Synthesis, Characterization and Antibacterial Studies on Mn(II) and Fe(II) Complexes of N, O Donor Salicyloylpyrazoleoxime Schiff Bases
Namdeo T. Dhokale1, Bhausaheb K. Karale2 and Arvind V. Nagawade1
1Department of Chemistry, Ahmednagar College, Ahmednagar (MS) India.
2Department of Chemistry, Radhabai Kale Mahila Mahavidyalaya, Ahmednagar (MS) India.
Corresponding Author E–mail: avnagawade@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/330118
Mn (II) and Fe(II) complexes with Schiff bases substituted salicyloyl pyrazole oximes were synthesized by the reaction of CFPHMP, FPHMP, DCFPHMP, CFPHMMP and BFPHMP (see introduction for definitions) with transition metal ion Manganese (Mn2+) and Iron (Fe2+). They were characterized by analytical and spectral methods. The elemental analysis data revealed that the Schiff base metal (II) complexes have 1:2 (M:L) stoichiometry. The molar conductivity data showed them to be non electrolytes. The Schiff bases are bidentate coordinating through the imine nitrogen and phenolic oxygen of salicyloyl pyrazole oximes. Based on analytical and spectral data, four-coordinate geometry is assigned for all complexes. The electronic absorption spectra suggest the square planer geometry for the complexes. The ligand salicyloyl pyrazole oximes and their Mn (II) and Fe (II) complexes were screened against bacillus subtilis, staphylococcus aureus, actinomycetes and pseudomonas.
KEYWORDS:Antibacterial; elemental analysis; transition metal complexes; square planer; electrolytes
Download this article as:| Copy the following to cite this article: Dhokale N. T, Karale B. K, Nagawade A. V. Synthesis, Characterization and Antibacterial Studies on Mn(II) and Fe(II) Complexes of N, O Donor Salicyloylpyrazoleoxime Schiff Bases. Orient J Chem 2017;33(1). |
| Copy the following to cite this URL: Dhokale N. T, Karale B. K, Nagawade A. V. Synthesis, Characterization and Antibacterial Studies on Mn(II) and Fe(II) Complexes of N, O Donor Salicyloylpyrazoleoxime Schiff Bases. Orient J Chem 2017;33(1). Available from: http://www.orientjchem.org/?p=29761 |
Introduction
Hydroxyoximes are special class of Schiff base ligands which contains hydroxyl group in addition to imine functionality. It is easy to make coordinate bond with bivalent metal through Oxygen atom of phenol by deprotonation and through Nitrogen atom of imine group. The oxime group also plays an important role to promote inter and intra molecular hydrogen bond, to stabilize metal-organic molecule framework and used in extractive metallurgy and separation of metal ions1-4. The chemistry of oxime metal complexes is interesting because of these species shows variety of reactivity modes. They have been widely studied in variety of biochemical5,6 and analytical applications7,8.
The oxime group attached to the pyrazole ring is very good donor group because of its potential to facilitate different coordination modes. Also it is well known that N atom of oxime group involve in bond formation with metal atom and O atom forms bridge between two organic molecules through hydrogen bond. A pyrazole oxime ligand contains two different bridging functions9-11. A number of pyrazole derivatives show biological activity as antipyretic 12, antimicrobial13 and antitumor activity14. Pyrazole derivatives are also used in agriculture as pesticides15-16.
Fluorine substituted heterocyclic compounds and their analogues are accepted by enzymes as substrate because fluorine is comparable with steric demand to hydrogen. When fluorine attached to molecule it increases the activity of molecules. Fluorinated analogue of natural occurring nucleic acid are found to associate with various biological activity such as, antiviral17, antitumor18-19, antifungal20 and antimicrobial activity21. All literature survey suggest that, the transition metal containing pyrazole moiety and fluorine substituent has great importance in the field of chemistry and large applications in agriculture, industrial and biological studies.
