4Nanoparticles
The Fe3O4–ZnO were prepared by commercial magnetite were coated with ZnO by chemical wet method. Fe3O4 was dried at 103°C for 3 h in an oven. ZnCl2 was used as a starting agent, and NaOH was used as a precipitant. A stock solution of 0.1 M ZnCl2 was prepared by dissolution of ZnCl2 into deionized water. Then an alkaline stock solution of 0.2 M NaOH was prepared in deionized water. Fe3O4 was added to the ZnCl2 solution n the volume ratio of 1:1. The synthetic reaction was fundamentally performed with stirring for 7 h. The products in aqueous solution were centrifuged (4,000 rpm; Sigma-301, Germany), washed with deionized water, and then dried at 100 °C for 3 h (23).
The pHpzc of ZnO–Fe3O4 composite was determined using the procedure as follows a 1-L solution of 0.1 M NaNO3 was prepared and divided into ten solutions with pH ranging from 2 to 11 adjusted by HCl and NaOH with suitable molarity. Thereafter, 0.2 g of the ZnO–TiO2 composite was added to the solutions. The mixture was shaken at 170 rpm for 48 h. Then, the
samples were centrifuged, and the pH of each solution was measured. The final pH values versus initial pH values were plotted to obtain pHzpc. The pHpzc can be determined at the point where the line of final pH is crossing the line of the initial pH.
 |
Figure 2: Typical XRD patterns of three adsorbents. Click here to View figure |
Characterization
The X-ray diffraction (XRD) studies were performed with a Philips XRD instrument (Siemens D-5000, ermany) using Cu Kα radiation (λ=1.5406 Å) at wide-angle range (2θ value 4–70°), an accelerating voltage of 40 kV, and an emission current of 30 mA. For characterization of the functional groups on the surface of the samples, Fourier transform infrared spectroscopy (FT-IR) spectra were recorded on a Perkin-Elmer (Germany) spectrometer under a dry air at room temperature by the KBr pellets method. The spectra were collected over the range from 400 to 4,000 cm−1. The surface morphology of ZnO, Fe3O4, and ZnO–Fe3O4 composite were obtained by field emission scanning electron microscopy (FE-SEM), which was carried out by a Mira microscope (Mira3, Tescan, Czech Republic). SEM images were further supported by energy dispersive X-ray (EDX)
 |
Figure 3: SEM image of (a) ZnO, (b) Fe3O4, and (c) ZnO-Fe3O4 composite. Click here to View figure |
Micro analysis to provide direct evidence for the purity, existence, and distribution of specific elements in a solid sample.Fig.1shows Infrared spectrum that shows Fe-O transplantation along the lower waves (700cm-1) and the resulting peaks along 3600 through 3200 cm-1 wave belong to O-H transplantations. The transplantations in the two bands of 406-512cm-1belong to ZnO. Fig.2 shows the diffraction of X-ray by means of spectrometer (XRD).
The main peaks at 2θ values of 31.72, 34.4, 36.2, 47.49, 56.51, 62.8, 66.28, 67.86, and 68.86 correspond to the (100), (002), (101), (102), (110), (103), (200), (112), and (201) planes of hexagonal wurtziteZnO (JCPDS card no. 36–1451).The main peaks at 2θ values of 18.27, 21.16, 30.11, 30.21, 35.42, 35.53, 37.03, 37.18, 43.12, and 57.09 correspond to the (011), (002), (112), (200), (121), (103), (022), (202), (004), and (321) planes of the orthorhombic Fe3O4 (JCPDS card no. 031156) (9). It can be seen from Fig. 2, after immobilizing the ZnO, the peaks related to the ZnO are still observed, which indicates growth of the ZnO crystal on the Fe3O4 nanoparticles.
SEM images of ZnO nanoparticles, Fe3O4nanoparticles, and ZnO–Fe3O4composite are shown in Fig.3a–c, respectively. ZnO was shaped as rods as shown in Fig.3a,c. As shown in Fig.3b, c,ZnO–Fe3O4composite was composed of spherical Fe3O4nanoparticles and ZnOnanorods.
EDX microanalysis was used to characterize the elemental composition of the ZnO–Fe3O4 composite. EDX pattern of the ZnO–Fe3O4 composite is depicted in Fig. 4.
