Copper(II) Carboxylate Containing Paddle Wheel Structure: Synthesis and Crystal Structure
Adnan M. Qadir
Department of Chemistry, Collage of Science, Salahaddin University, Erbil 44001, Kurdistan Region, Iraq.
Corresponding Author E-mail: aq701998@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/330249
Article Received on : January 09, 2017
Article Accepted on : March 22, 2017
A dinuclear copper(II) complex of the type [Cu2(2-nb)4(MeOH)2] (2-nb = 2-nitrobenzoate) has been synthesized and characterized by single crystal X-ray diffraction method. The compound belongs to monoclinic space group P21/c with a = 19.9374(10) ٫ b = 21.1404(11) ٫c = 7.6533(4) Å, α = 90٫ β = 96.817(3)٫ γ = 90º٫ V = 3246.8(3) Å 3٫ Z = 4٫ F(000) = 1736.0.0٫ Dc = 1.774Mg/m3, μ = 1.422 mm-1. The Cu(II) complex is a cocrystal containing two inorganic units. Each Cu(II) adopted square pyramidal geometry.
KEYWORDS:Paddle wheel; Nickel; Synthesis; Carboxylate
Download this article as:| Copy the following to cite this article: Qadir A. M. Copper(II) Carboxylate Containing Paddle Wheel Structure: Synthesis and Crystal Structure. Orient J Chem 2017;33(2). |
| Copy the following to cite this URL: Qadir A. M. Copper(II) Carboxylate Containing Paddle Wheel Structure: Synthesis and Crystal Structure. Orient J Chem 2017;33(2). Available from: http://www.orientjchem.org/?p=31049 |
Introduction
Generally, most copper(II) carboxylates display the dinuclear paddle-wheel cage structure which consists of two copper centers bridged by four carboxylate molecules and each copper(II) is attached to a neutral ligand . The dimeric copper (II) carboxylates with the formula [Cu2(RCOO)4 L2] are known to contain two or more antiferromagnetically coupled metal centers [1-4]. Dinuclear copper(II) compounds with four syn–syn bridging carboxylato ligands are of special interest due to their fungicidal activity which should arise from the unique coordination sphere around central copper(II) ions[5]. A paddle-wheel structure type was first reported in 1953 for the case of cupric acetate dehydrate [6]. Carboxylate ligands can coordinate to metal center in monodenate, bidentate or bridging mode[7]. Copper is a significant transition metal in biology and coordination chemistry [8,9]. Dinuclear copper centres are present in the oxygen breathing protein hemocyanin and in a number of enzymes which have oxidase or oxygenase function[10] . Different members of metalloproteins of the type-3 with binuclear copper sites are known: hemocyanins, tyrosinase, catechol oxidase with bridging oxygen atoms and cytochrome coxidase with bridging sulfur atoms. In most of these,copper is ligated by histidine and cysteine amino acids[11,12]. In this work, we report synthesis and crystal structure of binuclear Cu(II) complex containing 2-nitrobenzoic acid. The complex exhibited a paddlewheel structure.
Experimental
Synthesis
A mixture of 2-nitrobenzoic acid (0.302 g, 2 mmol) and K2CO3(0.138 g, 1 mmol) in H2O (10ml) was added to Cu(NO3)2 (0.188 g, 1 mmol) in H2O (10ml). The mixture was stirred for 30 minutes. The precipitate formed was filtered and recrystallized from MeOH.
Crystal structure determination
Single crystal of the title compound with dimensions of 0.44 mm × 0.42 mm × 0.12 mm was mounted on Bruker APEX-II CCD diffractometer equipped with a graphite-monochromated MoKα radiation (λ = 0.71073 Å) at 100.0(2) K. The compound crystallizes in monoclinic space group P21/c with a = 19.9374(10) ٫ b = 21.1404(11) ٫c = 7.6533(4) Å α =90٫ β = 96.817(3)٫ γ = 90º٫ V = 3246.8(3) Å3٫ Z = 4٫ F(000) = 1736.0.0٫ Dc = 1.774Mg/m3, μ = 1.422 mm-1.The structure was solved by direct method using XS solution program and refined by least-squares techniques using XL [13] refinement package. Empirical absorptions were applied to all intensity data. All hydrogen atoms were placed in geometrically calculated positions and allowed to ride on the parent carbon atoms. The full final matrix least square refinement gave R= 0.0356 and wR= 0.0805.
Results and Discussion
Crystal Structure
Crystal structure of the Cu(II) complex is depicted in Fig. 1. Crystallographic data and refinement are given in Table-1. Selected bond lengths and angles are given in Table-2.
The title compound belongs to monoclinic P21/c space group with four molecules in the unit cell. The Cu(II) complex is cocrystal with two neutral inorganic components (1 and 1′). In both components, two Cu(II) centers are bridged by four carboxylate groups of the four ligands.