In this present study we report synthesis and characterization of Manganese (Mn2+) and Iron (Fe2+) complexes with N and O donor Schiff bases, 4-chloro-2-[(E)-[1-(4-fluorophenyl)-1H-pyrazol-4-yl](hydroxyimino)methyl]phenol, (CFPHMP), 2-[(E)-[1-(4-fluorophenyl)-1H-pyrazol-4-yl](hydroxyimino)methyl]-4-methylphenol (FPHMP), 2,4-dichloro-6-[(E)-[1-(4-fluorophenyl)-1H-pyrazol-4-yl](hydroxyimino)methyl]phenol (DCFPHMP), 4-chloro-2-[(E)-[1-(4-fluorophenyl)-1H-pyrazol-4-yl](hydroxyimino)methyl]-5-methylphenol (CFPHMMP) and 4-bromo-2-[(E)-[1-(4-fluorophenyl)-1H-pyrazol-4-yl](hydroxyimino)methyl]phenol (BFPHMP). The antibacterial activity of Schiff bases and their manganese (Mn2+) and Iron (Fe2+) complexes were tested against bacillus subtilis, staphylococcus aureus, actinomycetes and pseudomonas.
Material and methods
Most of the chemicals used were analytical grade. The solvents were research grade and used after purification. MnSO4 and FeSO4 were used without purification. Both water and ethyl alcohol were distilled before use. The ligand salicyloyl pyrazole oxime was prepared using the method reported in our previous investigation22. Melting points of the complexes were determined in open glass capillaries and are uncorrected. The conductance of synthesized complexes was measured using 10-3 M solution of complexes in DMF on Elico digital conductivity meter (Model CM-180). The elemental analysis was recorded on Thermo Scientific (FLASH 2000) elemental analyzer and the FTIR spectra were recorded in the range 4000-350 cm-1 on Shimadzu FTIR spectrophotometer. The thermal analysis was recorded on Shimadzu Thermometric Analyzer (TGA-50). UV-Visible spectra were recorded on Shimadzu UV-Visible double beam spectrophotometer (UV-1800). The percentages of metal ions in complex were determined using standard volumetric methods 23.
Synthesis of metal complexes
Metal sulphate (One equivalent) was dissolved in distilled water and acidified by concentrated Hydrochloric acid. The resulting acidic solution was made warm using hot water bath. It was further treated with alcoholic ligand CFPHMP, FPHMP, DCFPHMP, CFPHMMP and BFPHMP solution (2 equivalents). The addition of ligand solution was made slowly drop after drop. Slight excess of ligand solution was added to ensure the complete complexation. The resulting mixed solution was then treated with alcoholic ammonia to make it alkaline. The solution was then digested on boiling water bath where colored complex precipitated out. The product was filtered, washed first with little hot distilled water and then by ethyl alcohol to remove excess of ligand. It was dried under ambient conditions.
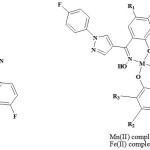 |
Scheme 1: Structures of Schiff bases and their Mn (II) and Fe (II) complexes. |
| Compound | R1 | R2 | R3 |
| Ia, Ib, Ic | Cl | H | H |
| IIa, IIb, IIc | CH3 | H | H |
| IIIA, IIIb, IIIc | Cl | H | Cl |
| IVa, IVb, IVc | Cl | CH3 | H |
| Va, Vb, Vc | Br | H | H |
Result and Discussion
The Schiff base Mn (II) complexes (Ib-Vb) and Fe (II) complexes (Ic-Vc) were synthesized and characterized by various spectroscopic techniques. The synthesized compounds are non hygroscopic, stable solids, insoluble in water and other solvents. All the complexes are soluble in DMF and DMSO. The analytical data and molar conductivity data of metal complexes are presented in Table-1 and Table-2. The elemental analysis data of the metal complexes are consistent with their general formulation ML2. The Mn (II) complexes are dark shiny brown in color where as Fe (II) complexes are brown in color. The colors of complexes are different from the color of ligand indicating that formation of color depends on type of metal ion. The melting points of complexes are different than that of ligand is an evidence for complexetion. The metal complexes were dissolved in DMF and the molar conductivities of their 10-3 M solutions were measured at room temperature on conductivity meter. The low molar conductivity values of metal complexes suggest their non electrolytic nature24. The structures of the complexes were predicted based on analytical, spectroscopic and thermal measurements.