According to the results of EDX analysis, the major elements were Zn, O, and Fe, indicating good hybridization between ZnO and Fe3O4.
Experimental Design
The phosphate solution was prepared by dissolving an accurately weighed sample of potassium dihydrogen phosphate (KH2PO4, anhydrous) in deionized water. Batch sorption experiments were performed in order to obtain factors for preparation and equilibrium data.
The adsorption experiments were performed in a 1000 Ml Erlenmeyer flask containing 30 ML of phosphate solution and mixtures were stirred at 150 RPM in different time interval. In order to determine the effects of various parameters, the experiments were conducted at different adsorbent amounts of 0.4 to 2 g/L, initial phosphate concentrations of 10 to 90 mg/L as pre tests and initial pH of 3 to 11. Removal efficiency of phosphate was calculated with Eq. 1.
![]()
Where, C0 and Ce (mg/L) are the initial and equilibrium liquid phase phosphate concentrations respectively.
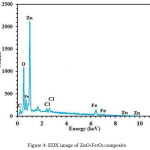 |
Figure 4: EDX image of ZnO-Fe3O4 composite Click here to View figure |
The solution pH was adjusted by HCl (0.10 M) or NaOH (0.10 M) solutions, and Na2SO42-, NaCl, NaHCO32-or NaCO3– solutions were added as sources of the competing anions when necessary. All experiments were carried out at room temperature which was the same to the application environment. The concentrations of the phosphate solutions were determined by the stannous chloride method (4500-P D)(28). The absorbance of the solutions was analyzed using spectrophotometer (UV/Vis Spectrophotometer, Hach-DR 5000) (24). The pH of the solution was determined by pH meter (Metron, Switzerland). The adsorption capability ( , mg/g) of phosphate in Zno-Fe3O4 was calculated by the following Eq. 2.
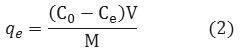
Where , V (L) is the volume of the solution and M (g) is the mass of the adsorbent.
Design Expert software (Design Expert version 7.1.o) was used for statistical analysis of experimental data.Design Expert is a statistical software package from Stat-Ease Inc. that is specifically devoted to performing design of experiments and offers comparative tests, screening, characterization, optimization, robust parameter design, mixture designs and combined designs(25). To optimize four main variables (Contact time, pH, ZnO-Fe3O4 doses and phosphate concentration) due to the influence on the response (phosphate removal), Response Surface Methodology (RSM) and Central Composite Design (CCD) were used. Independent variables including the Contact time(A), pH(B), ZnO-Fe3O4 doses(C) and phosphate concentration(D) were set at five coded level: -1(minimum, -0.5, 0(center point), +0.5, and +1(maximum). Preliminary tests were done for determination of narrower and effective range before designing the research. In order to obtain the optimum values of four independent variables, phosphate removal was analyzed as a response. The quadratic model as Eq. 3 has been used for analysis of experimental data and prediction of optimum condition:
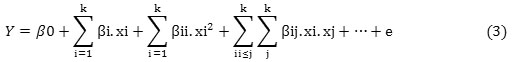
Where:
Y = Response, I = Linear constant, j = Quadratic constant, β = Regression constant
k = Number of studied factors and, e = Random error
Six experiments were repeated in central point for evaluation of pure error. Analysis of variances (ANOVA) was used for graphical analyses of the data to obtain the interaction between the process variables and the responses.ANOVA is a collection of statistical models used to analyze the differences among group means and their associated procedures such as “variation” among and between groups (26). The quality of model fitness determined by R2 (coefficient of determination) and p-value with confidence level of 95% was used for influences of individual variables as well as their interactive influences evaluation. To control the significance and Adequacy of model, calculated F-value (Fisher variation ratio), probability value and Adequate precision were evaluated. Finally to find optimum condition of adsorption for phosphate removal, response variables have been compared in a numerical optimization, where all independent variables selected in “in range” and phosphate removal selected in “maximize”.
Results and Discussion
In this experimental study 30 run of CCD were performed that results are shown in Table2. Table 2 shows Central composite design for the study of four experimental variables for phosphate removal efficiency and obtained results.
