An Investigation of pH Effects on the Properties of the Fabricated Banana Flower Extracts-based Organic Solar Cell
Sutikno, Ian Yulianti and Dany Sigit Saputra
Physics Department, Faculty of Mathematics and Natural Sciences, Universitas Negeri Semarang (Unnes) D7 Building, 2nd Floor, Sekaran Campus, Gunungpati, Semarang, Republic of Indonesia, 50229.
Corresponding Author E-mail: sutiknomadnasri@mail.unnes.ac.id
DOI : http://dx.doi.org/10.13005/ojc/330137
The purpose of this research is to fabricate banana flower extracts-based dye-sensitized solar cell (DSSC) using spin coating method. DSSC is made of photoactive material of banana extract by spincoating method and thermal evaporation, using indium tin oxide (ITO) substrate. Spincoater speed, coating duration and heating temperature are fixed as controlled variables, anthocyanin pH is fixed as independent variable, while optical and electrical properties of solar cell device are fixed as dependent variables. The anthocyanin content decreased as well as the pH increased and the voltage knee also increased. The pH enhance has caused the current generation of device decreased.
KEYWORDS:DSSC; Organic photovoltaics; Photovoltaics; Synthesis method
Download this article as:| Copy the following to cite this article: Sutikno S, Yulianti I, Saputra S, D. An Investigation of pH Effects on the Properties of the Fabricated Banana Flower Extracts-based Organic Solar Cell. Orient J Chem 2017;33(1). |
| Copy the following to cite this URL: Sutikno S, Yulianti I, Saputra S, D. An Investigation of pH Effects on the Properties of the Fabricated Banana Flower Extracts-based Organic Solar Cell. Orient J Chem 2017;33(1). Available from: http://www.orientjchem.org/?p=30354 |
Introduction
Dye-sensitized solar cell (DSSC) is the third generation solar cell which is considered as future alternative energy source, a dye-sensitized photovoltaic cell of TiO2. The basic principle of DSSC is transforming light energy into electrical energy. When a dye molecule absorbs light, an electron is replaced with an excited state, and this can jump to conduction band of TiO2. Equal to this process, the photoelectric effect is obtained by dye pigments [1]. The production cost of silicon based-conventional solar cell is relatively high and it consumes more energy. This device has been attracting many researchers at last decade since the first report made by Gratzel dan coworkers [2] due to DCCS is produced at relatively low cost [1,3], easy prepared and fabricated, simple design structure [4], light, potentially applicable in flexible devices as if compared with conventional pn junction devices [5], transparent module construction, better performance on wider tap, lower sensitivity to the angle of incidence and condition of darkness [6], and has a high conversion efficiency [7]. Silicon based-conventional solar cell has achieved efficiency as high as 20% while highest organic solar cell efficiency ever achieved is 1,49% [8].
The selected material advantages include [1,7,]: available in nature in large quantities [1,4], easy to use, non-toxic, ecofriendly and easily biodegradable. The types of materials that will be developed [2] include flowers, plant leaves, fruits, fruit peels and roots. The photoactive materials extracted from various types of flowers such as red cabbage, blue bean [2], raspberry (Rubus Ideaus), black carrot (Daucuscarota L.), rosella (Hibiscus Sabdariffa L.) [9], red bougenville flower [1], black tea, banana flower [10,11], Allium Cepa L. [11], beans or promegranate skin [5], flowers canaries and radishes. The leaves of plants that can be extracted the photoactive ingredient include: basella alba rubra or malabar spinach or typhoon spinach [7], and mapple leaf. Fruits ever extracted by some researchers to compose DSSC include: prickly pir, beets [5], achoite seed, mulberry, strawberry and blackberry. Anthocyanin also can be extracted of grap skins [11] and bieten roots [12], Beta vulgaris [4], red potatoes, pomea pescaprae, and Imperata cylindrica (L.) Beauv and Paspalum conjugatum Berg [1]. The anthocyanin quantity in the sample decreased as well as it’s pH increased from 3 up to 7 [11]. The pigment is present on the different parts of plants which including flower petals, fruits, leaves, stems, and roots [13]. The acid treatment has caused pigment spectrum absorption spreading and cell efficiency increasing. The acid treatment can increase efficiency of pigment cell I. Pescaprae from 0,45% up to 0,53%. How does the effect pH level of banana anthocyanin extract on the electrical property of the fabricated DSSC device?
Materals and Method
The materials used in this research comprise indium titanium oxide (ITO) substrate 1800Å and sheet resistance 7 W/sq, banana flower extract anthocyanin, aluminium foil, copper (Cu) foil, nitrate ammonium, DI-water, poly ethylene glycol (PEG), ammonia, ethanol, and acetyl acetone. The production equipments consist of spincoater with maximum speed 10.000 rpm, oxidation/vacuum furnace, evaporator, sputtering reactor, ultrasonic cleaning bath, and IV-meter. Additionally, the characterization equipments utilized include FTIR, Ocean Optic Vis-Nir USB4000 spectrophotometer, I-V meter (Four point probe meter) and UV-vis spectrometer.