Table 1: Physical, Analytical and molar conductance of Mn (II) complexes.
| Complex | M. P. (0C) | % Yield | Found (Calcd.) % | ΛM | |||
| M | C | H | N | ||||
| Mn(CFPHMP)2 (Ib) | >300 | 90 | 7.49(7.67) | 54.04(53.65) | 3.11(2.81) | 11.98(11.75) | 17.3 |
| Mn(FPHMP)2 (IIb) | 224-226 | 82 | 7.97(8.13) | 60.03(60.45) | 3.46(3.88) | 12.61(12.44) | 12.1 |
| Mn(DCFPHMP)2 (IIIb) | 288-290 | 74 | 6.28(7.0) | 48.41(48.94) | 2.65(2.31) | 10.51(10.70) | 17.3 |
| Mn(CFPHMMP)2 (IVb) | 202-204 | 82 | 7.97(7.38) | 54.61(54.86) | 3.59(3.25) | 11.01(11.29) | 19.9 |
| Mn(BFPHMP)2 (Vb) | 212-214 | 76 | 6.76(6.82) | 47.57(47.73) | 2.94(2.50) | 10.22(10.44) | 35.4 |
Table 2: Physical, Analytical and Molar Conductance of Fe (II) Complexes.
| Complex | M. P. (0C) | % Yield | Found (Calcd.) % | ΛM | |||
| M | C | H | N | ||||
| Fe(CFPHMP)2 (Ic) | >300 | 93 | 7.07(7.79) | 53.88(53.58) | 2.71(2.81) | 11.54(11.72) | 60.6 |
| Fe(FPHMP)2 (IIc) | 280-282 | 77 | 8.93(8.26) | 60.11(60.37) | 3.99(3.87) | 12.86(12.42) | 26.5 |
| Fe(DCFPHMP)2 (IIIc) | 222-224 | 75 | 7.44(7.10) | 48.51(48.89) | 2.70(2.31) | 10.47(10.69) | 33.3 |
| Fe(CFPHMMP)2 (IVc) | 224-226 | 65 | 7.89(7.49) | 54.34(54.79) | 3.55(3.25) | 10.93(11.28) | 33.6 |
| Fe(BFPHMP)2 (Vc) | 234-236 | 70 | 7.14(6.93) | 47.91(47.67) | 2.86(2.50) | 10.23(10.42) | 32.5 |
IR spectra
The FTIR spectra of metal complexes were recorded over the range 4000-350 cm-1. The most important bands in IR spectra of free ligand and its metal complexes are presented in table-3 and table-4. To study the binding mode of ligand and metal, IR spectra of free ligand and corresponding metal complexes were compared. The important vibrational bands have been assigned on the basis of reported assignments25-26. The free ligand exhibit broad band in the 3391-3248 cm-1 range assignable to free ν(O-H) stretching frequency which is obviously absent in the spectra of metal complexes indicating deprotonation and involved in the formation of metal oxygen bond of phenolic group. The band in the 3166-3131 cm-1 range assignable to hydrogen bonded ν(O-H) stretching frequency and shifting to lower frequency, indicative of the coordination of nitrogen atom and strong hydrogen bonding. Evidence for this coordination also arise from the small shift towards lower frequency of the band in the 1563-1542 cm-1 range assigned to ν(C=N) stretching and ν(N-O) stretching in the 1282-1226 cm-1range. The absorption peaks due to ν(C-O) observed in the 1097-1073 cm-1 region. The spectra of free ligand moved towards low frequency in the spectra of the complexes. This supports bonding of the metal ions to the phenolic OH after deprotonation27-28. The oxygen and nitrogen coordination to the metal ion is proved by the bands that appears in the range 557-540 cm-1 and 464-447 cm-1 assigned to ν(M-O) and ν(M-N) respectively29-30
Table 3: The significant peaks in FTIR spectra of free ligand and its Mn(II) complexes.