To be significant of model components is necessary to develop a proper model. CCD showed in table 2 has been used for mathematical equations for prediction of results (Y) as function of contact time (A), pH (B), ZnO-Fe3O4 dose(gr/L (C) and phosphate concentration (mg/L) (D) , their reciprocal effects, include: (AB, AC, AD, BC, BD, DC) and second order effects(A2, B2, C2 and D2). Analysis of variances (ANOVA), have been used for graphical analysis of data and to determine reciprocal effects of independent variables and response. The equation from the first analysis of variances was modified by elimination of insignificant terms. Table 3 shows data quadratic model with independent variables and other statistical
Table 2: Central composite design for the study of four experimental variables for phosphate removal efficiency and obtained results
|
Std |
Run number |
Factors |
Phosphate removal efficiency (%) |
||||
|
A-Contact timea(code) |
B-pH(code) |
C-ZnO-Fe3O4 doseb(code) |
D-Phosphate Concentrationc(code) |
Actual value |
Predicted value |
||
|
25 |
1 |
50(0) |
3(0) |
1(0) |
30(0) |
89 |
93.66 |
|
22 |
2 |
50(0) |
3(0) |
1.25(+0.5) |
30(0) |
98 |
101.43 |
|
2 |
3 |
60(+1) |
2.5(-1) |
0.5(-1) |
15(-1) |
76 |
77.44 |
|
24 |
4 |
50(0) |
3(0) |
1(0) |
37.5(+0.5) |
91 |
93.71 |
|
12 |
5 |
60(+1) |
3.5(+1) |
0.5(-1) |
45(+1) |
78 |
75.2 |
|
18 |
6 |
55(+0.5) |
3(0) |
1(0) |
30(0) |
87 |
88.62 |
|
30 |
7 |
50(0) |
3(0) |
1(0) |
30(0) |
92 |
93.66 |
|
11 |
8 |
40(-1) |
3.5(+1) |
0.5(-01) |
45(+1) |
65 |
66.36 |
|
10 |
9 |
60(+1) |
2.5(-1) |
0.5(-01) |
45(+1) |
84 |
82.86 |
|
17 |
10 |
45(-0.5) |
3(0) |
1(0) |
30(0) |
82 |
80.4 |
|
3 |
11 |
40(-1) |
3.5(+1) |
0.5(-01) |
15(-1) |
63 |
62.44 |
|
13 |
12 |
40(-1) |
2.5(-1) |
1.5(+1) |
45(+1) |
67 |
66.63 |
|
8 |
13 |
60(+1) |
3.5(+1) |
1.5(+1) |
15(-1) |
98 |
92.5 |
|
19 |
14 |
50(0) |
2.75(-0.5) |
1(0) |
30(0) |
90 |
90.4 |
|
9 |
15 |
40(-1) |
2.5(-1) |
0.5(-01) |
45(+1) |
48 |
50.8 |
|
5 |
16 |
40(-1) |
2.5(-1) |
1.5(+1) |
15(-1) |
75 |
75.22 |
|
16 |
17 |
60(+1) |
3.5(+1) |
1.5(+1) |
45(+1) |
58 |
62.5 |
|
6 |
18 |
60(+1) |
2.5(-1) |
1.5(+1) |
15(-1) |
98 |
99.27 |
|
23 |
19 |
50(0) |
3(0) |
1(0) |
22.5(-0.5) |
100 |
97.31 |
|
29 |
20 |
50(0) |
3(0) |
1(0) |
30(0) |
98 |
94 |
|
27 |
21 |
50(0) |
3(0) |
1(0) |
30(0) |
95 |
94 |
|
20 |
22 |
50(0) |
3.25(+0.5) |
1(0) |
30(0) |
93 |
92.7 |
|
14 |
23 |
60(+1) |
2.5(-1) |
1.5(+1) |
45(+1) |
83 |
81 |
|
4 |
24 |
60(+1) |
3.5(+1) |
0.5(-01) |
15(-1) |
78 |
81 |
|
1 |
25 |
40(-1) |
2.5(-1) |
0.5(-01) |
15(-1) |
38 |
35.65 |
|
26 |
26 |
50(0) |
3(0) |
1(0) |
30(0) |
96 |
94 |
|
7 |
27 |
40(-1) |
3.5(+1) |
1.5(+1) |
15(-1) |
88 |
91 |
|
28 |
28 |
50(0) |
3(0) |
1(0) |
30(0) |
92 |
93 |
|
15 |
29 |
40(-1) |
3.5(+1) |
1.5(+1) |
45(+) |
76 |
72 |
|
21 |
30 |
50(0) |
3(0) |
0.75(-0.5) |
30(0) |
98 |
95 |
a Unit of time:min; b unit of dosage: g/L; c unit of concentration: mg/L parameter.