The organic solar cell structure if seen from the side view is designed as Figure 1. First, the ITO substrate was cleaned using ultrasonic cleaning bath in the acetone isoprophyl alcohol, and then dried using dry nitrogen. A conductive layer of banana flower extract anthocyanin with thickness of 40 mm was coated on the substrate using spincoater. At this step, some parameters were controlled to optimize the conductive layer thickness. The thickness, optical property and microstructure were also optimized. The aluminium electrode in thickness of 120 nm was formed through thermal evaporation at vacuum condition 1,33 x 10-4 Pa.
The cell active electrode area equals to 6 mm2. The relatioship between voltage and current is measured (Four point probe meter) at two conditions namely dark and illuminated with incident light for wavelength of 500 nm and intensity of 16,7 mW/cm2. This solar cell was illuminated on the ITO side using Xe lamp of 50W.
In this research, when thin films of PEG and banana flower extract were coated, the generated electrical current either on the dark or light condition was fixed as dependent variable, while spincoater speed was fixed as independent variable and the room temperature was controlled at 25°C. The electrical conductiviy was measured using four-point probe meter. Aluminium contact pad was metallized at the controlled temperature and thermal pressure namely 961,93°C and 1,33 x 10-4 Pa. The spincoater speed was controlled at 500 rpm and rotated during 30 seconds.
The relationship between pH of banana flower extract with range of absorption wavelength, electrical conductivity and microstructure were accurately investigated and analyzed. The photoactive layer is also analyzed spesifically. The solar cell performance is determined by analyzing the relationship between current and voltage behavior.
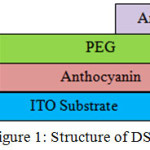 |
Figure 1: Structure of DSSC |
Results and Discussion
The fresh banana flower was sliced, blended and extracted into anthocyanin extract through some steps. The extraction result was filtered and heated while stirring at speed of 500 rpm. The solution was prepared by mixing 30 g aquadest, 150 g ethanol, and 6 g acid acetate during 30 seconds to achieve homogeneous. Then amount of 20 g pieces of banana flower and solution were put in the blender to be crushed. The extracted banana flower were prepared in weight fractions of 0,1, 0, 2, 0,3 and 0,4. The formula used to compute the weight fraction is as the following:
![]()
where X = weght fraction, a = mass of banana flower and b = mass of solvent. The extraction fraction is listed as Table 1.
Table 1: Weight fractions made for preparation of banana flower extraction.
|
Weight fraction (X) |
Banana flower (gram) |
Solution (gram) |
|
|
A |
0,1 |
21 |
186 |
|
B |
0,2 |
47 |
186 |
|
C |
0,3 |
80 |
186 |
|
D |
0,4 |
124 |
186 |
The solution used for distillation process consists of aquadest (30 g), ethanol 96% (150 g) and acid acetate ( 6 g). For sample A, ethanol was separated of solution by distillation process for 2 hours at temperature 95°C and it started to drip in the tenth minute. Sample of weight fraction 0,2 was distilled at temperatures 75-90°C for 145 minutes and vaporization speed 0,86 ml/minute, while the sample of weight fraction of 0,2 was distilled at the same temperature for 150 minutes and vaporization speed 0,53 ml/minute. The solution volume before distillation process equalled to 107 ml and after distillation it became 23 ml and dark purple. Then this solution were prepared at variated pH levels namely 3, 4, 5 and 6 and they were marked A, B, C, and D respectively.
Figure 2 is a SEM image with magnification of 100X of banana flower extract layer at different pH (2, 3, 4 and 5). In Figure 2(a), the bright spots which are visible clumps of sparse and increasingly pH rises, these clots are figure numerous and meetings.
 |
Figure 2: Microstuctures of thin films of banana flower extract for pH levels: (a) 2, (b) 3, (c) 4, and 5 |
On the Figure 3, it is exhibited the relationship between transmittance of banana flower extract layer to ultraviolet exposure for wavelengths of 300-1000 nm at variated pH levels. Based on this relation, it can be taken a relation that as the pH level of solution increases the transmittance increseas as well, and at the pH 6 will fall back.
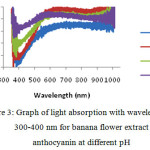 |
Figure 3: Graph of light absorption with wavelengths of 300-400 nm for banana flower extract anthocyanin at different pH |
To characterize thin films of banana flower extracts, the solution of banana flower extract is coated on the preparate glass in size of 1 cm × 1,25 cm using spincoater at speed of 500 rpm for 30 seconds. This substrate size is adapted to electrical property characterization equipment which will be used. The substrates were immersed in the ethanol and shakened gently for 1 hour, cleaned using acetyle acetone, alcohol and aquadest and dried using spincoater to remove water. Then, the coated films were also dried on the oven at temperature 250°C for 10 minutes. The pH effect of solution on the film properties were investigated.
Four samples with each weight fraction of A(0,1), B(0,2), C(0,3) and D(0,4) were characterized using ocean optic Vis-Nir USB4000. This characterization aims to determine absorbances of banana flower extract for wavelengths of visible light up to infrared (350-100nm).