|
Compound |
νO-H |
νC=N |
νN-O |
νC-O |
νM-O |
νM-N |
||||||
|
a |
b |
a |
b |
a |
b |
a |
b |
a |
b |
a |
b |
|
|
I |
3391, 3131 |
3165 |
1563 |
1541 |
1232 |
1232 |
1097 |
1053 |
– |
540 |
– |
464 |
|
II |
3350, 3134 |
3169 |
1542 |
1544 |
1231 |
1247 |
1097 |
1095 |
– |
549 |
– |
462 |
|
III |
3310, 3166 |
3161 |
1545 |
1539 |
1242 |
1230 |
1088 |
1083 |
– |
545 |
– |
451 |
|
IV |
3327, 3157 |
3153 |
1548 |
1543 |
1226 |
1230 |
1093 |
1097 |
– |
553 |
– |
457 |
|
V |
3248, 3163 |
3142 |
1546 |
1537 |
1282 |
1230 |
1073 |
1097 |
– |
555 |
– |
460 |
Table 4: The significant peaks in FTIR spectra of free ligand and its Fe(II) complexes.
|
Compound |
νO-H |
νC=N |
νN-O |
νC-O |
νM-O |
νM-N |
||||||
|
a |
c |
a |
c |
a |
c |
a |
c |
a |
c |
a |
c |
|
|
I |
3391, 3131 |
3162 |
1563 |
1545 |
1232 |
1233 |
1097 |
1057 |
– |
546 |
– |
461 |
|
II |
3350, 3134 |
3147 |
1542 |
1543 |
1231 |
1228 |
1097 |
1095 |
– |
549 |
– |
455 |
|
III |
3310, 3166 |
3130 |
1545 |
1539 |
1242 |
1228 |
1088 |
1082 |
– |
551 |
– |
447 |
|
IV |
3327, 3157 |
3138 |
1548 |
1539 |
1226 |
1230 |
1093 |
1095 |
– |
557 |
– |
459 |
|
V |
3248, 3163 |
3157 |
1546 |
1539 |
1282 |
1230 |
1073 |
1095 |
– |
553 |
– |
447 |
Thermal Analysis
Thermal behaviors of complexes have been studied using TGA in air as atmosphere. Thermogravimetric analysis of metal complexes was carried out from 300C to 9000C at heating rate of 100C per minutes. All the complexes investigated shows similar behavior in their thermograms. According to Thermogravimetric data of manganese and iron complexes exhibit high stability and melt with decomposition in the range 200-2800C. In TGA thermograms of complexes does not show any significant weight loss up to 2000C indicating there is no lattice or coordinated water molecules. Like most of the organic metal complexes Mn (II) and Fe (II) complexes of salicyloyl pyrazole oxime decompose by producing fine powder. During decomposition gaseous products such as CO2, NH3 etc. formed as a result of fragmentation of ligand. Further increase in temperature, the complexes decompose slowly involving loss of ligand in the temperature range 600-850 0C leading to formation of air stable metal oxide as the end product.