Table 1: Crystallographic Data And Refinement For Cu(Ii) Complex
|
Crystal data |
|
|
Chemical formula |
C30H24Cu2N4O18 |
|
Formula weight |
855.61 |
|
Cell system, space group |
monoclinic, P21/c |
|
a (Å) |
19.9374(10) |
|
b (Å) |
21.1404(11) |
|
c (Å) |
7.6533(4) |
|
α(°) |
90 |
|
β(°) |
96.817(3) |
|
γ(°) |
90 |
|
Volume (Å3) |
3246.8(3) |
|
Z |
4 |
|
Dc(Mg m–3) |
1.774 |
|
μ (mm-1) |
1.422 |
|
Crystal size (mm) |
0.44 × 0.42 × 0.12 |
|
2Θ range for data collection |
5.638 to 69.496 |
|
Index ranges |
-27 ≤ h ≤ 31, -33 ≤ k ≤ 33, -12 ≤ l ≤ 10 |
|
Reflection collected |
75054 |
|
Independent reflections |
12506 [Rint = 0.0470, Rsigma = 0.0465] |
|
Data/restraints/parameters |
12506/6/495 |
|
F (000) |
1736 |
|
Goodness-of-fit on F2 |
1.054 |
|
R1 [I > 2σ(I)] |
R1 = 0.0356 |
|
wR2 [I > 2σ(I)] |
wR2 = 0.0805 |
|
R1 (all data) |
R1 = 0.0626 |
|
wR2 (all data) |
wR2 = 0.0897 |
|
Min. and max. resd. dens. [e/Å3], |
0.46/-0.81 |
In compound 1, Cu1 has distorted square-pyramidal geometry coordinated by O1,O2,O3 and O4 from four carboxylate molecules in the basal plan and O5 from MeOH molecule in the apical position. The angular and structural parameter (τ) is 0.16.The bond lengths Cu1-O1, Cu1-O2, Cu1-O3 and Cu1-O4 are 1.9671(10), 1.9722(10), 1.9670(10) and 1.9638(10) Å, respectively, while the apical bond length Cu1-O5 is 2.1451(10) Å. The values are in agreement with bond distances for other binuclear Cu(II) complex. The bond angles O5-Cu1-Ocarboxylate range from 93.19(4) to 97.78(4)°.The Cu1-Cu11bond distance is 2.6210 (4) Å in the dinuclear unit.
In compound 1′, Cu1′ has almost regular square-pyramidal geometry (τ =0.0105) coordinated by O1′,O2′,O3′ and O4′ from four carboxylate ligands in the basal plan and O5′ from MeOH molecule in the apical position. The Cu1′-Ocarboxylate bond lengths range 1.9586(11) from to 1.9798(10) Å, while the apical bond length Cu1′-O5′ is 2.1898(11) Å. The bond angles O5′-Cu1′-O1′, O5′-Cu1′-O2′, O5′-Cu1′-O3′ and O5′-Cu1′-O4′ are 88.06(4), 93.63(4), 102.69(4) and 96.84(4)°, respectively. The Cu1′-Cu1′2 bond distance is 2.6210 (4) Å.
Table 2: Selected Bond Lengths (Å) and Angles (°) For The Complex
|
Bond length (Å) |
|||
| Cu1-O1Cu1-O2Cu1-O3Cu1-O4Cu1-O5Cu1-Cu11 | 1.9671(10)1.9722(10)1.9670(10)1.9638(10)2.1451(10)2.6210(4) | Cu1′-O1’Cu1′-O2’Cu1′-O3’Cu1′-O4’Cu1′-O5’Cu1′-Cu1′2 | 1.9622(10)1.9658(10)1.9586(11)1.9798(10)2.1898(11)2.6134(4) |
|
Bond angle (°) |
|||
| O5-Cu1-O1O5-Cu1-O2O5-Cu1-O3O5-Cu1-O4O1-Cu1-O3O2-Cu1-O4 | 97.15(4)93.19(4)93.60(4)97.78(4)169.18(4)169.02(4) | O5′-Cu1′-O1’O5′-Cu1′-O2’O5′-Cu1′-O3’O5′-Cu1′-O4’O1′-Cu1′-O3’O2′-Cu1′-O4′ | 88.06(4)93.63(4)102.69(4)96.84(4)168.88(4)169.51(4) |
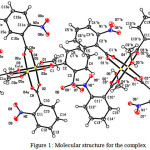 |
Figure 1: Molecular structure for the complex
|
Supplementary Material
CCDC 1420202 contains the supplementary crystallographic data for this paper. These data can be obtained free of charge at www.ccdc.cam.ac.uk/conts/retrieving.html or from the Cambridge Crystallographic Data Centre (CCDC), 12 Union Road, Cambridge CB2 1EZ, UK (Fax: +44-1223-336033; email: deposit@ccdc.cam.ac.uk or www:http://www.ccdc.cam.ac.uk).
Reference
- Chadjistamatis, I.; Terzis, A.; Raptopoulou, C.P. and Perlepes, S.P. Inorg. Chem. Commun. 2003, 6,1365 .
CrossRef - Baranwal, B.P.; Das,S.S. and Singh, P. Synth. React. Inorg. Met. Org. Chem.1998,28, 1689.
CrossRef - Baran, E.J.; Etcheverry, S.B.; Torre M.H. and Kremer, E. Polyhedron 1994,13,1859.
CrossRef - Campbell and Haw, J.F. Inorg Chem 1998, 27,3706.
- Kasuni, M.; nik,K.G.; Egedin, P. and Golobi, A. Acta Chim. Slov. 2010 57, 350.
- Niekerk, J.N.V. and Schoening, F.R.L. Acta Cryst. 1953, 6,227.
CrossRef - Qadir, A.M. Asian J. Chem. 2013, 25, 8829.
CrossRef - Thomas, A.M.; Nethaji, M.; Mahadevan,S. and Chakravarty, A.R. Inorg. Biochem. 2003, 94,171.
CrossRef - Zhang, L.; Niu, S.Y.; Jin, J.; Sun, L.P.; Yang G.D. and Yie, L. Chem. J. Chin. Universities 2009, 30, 236.
- Bullock, G.; Hartstock,F.W. and Thomps, L. Can. J . Chem.1983, 61, 57.
CrossRef - Solomon, E.I.; Sundaram U.M. and Machonkin, T.E. Chem. Rev. 1996, 96, 2563.
CrossRef - Borghi, E. and Solari, P.L. J. Synchrotron Rad. 2005, 12, 102.
CrossRef - Shldrick, M. Acta crystallogr. A 2008, 64, 112.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.