Next step is metallization process to make aluminium contact. Four DSSC samples (A, B, C and D) of solution pH each is 3, 4, 5 and 6 were metallized using evaporator which is installed by heating element (wolfram wires) to form aluminum pad. The electrical properties of fabricated DSSCs were characterized using IV-meter. The used organic materials can function well as a semiconductor material. Nearby at all pH levels, the Knee voltages were found. Additionally, at pH 3, the knee voltage was not found (in the Fig. 4 it is indicated by blue line), at pH 4, the Knee voltage was found about 1 V and shows the semiconductor typical. For pH 5 and pH 6 the Knee Voltages were found in 5,5V and 4,5V, respectively. There is a trend that the more the pH the more the Knee voltage.
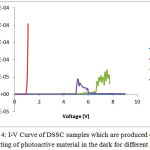 |
Figure 4: I-V Curve of DSSC samples which are produced of first coating of photoactive material in the dark for different pH |
At the low pH (4), the generated maximum current is relatively higher than that of high pH (pH 5 and 6). The voltage increase on the forward bias 0 up to 1 Volt has generated the electrical current sharply for DSSC sample of solution pH 3 (it is indicated by red color line). The anthocyanin gist of sample decreased when pH incrased from pH 3 to pH 7 (Ekici et al., 2014). The generated electrical current at pH 5 and pH 6 were relatively slower than that of pH 3 were caused the anthocyanin content at high pH decreased.
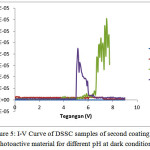 |
Figure 5: I-V Curve of DSSC samples of second coating of photoactive material for different pH at dark condition |
Figure 5 contains a DSSC curve with thicker photoactive material layer than the tested samples as indicated in Figure 4. In Figure 5, the Knee voltages of pH 4 sample is not found, while for samples with pH 5 and pH 6 they were consistently found namely Knee voltages 6.5 V and 4.5 V respectively. The acid treatment leads to absorption spectrum of pigment widening and cell efficiency increasing.
Conclusion
The anthocyanin level in the solution of banana flower extract lessened as well as the pH of solution raised. The pH level increase of photoactive layer of DSSC sample has triggered the Knee voltage enhance and also the produced electrical current reduce. The acid treatment has created absorption pigment spectrum widening and cell efficieny augmenting.
Acknowledgment
We thank the Ministry of Research, Tehcnology, and Higher Education for funding this research through Competitive Grant Scheme under Contract No. 055/SP2H/LT/DRPM/2016. We also thank Mutaqin, S.Pd. (Technician of Physics Laboratory, Semarang State University) for his technical help and Dr. Fachrurrosyid (Researcher in the Physics Laboratory, Sebelas Maret University) for metallization service.
References
- Prima, E. C.; Yuliarto, B.; Suendo, V. Suyatman J. Mater. Sci. Mater. Electron. 2014, 25, 4603-4611
CrossRef - Gokilamani, N.; Muthukumarasamy, N.; Thambidurai, M.; Ranjitha, A.; Velauthapillai, D. J. Sol-Gel Sci. Technol. 2013, 66, 212-219
CrossRef - Isah, K. U.; Ahmad, U.; Idris, A.; Kimpa, M. I.; Uno, U. E.; Ndamitso, M. M.; Alu, N. Mater. Renew. Sustain. Energy 2014, 3(39), 1-5
- Tripathi, M.; Upadhyay, R.; Pandey, A.; Dubey, P. K. Ionics 2013, 19(8), 1179-1183
CrossRef - Gokilamani, N.; Muthukumaramy, N.; Thambidurai, M.; Rajintha, A.; Velauthapillai, D.; Senthil, T.S.; Balasundaraprabhu, R. J. Mater. Sci.: Mater. Electron. 2013, 24, 3394-3402
CrossRef - Szostak, R.; Souza, E.C.F.; Antunes, S.R.M.; Borges, C.P.F.; Andrade, A.V.C.; Rodrigues, P.R.P.; Anyunes, A.C. J. Mater. Sci.: Mater. Electron. 2015, 26(4); 2257-2262
CrossRef - Gokilamani, N.; Muthukumarasamy, N.; Thambirudai, M.; Rajintha, A.; Velauthapillai, D. J. Sol-Gel Sci. Technol. 2014, 69; 17-20
CrossRef - Gokilamani, N.; Muthukumarasamy, N.; Thambidurai, M.; Rajintha, A.; Velauthapillai, D. Appl. Nanosci. 2015, 5, 297-303
CrossRef - Tekerek, S; Kudret, A; Alver, Ü. Indian J. Phys. 2011, 1, 1469 -1476
CrossRef - Sutikno, Dharmaputera; N.M.; Rahayu, S. J. Adv. Agric. Technol. 2014, 1(2), 89 -93
- Ekici, L.; Simsek, Z.; Ozturk, I.; Sagdie, O.; Yetim, H. Food Anal. Methods 2014, 7, 1328-1336
CrossRef - Tripathi, M; Chawla, P. Ionics 2015, 21, 541-546
CrossRef - Sutikno, Hakim, M.L.; Sugianto. Orient. J. Chem. 2016, 32(1), 165-170
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.