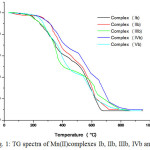 |
Figure 1: TG spectra of Mn(II)complexes Ib, IIb, IIIb, IVb and Vb. |
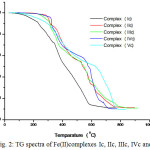 |
Figure 2: TG spectra of Fe(II)complexes Ic, IIc, IIIc, IVc and Vc. |
Electronic spectra
The electronic spectra in ultra violet and visible region of selected ligands and their complexes were recorded. The solution of known concentration (1 x 10-4M) of ligands and its corresponding complexes prepared in DMf and their UV-Visible spectra were recorded in the region 190-700nm. The ligand shows absorption band at 300-350 nm is assigned to π →π* of the aromatic chromophore. In addition to the band observe in the range 273-290 nm can be attributed to n→π* transitions. In metal complexes exhibits two bands in the region 20,704 – 17,271 cm-1 and 15,924- 15,948cm-1) attributed to d→d transitions. This is probable indicative of four coordinate square planer geometry31.
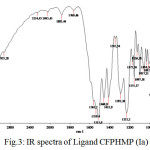 |
Figure 3: IR spectra of Ligand CFPHMP (Ia) |
1H NMR spectra of all synthesized complexes recorded in DMSO as solvent and TMS as internal standard. However due to presence of metal ion, proton resonance was not effected and gave broad peaks indicating the formation of metal complexes.
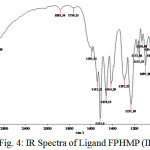 |
Figure 4: IR Spectra of Ligand FPHMP (IIa) Click here to View figure |
XRD pattern of metal complexes were obtained in solid form. The X-ray diffractogram of all complexes showed broad peak, which indicate polycrystalline nature32. Though polycrystalline nature of complexes was observed they were generally not soluble in non polar solvents.
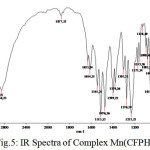 |
Figure 5: IR Spectra of Complex Mn(CFPHMP)2 (Ib) |
Biological Activity
The antibacterial activity of salicyloyl pyrazole oxime and their metal complexes were screened against bacillus subtilis, staphylococcus aureus, actinomycetesand pseudomonas by the well diffusion method33. The test solutions were prepared in DMF, nutrient agar was used as culture medium. The zone of inhibition was measured in mm and presented table-5. All compounds were dissolved DMF at 100µg/ml. DMF was used as solvent and also for control. The result showed that the complex IIIa had highest effect and Ia, IVb lowest effect against B. Subtilis than all other compounds. The metal complexes had higher antibacterial activity than free ligands against S. Aureus. Also in Actinomycetes the metal complexes had higher activity than free ligands except complex IIIa, IIIb and Vb while the complex Ib showed highest antibacterial activity. All the free ligands and their Mn (II) and Fe (II) complexes showed similar activity against Pseudomonas. The dissimilarity in the activity of the different metal complexes against different bacteria depends either on the impermeability of the cells of the microbes or difference in the ribosomes in the microbial cells34-35.
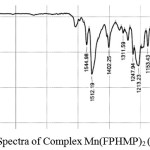 |
Figure 6: IR Spectra of Complex Mn(FPHMP)2 (IIb) |
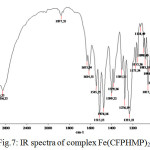 |
Figure 7: IR spectra of complex Fe(CFPHMP)2 (Ic) |
Table 5: Antibacterial effect of free ligands and their Mn (II) and Fe (II) complexes against bacillus subtilis, Staphylococcus aureus, actinomycetesand pseudomonas.