As shown in table 3 the ANOVA of the quadratic regression model confirmed that the model is significant. High R2 coefficient (R2>0.95 and have a good agreement with Adj.R2 (Adj.R2=0.94) shows a satisfactory adjustment of quadratic models to experimental results (27).Adequate Precision (AP) of model for phosphate removal was 24.1 that is greater than 4 that indicated adequate signal and it is concluded that predicted data could be used to navigate design space defined by the CCD (26). F-value of model obtained from ratio of mean square of regression to mean square of residual. As shown in table 3, F value and P- value (pro>F) indicate that the phosphate removal model is significant again. It can be seen that P-value is very small (P-value<0.0001) and the less P-value the more significant model.
Based on the sequential model sum of squares, the phosphate removal models, was selected based on the highest order polynomials where the additional terms were significant and the model was not aliased. The model of phosphate removal efficiency was represented as Y, The quadratic model for term of Y was selected as suggested by the software and is shown in Eq. 4. The independent variables in the model were contact time, pH, ZnO-Fe3O4 dose and phosphate concentration.
Table 3: ANOVA results for response parameters
|
Source |
Sum of square |
Df |
Mean square |
F-Value |
P-Value |
|||||||
|
Pro.>F |
||||||||||||
|
Model |
7015.49 |
14 |
510.11 |
33.56 |
<0.0001S |
|||||||
|
A-contact time |
1112.74 |
1 |
1112.74 |
74.52 |
<0.0001 S |
|||||||
|
B-pH |
80.74 |
1 |
80.74 |
5.41 |
0.0345 S |
|||||||
|
C-ZnO-Fe3O4 |
773.88 |
1 |
773.88 |
51.83 |
<0.0001 S |
|||||||
|
D-Phosphate concentration |
214.56 |
1 |
214.56 |
14.37 |
0.0018 S |
|||||||
|
AB |
540.56 |
1 |
540.56 |
36.2 |
<0.0001 S |
|||||||
|
AC |
315.06 |
1 |
315.06 |
21.1 |
0.0004 S |
|||||||
|
AD |
95.06 |
1 |
95.06 |
6.37 |
0.0234 S |
|||||||
|
BC |
105.06 |
1 |
105.06 |
7.04 |
0.0181S |
|||||||
|
BD |
126.56 |
1 |
126.56 |
8.48 |
0.0107S |
|||||||
|
CD |
564.06 |
1 |
564.06 |
37.78 |
<0.0001S |
|||||||
|
A2 |
222.75 |
1 |
222.75 |
14.92 |
0.0015S |
|||||||
|
B2 |
12.27 |
1 |
12.27 |
0.82 |
0.3790 NS |
|||||||
|
C2 |
50.47 |
1 |
50.47 |
3.38 |
0.0859 NS |
|||||||
|
D2 |
9.15 |
1 |
9.15 |
0.61 |
0.4460 NS |
|||||||
|
Residual |
223.97 |
15 |
14.93 |
|||||||||
|
Lack of fit |
170.64 |
10 |
17.06 |
1.6 |
0.3151NS |
|||||||
|
Pure error |
53.33 |
5 |
10.67 |
|||||||||
|
Core Total |
7239.47 |
29 |
||||||||||
|
Other statistical parameters |
||||||||||||
|
Mean=82.47 |
S.D.=3.86 |
C.V.=4.69 |
PRESS=1451 |
R2=0.97 |
Adj.R2=0.94 |
Pred.R2=0.8 |
A.P=24.1 |
|||||
S: Significance: NS: Not significance; S.D: Standard deviation C.V: Coefficient of variance; PREES: Predicted residual error sum of squares; A.P: Adequate precision
All insignificant terms from statistical point of view were removed and the empirical model for phosphate removal as dependent variable was resulted as equation 4:
Y= 93.65 + 8.21A + 2.2B + 6.84C – 3.6D -5.18AB – 4.44AC – 2.44AD – 2.56BC – 2.81BD – 5.94CD – 36.59A2 (4)
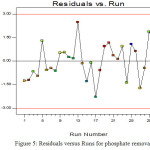 |
Figure 5: Residuals versus Runs for phosphate removal Click here to View figure |
Figure 5 shows residuals versus runs for phosphate removal. This plot can be used to study independence of residuals. If there were sinusoidal variations therefore, data independence would be acceptable (26). As shown in Fig. 5, there are no evidences for rejection of data independence hypothesis and this is concluded that selected model is suitable for data analysis
The Reciprocal Effects of All Independent Variables on Phosphate Removal
Figure 6 shows Contour plot for phosphate removal as a function of contact time and pH(a), contact time and ZnO-Fe3O4(b), contact time and phosphate concentration(c), pH and ZnO-Fe3O4(d), pH and phosphate concentration(e) and ZnO-Fe3 and phosphate concentration(e)
Contour plots show the reciprocal effects of tow variables while, other are constant. This kind of plot indicates a correct geometrical shape of reciprocal effects of variables and they will give useful information about system in the range of experimental design. Optimization of process defined as setting the levels of variables to achieve maximum phosphate removal.