| Compound | Zone of inhibition in mm | |||
| B. Subtilis | S. Aureus | Actinomycetes | Pseudomonas | |
| I | 19 | 12 | 13 | 22 |
| II | 18 | 15 | 17 | 25 |
| III | 16 | 17 | 14 | 27 |
| IV | 18 | 16 | 16 | 31 |
| V | 18 | 12 | 17 | 23 |
| Ia | 12 | 15 | 24 | 26 |
| IIa | 16 | 18 | 23 | 24 |
| IIIa | 26 | 20 | 14 | 21 |
| IVa | 15 | 18 | 18 | 28 |
| Va | 14 | 21 | 17 | 30 |
| Ib | 17 | 15 | 28 | 25 |
| IIb | 15 | 23 | 19 | 28 |
| IIIb | 18 | 24 | 12 | 27 |
| IVb | 12 | 19 | 17 | 26 |
| Vb | 16 | 19 | 15 | 29 |
| Control | 08 | 10 | 10 | 20 |
 |
Figure 8: IR spectra of complex Fe(FPHMP)2 (IIc) |
Conclusion
The available experimental data suggests that prepared salicyloyl pyrazole oxime ligands posses two coordination sites. The physical and spectroscopic characterization of the complex reveled that oxygen atom of phenol group and nitrogen atom of oxime group are involved in the coordination with the metal ion. The elemental analysis data suggest that the Schiff base metal (II) complexes have general formula ML2, Where M is Mn (II) and Fe (II).The complexes have good solubility in DMF and DMSO but were found to be insoluble in water and other organic solvents. The thermal analysis data suggested that complexes have high stability below 2000C and decomposes slowly after 2000C giving formation of corresponding metal oxides. The electronic absorption spectra reveled probably square planer geometry for the complexes. The magnitudes of molar conductance of these complexes suggested that complexes are non electrolytic in nature. The antibacterial data reveled that ligand and their metal complexes show considerable antimicrobial activity. However, the zone of inhibition of ligand varies with organisms as well as metal ions. Thus it can be concluded that most of the ligands metal complexes posses moderate antimicrobial activity.
Acknowledgment
Authors are thankful to SAIF, Punjab University, Chandigarh and Department of Microbiology, K. J. Somaiya College, Kopargaon for providing antimicrobial activity.
References
- Chaudhari, P. CoordHYPERLINK “http://www.sciencedirect.com/science/article/pii/S001085450300081X”. Chem. Rev. 2003, 243 (1-2), 143-190.
- Dong, W. K.; Feng, J. H.; Yang, X. Q. Synth. React. HYPERLINK “http://www.tandfonline.com/doi/full/10.1080/15533170701301587″Inorg.MetHYPERLINK “http://www.tandfonline.com/doi/full/10.1080/15533170701301587”. Org. Nano-Chem. 2007, 37 (3), 189-192.
- Dong, W. K.; Li, L.; Li, C. F.; Xu, L.; Duan, J. G. SpectrochimHYPERLINK “http://www.sciencedirect.com/science/article/pii/S1386142508000759”. HYPERLINK “http://www.sciencedirect.com/science/article/pii/S1386142508000759″ActaHYPERLINK “http://www.sciencedirect.com/science/article/pii/S1386142508000759” Part-A 2008, 71(2), 650-654.
- Sastre, A. M.; Szymanowski, J. J. Solvent HYPERLINK “http://www.tandfonline.com/doi/full/10.1081/SEI-200035610″ExtrHYPERLINK “http://www.tandfonline.com/doi/full/10.1081/SEI-200035610”. HYPERLINK “http://www.tandfonline.com/doi/full/10.1081/SEI-200035610″Ion Exch. 2004, 22 (5), 737-759.
- Luo, C.; Tong, M.; Maxwell, D. M.; Saxena, A. Chem-BiolHYPERLINK “http://www.sciencedirect.com/science/article/pii/S0009279708002469”. Interact. 2008, 175(1-3), 261-266.
- Naur, P.; Petersen, B. L.; Mikkelsen, M. D.; Bak, S.; Rasmussen, H.; Olsen, C. E.; Halkier, B. A. Plant HYPERLINK “http://www.plantphysiol.org/content/133/1/63.full.pdf”Physio. 2003, 133, 63-72.
- Gergely, A.; Gyimesi-Forras, K.; Horvath, P.; Hosztafi, S.; Kokosi, J.; Nagy, P. I.; Szasz, G.; Szentesi, A. CurrHYPERLINK “http://benthamscience.com/journals/current-medicinal-chemistry/volume/11/issue/19/page/2555/”. Med. ChemHYPERLINK “http://benthamscience.com/journals/current-medicinal-chemistry/volume/11/issue/19/page/2555/”. 2004, 11(19), 2555-2564.
- Urbansky, E. T.; Freeman D. M.; Elovitz, M. S. Water Res. 2000, 34 (9), 2610-2613.
CrossRef - Nanda, P. K.; Ray, D. InorgHYPERLINK “http://www.sciencedirect.com/science/article/pii/S0020169305002483”. HYPERLINK “http://www.sciencedirect.com/science/article/pii/S0020169305002483″Chim.Acta. 2005, 358 (13), 4039-4044.
- Milios, C. J.; Kyritsis, P.; Raptopoulou, C. P.; Terzis, A.; Vicente, R.; Escuer A.; Perlepes, S. P. Dalton Trans 2005, 3, 501-511.
CrossRef - Zhan, S.; Hu, C.; Chen, X.; Meng, Q.; Lu, C.; Wang G.; Zheng, P. Polyhedron 1999, 18 (11), 2035-2039.
CrossRef - Ivanenkov, Y. A.; Balakin, K. V.; Tkachenko, S. E. Drugs in R HYPERLINK “http://link.springer.com/article/10.2165/0126839-200809060-00005″&HYPERLINK “http://link.springer.com/article/10.2165/0126839-200809060-00005” HYPERLINK “http://link.springer.com/article/10.2165/0126839-200809060-00005″D 2008, 9 (6), 397-434.
- Almeida da Silva, P. E.; Ramos, D. F.; Bonacorso, H. G.; de la Iglesia, A. I.; Oliveira, M. R.; Coelho, T.; Navarini, J.; Morbidoni, H. R.; Zanatta, N.; Martins, M. A. P. Int. J. HYPERLINK “http://www.sciencedirect.com/science/article/pii/S092485790800143X”AntimicrobHYPERLINK “http://www.sciencedirect.com/science/article/pii/S092485790800143X”. Agents 2008, 32 (2), 139-144.
- Reisner, E.; Arion, V. B.; Keppler B. K.; Pombeiro, A. J. L. InorgHYPERLINK “http://www.sciencedirect.com/science/article/pii/S0020169306007638”. HYPERLINK “http://www.sciencedirect.com/science/article/pii/S0020169306007638″Chim.Acta. 2008, 361 (6), 1569-1583.
- Lemaire, G.; Mnif, W.; Pascussi, J. M.; Pillon, A.; Rabenoelina, F.; Fenet, H.; Gomez, E.; Casellas, C.; Nicolas, J. C.; Cavailles, V.; Duchesne, M. J.; Balaguer, P. ToxHYPERLINK “http://toxsci.oxfordjournals.org/content/91/2/501.full.pdf+html”. Sci. 2006, 91 (2), 501-509.
- Singh, N.; Sangwan N. K.; Dhindsa, K. S. Pest. HYPERLINK “http://onlinelibrary.wiley.com/doi/10.1002/(SICI)1526-4998(200003)56:3%3C284::AID-PS128%3E3.0.CO;2-M/pdf”Manag.Sci. 2000, 56 (2), 284-288.
- Rani, P.; Srivastava, V. K.; Kumar, A. Eur. J. Med. Chem. 2004, 39 (5), 449-452.
CrossRef - Lucey, N. M.; McElhinhey, R. S.; Hoshi, A.; Kimura, K.; Fujji, S.; Ogawa, M.; J. Chem. res.(S), 1985, 240.
- Bodey, G. P.; Alberto, P. Eds. Elsevier Science, 1984.
- Gadakh, A. V.; Pandit, C.; Rindhe, S.; Karale, B. K. BioHYPERLINK “http://www.sciencedirect.com/science/article/pii/S0960894X10009753″orgHYPERLINK “http://www.sciencedirect.com/science/article/pii/S0960894X10009753”. HYPERLINK “http://www.sciencedirect.com/science/article/pii/S0960894X10009753″&HYPERLINK “http://www.sciencedirect.com/science/article/pii/S0960894X10009753” Med. Chem. Let. 2010, 20 (18), 5572-5576
- Basser, M. A.; Jadhav, V. D.; Phule, R. M.; Archana, Y. V.; Vibhute, Y. B. Orient. J. Chem, 2000, 16, 553-556.