It should be considered that elliptical curve shows a strong reciprocal effect between tow variables and circular curve shows no relationship between variables (27).
As shown in fig 6 all plots are as elliptical curves that indicate there are reciprocal effects among variables.
The effects of contact time on phosphate removal are shown in figures 6- a, 6-b and 6-c. As shown in figures 6-a, 6-b and 6-c phosphate removal increase to the time and gradually decreased until reaction reaches a near equilibrium after 53.3min. the high removal at the initial time can be attributed to the abundance of free active sites on the surface of ZnO- Fe2O3and this trend would be
As the active sites are occupied by phosphate, adsorption rates are decreased due to having little available active sites on the adsorbents (23).The effects of primary concentration of phosphate on removal efficiency are shown in figures 6-c, 6-e and 6-f. As it can be seen from the figures, the efficiencies decrease from 100 to 77.11 with the increase of concentration from 15 to 45 mg/l (this range of concentration was selected for development of model).
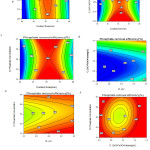 |
Figure 6: Contour plot for phosphate removal as a function of (a) contact time and pH, (b)contact time and ZnO-Fe3O4, (c)contact time and phosphate concentration, (d) pH and ZnO-Fe3O4, (e) pH and phosphate concentration and (e) ZnO-Fe3 and phosphate concentration |
The increase of adsorption capacity or decrease of adsorption efficiency in this case is probably due to stability of active locations on the adsorbent against the increase of phosphate numbers, In other words saturation of adsorbent surface in higher concentrations of contaminant. Another reason could be the particle response such as accumulation and association due to higher concentrations of adsorbent since accumulation results in decrease of active surface of absorbent. Figures 6-a 6-d and 6-e show phosphate removal as a function of pH.
The effect of primary pH of solution on the phosphate removal efficiency was studied in pH of 3 to 11. It can be seen that removal efficiencies decrease of the pH increases from 3 to 11. The results from the fact that, the solution pH plays an important role in the adsorption process so that the adsorption process in low pH has higher efficiencies in absorbing contaminants compared to alkaline pH. The higher efficiency of adsorption in acidic pH could be due to electrostatic adsorption between negative loaded phosphate and the surface with positive load of adsorbent which might be as a dominant solution. Also in lower pHs, H+ ion increases on surface. But in higher pH, the adsorbent surface receive a negative load, hence the electrostatic repulsive force between ions gets more negative loads and the electrostatic repulsive force between ions with negative load of absorbed material and the surface with negative load of adsorbent has reduced the adsorption phosphate.
It should be considered thatthe pH range of wastewater, surface water and groundwater is normally higher than 3.5, therefore pH of effluent must be neutralized before to discharge to the environment or reuse.
The effect of adsorbent dose on the removal efficiency was studied with the primary adsorbent dose of 2-0.4 gr/l. the results of effects of adsorbent doses on the remove efficiency are shown in Fig.6-b, 6-d and 6-f. As it can be seen, when adsorbent dose increases from 0.2 to 2 gr/l, the remove efficiency increases from 86.77 to 100 percent, really the removal efficiency increases with the adsorbent dose increase. The increase of adsorption percent in this case is due to increase of adsorption positions available for phosphate. But decrease of adsorption capacity with the increase of adsorbent amount might stem from unsaturation of active positions on adsorbent surface during the adsorption process.