- Dhokale, N. T.; Karale B. K.; Nagawade, A. V. Res. J. of Chem. Sci. 2014 Vol. 1(NCCICR-2014), 100-105.
- Vogel’s, Text book of practical organic Chemistry, 5th edition, J. Wiley, New York, 2012.
- Geary, W. J. Coord. Chem. Rev., 1971, 7, 81-122.
CrossRef - Keeney, M. E.; Osseo-Asare, K. Polyhedron 1984, 3 (6), 641-649.
CrossRef - Percy, G. C.; Thornton, D.A. InorgHYPERLINK “http://www.sciencedirect.com/science/article/pii/0020165071800405”. HYPERLINK “http://www.sciencedirect.com/science/article/pii/0020165071800405″andHYPERLINK “http://www.sciencedirect.com/science/article/pii/0020165071800405” HYPERLINK “http://www.sciencedirect.com/science/article/pii/0020165071800405″NuclHYPERLINK “http://www.sciencedirect.com/science/article/pii/0020165071800405”. Chem. HYPERLINK “http://www.sciencedirect.com/science/article/pii/0020165071800405″lettHYPERLINK “http://www.sciencedirect.com/science/article/pii/0020165071800405”. 1971, 7 (7), 599-604.
- Bukhari, I. H.; Arif, M.; Akbar, J.; Khan, A. H. Pakistan J. Biol. Sci., 2005, 8 (4), 614-617.
CrossRef - Basavaraju, B.; BhojyaNaik H. S.; Prabhakasa, M. C. E-Journal Chem., 2007, 4(1), 39-45.
CrossRef - Hamrit, H.; Diebbar-sid, S.; Benali-Baitich, O.; Khan M. A.; Bouet, G. SynthHYPERLINK “http://www.tandfonline.com/doi/abs/10.1080/00945710009351872”. React. HYPERLINK “http://www.tandfonline.com/doi/abs/10.1080/00945710009351872″InorgHYPERLINK “http://www.tandfonline.com/doi/abs/10.1080/00945710009351872”.HYPERLINK “http://www.tandfonline.com/doi/abs/10.1080/00945710009351872” Met-Org. Chem. 2000, 30 (10), 1835-1848.
- Mohapatra, B. B.; Saraf, S. K. J. Ind. Chem. Soc. 2003, 80 (7), 696-699.
- Mehta, B. H.; Swar, Y. A. Asian J. of Chem. 2001, 13 (3), 928-932.
- Hussain, R.; Juneja, H. D. Int. J. HYPERLINK “http://www.sadgurupublications.com/ContentPaper/2009/5_7(2)2009.pdf”Chem. Sci. 2009, 7 (2), 632-638.
- Perez, C.; Pauli, M.; Bazerque, P. Acta. Biol. Med. Exp. 1990, 15, 113-115.
- Kurtoglu, M.; Ispir, E.; Kurtoglu N.; Serin, S. Dyes HYPERLINK “http://www.sciencedirect.com/science/article/pii/S0143720807000745?np=y”Pigm. 2008, 77 (1), 75-80.
- El-Tabl, A. S.; El-Saied, F.A.; Plass, W.; Al-Hakimi, A. N. SHYPERLINK “http://www.sciencedirect.com/science/article/pii/S1386142507006567″pectrchimHYPERLINK “http://www.sciencedirect.com/science/article/pii/S1386142507006567”. HYPERLINK “http://www.sciencedirect.com/science/article/pii/S1386142507006567″Acta. A 2008, 71 (1) 90-99.

This work is licensed under a Creative Commons Attribution 4.0 International License.