In this study factors including the contact time, pH, -ZnO-Fe3O4 dose and phosphate concentration were selected in range and response (phosphate removal) was selected as maximize. Results from numerical optimization showed that in contact time of 53.3 min, pH = 3.1, ZnO-Fe3O4 dose of 1.35 g/L and phosphate concentration of 36.2 mg/L, the phosphate removal efficiency were 100% . In this condition desirability was 100% (Fig. 7).
Five excess tests were performed at the optimum point to confirm adequacy and validity of optimization procedure. The results of these tests are given in Table 4. As shown in Table 4 results from experiments will agree to results from model. This agreement confirms accuracy and approach of model.
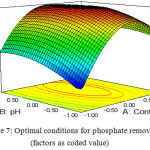 |
Figure 7: Optimal conditions for phosphate removal (factors as coded value) Click here to View figure |
Table 4: Verification experiments at optimum conditions
|
Optimum conditions |
Responses |
Error |
S.D |
|
|
Predicted |
Actual |
|||
|
ZnO-Fe3O4 =1.35g/L |
100 |
97.6 |
2.4 |
±3.5 |
|
Contact time=53.3min |
||||
|
pH=3.1 |
||||
|
Phosphate concentration=36.2mg/L |
||||
Conclusions
In the current study, commercial magnetite was coated with ZnO using chemical wet method. The phosphate adsorption can be well modeled by the Response Surface Methodology. The removal efficiency of phosphate was optimized. Maximum adsorption of ZnO–Fe3O4 was obtained at pH of 3.1, contact time of 53.3 min, adsorbent dosage of 1.35 g/L and initial phosphate concentration of 36.2 mg/L. An empirical model for phosphate removal was developed as quadratic equation. Results from experiments will agree to results from model. This agreement confirms accuracy and approach of obtained
Acknowledgments
Authors would like to thankGuilan University of Medical Sciences of Iran for their contributions and funding.
Reference
- Jafari, J;Mesdaghinia, A.;Nabizadeh, R.;Farrokhi, M.;Mahvi, A.H. Investigation of anaerobic fluidized bed reactor/ aerobic moving bed bio reactor (AFBR/MMBR) system for treatment of currant wastewater. Iranian Journal of Public Health.2013, 8(42), 860-867
- Mezenner,NY.; Bensmaili,A. Kinetics and thermodynamic study of phosphate adsorption on iron hydroxide–eggshell waste. J of Chem Eng. 2009, 147, 87–96.
CrossRef - Anirudhan,T.S.; Noeline,B.F.; Manohar.D.M.; Phosphate removal from wastewaters using a weak anion exchanger prepared from a lignocellulosicresidue.Environ.Sci.Technol. 200640, 2740–2745.
- Danalewich,J.R.;Papagiannis,T.G.;Belyea,R.L.;Tumbleson,M.E.,Raskin,L.Characterization of dairy wastestreams, current treatment practices, and potential for biological nutrient removal. Water Res. 1998, 32, 3555–3568.
CrossRef - Kwon, H.B.; Lee,C.W.; Jun,B.S.; Yun,J.D.; Weon,S.Y.; Koopman. B. Recycling waste oyster shells for eutrophication control. Resource Conservation and Recycling.2004, 41(1), 75-82.
CrossRef - H.M.T.G.A.; PITAWALA.; D.G.G.P.; KARUNARATNE.; N. SILVA.Removal of aqueous phosphate and phenol by adsorption on clayey peat.laterite and red earth. 2013,15, 57-67.
- Seida,Y.; Nakano,Y. Removal of phosphate by layered double hydroxides containing iron, Water Res. 2002. 36, 1306–1312.
CrossRef - Osama Eljamal.;Ahmed M.E. Khalil.; Yuji Sugihara.; NobuhiroMatsunaga. Phosphorus removal from aqueous solution by nanoscale zero valent iron in the presence of copper chloride.chemical Engineering Journal. 2016, 293, 225-231
- Zhao,D.Y.; A.K.,Sengupta.Ultimate removal of phosphate from wastewater using a new class of polymeric ion exchangers.Water Res. 1998, 32, 1613–1625.
CrossRef - Raj T,Rehana.; Rajeev kumar,K. Synthesis of ZnO Nanostructures Using Domestic Microwave Oven Based Remote Plasma Deposition System.Nano science and nanotechnology.2012, 2, 66-70 DOI: 10.5923/j.nn.20120203.04.
CrossRef - Malakootian,M.;Yousefi,N.;Haghighifard,NA.; Kinetics Modeling and Isotherms for Adsorption of Phosphate from Aqueossolotion by modified Cilinoptilolite.J of water and WasteWater2011, 480, 21-29.
- Zhou,Q.;Wang,X.; Liu J- Zhang,L. “Phosphorus removal from wastewater using nano-particulates of hydratedferric oxide doped activated carbon fiber prepared by Sol–Gel method. J of chemical engineering.2012,202, 619-626.
CrossRef - Liu,H.;Sun,X.;Yin,Ch.;Hu,Ch.Removal of phosphate by mesoporous ZrO2. J of Hazardous Materials.2008, 151, 616-622.
CrossRef - Arias,C.A.; Del Bubba,M.; Brix,H. Phosphorus removal by sands for use as media in subsurface flow constructed reed beds, Water Res. 2001,35,1159–1168.
CrossRef - Agyei,N.M.;Strydom,C.A.;Potgieter,J.H. The removal of phosphate ionsfrom aqueous solution by fly ash, slag, ordinary Portland cement and relatedblends, Cem.Concr. Res. 2002, 32, 1889–1897.
CrossRef - Zeng,L.; Li,X.M.; Liu,J.D. Adsorptive removal of phosphate from aqueous solutions using iron oxide tailings, Water Res. 2004, 38, 1318–1326.
CrossRef - Karaca,S.; Gurses,A.; Ejder,M.; Acikyildiz,M. Adsorptive removal of phosphate from aqueous solutions using raw and calcinated dolomite, J of Hazard. Mater.2006, 128, 273–279.
CrossRef - Ozcar M.Adsorption of phosphate from aqueous solution onto alunite. Chemosphere.2003, 51(4),321-327
CrossRef - M.A, BEHNAJADY; N. MODIRSHAHLA; M. SHOKRI, B. RAD. Enhancement of photocatalytic activity of TiO2 nanoparticles by silver doping: photodeposition versus liquid impregnation methods, Global NEST Journal. 2008, 10(1) 1-7
- Chen,C.;Gunawan,P.;Xu,R. Self-assembledFe3O4-layered double hydroxide colloidal nanohybridswith excellent performance for treatment of organic dyes in water. J of Materials Chemistry.2011, 21, 1218–1225.
CrossRef - Geng,Z.; Lin,Y.; Yu,X.; Shen,Q.; Ma,L.;Li,Z. Highly efficient dye adsorption and removal: a functional hybrid of reduced graphene oxide-Fe3O4 nanoparticles as an easily regenerative adsorbent. J of Materials Chemistry.2012, 22, 3527–3535.
CrossRef - Moradi, M.; F. Ghanbari. Application of response surface method for coagulation processin leachate treatment as pretreatment for Fenton process: Biodegradability improvement.J. Water Process Eng. 2014, 4, 67-73.
CrossRef - Mason, R.L.; R.F. Gunst, J.; L.Hess. Statistical Design and Analysis of Experiments:With Applications to Engineering and Science.2nd Edition., John Wiley and Sons Inc., CanadaISBN: 9780471372165,,2003, 728.
CrossRef - M Farrokhi.; SC Hosseini.; JK Yang.; M Shirzad-Siboni.Application of ZnO–Fe3O4 Nanocomposite on the Removal of Azo Dye from Aqueous Solutions: Kinetics and Equilibrium Studies, Water, Air, & Soil Pollution.2014,225(9), 1-12
CrossRef - APHA,Standard methods for the examination of water and wastewater. 20 edition. 1998.
- Douglas C.Montgomery.Design and Analysis of Experiments, 8th Edition, John Wiley and Sons Inc; 8th edition 2012.
- Raymond H. Myers.; Douglas C. Montgomery. Christine M. Anderson-Cook, “Response Surface Methodology: Process and Product Optimization Using Designed Experiments,” John Wiley & Sons Inc; 3 edition 2009.
- Trinh, T.K.; Kang, L.S. Application of response surface method as an experimental design to optimize coagulation tests. Environmental Engineering Re-search.2010, 15, 63-70.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.









