Chemical Composition of The Essential Oil of Carduncellus Helenioides (Desf.)Hanelt from Algeria
F. Meratate1*, A. Lalaoui2, K. Rebbas3, O. K. Belhadad1, N. I. Hammadou4, H. Meratate5, I. Demirtas7, S. Akkal6and H. Laouer 8
1 Department of chemistry, University of M’sila, Algeria.
2 University of Algiers, Algeria.
3 Department of Natural and Life Sciences, University of M’sila, Algeria.
4 Faculty of medicine, University of Algiers, Algeria.
5 Department of pharmacy, Algeria.
6 Department of Chemistry, Çankırı Karatekin University, Çankırı, Türkiye.
7 Department of chemistry, University of Constantine , Algeria.
8 Department of Natural and Life Sciences, University of setif 1, Algeria .
Corresponding Author E-mail : faizachimie@hotmail.fr
DOI : http://dx.doi.org/10.13005/ojc/320304
Article Received on : April 23, 2016
Article Accepted on : May 30, 2016
The essential oil extracted from Carduncellus helenioides was analyzed using GC/EIMS. It was characterized by diepicedrene-1-oxide (10.6%), isoaromadendrene epoxide (7.1 %), caryophyllene oxide (6.20 %) , eudesmol (6.17 %) and aromadendrene oxide (1.3 %) as major constituents. The antibacterial activity of the essential oil of this plant were carrying out by disc diffusion method against four bacterial strains and the oil was only active against Staphylococcus aureus ATCC 25923.
KEYWORDS:Carduncellus helenioides; essential oil; caryophyllene oxide; isoaromadendrene epoxide; antibacterial activity
Download this article as:| Copy the following to cite this article: Meratate F, Lalaoui A, Rebbas K, Belhadad O. K, Hammadou N. I, Meratate H, Demirtas I, Akkal S, Laouer H. Chemical Composition of The Essential Oil of Carduncellus Helenioides (Desf.)Hanelt from Algeria. Orient J Chem 2016;32(3). |
| Copy the following to cite this URL: Meratate F, Lalaoui A, Rebbas K, Belhadad O. K, Hammadou N. I, Meratate H, Demirtas I, Akkal S, Laouer H. Chemical Composition of The Essential Oil of Carduncellus Helenioides (Desf.)Hanelt from Algeria. Orient J Chem 2016;32(3). Available from: http://www.orientjchem.org/?p=18148 |
Introduction
The term of medicinal plants include a various types of plants used in herbalism and some of these plants have a medicinal activities. These medicinal plants consider as a rich resources of ingredients which can be used in drug development and synthesis 1. Essential oils are concentrated liquids of complex mixtures of volatile compounds and can be extracted from several plant organs. Essential oils are a good source of several bioactive compounds, which possess antioxidative and antimicrobial properties 2. Asteraceae (Compositae) is the richest vascular plant family in the world, with 1600 to 1700 genera and 24,000 to 30,000 species. They are easily distinguished by the florets grouped in capitula, and the fruit, a cypsela often with a pappus 3.
The genus Carthamus includes 18 species of which C.tinctorius L. is the only cultivated species 4. Carthamus species probably originate from Southern Asia and is known to have been cultivated in China, India, Iran and Egypt 5.
Carduncellus helenioides (syn.Carthamus helenioides Desf.) is a high powerful plant (30-100 cm). Stem erect, simple or rower, firm and ridged. Yellow flowers. hairless, leathery leaves, ovate-lanceolate with great ribs, whole or scarcely denticulate, except sometimes the latest; stalked the lower and upper amplexicaul. Big heads of 4-5 cm in diameter, inverted and worn by a thickened stem.Involucre extra large, oval-lanceolatefoliaceus and finely serrated bracts. Involucre itself to ciliated bracts, entire last ending in Appendix ciliated and scarious. This is a clay Asteraceae places; Algerian-Moroccan 6.
In this study we sought to characterize the essential oils extracted from C.helenioides, as well as to evaluate their antibacterial activities. As far as we know, there is no report on volatile constituents of C.helenioides and this is the first study on the chemical composition and antibacterial effects of essential oils from this plant.
Material and Methods
Plant material
The Aerial parts (leaves, stems and flowers) of Carduncellus helenioides were collected during their flowering stage from M’sila in May 2015 (Figure 1). The plant was identified by Dr khellaf Rebbas (university of M’sila).
 |
Figure 1: Carduncellus helenioides Click here to View Figure |
Essential oil isolation
Isolation of essential oils was performed using hydrodistillation of aerial parts of Carduncellus helenioides using a Clevenger-type apparatus over 3 hours. The oils were dried over sodium sulphate7.
Qualitative and Quantitative analyses
Gas Chromatography with Electron Impact Mass Spectrometry (GC/EIMS) was performed using a Agilent technologies gas chromatograph equipped with a HP-5ms capillary column (30 m×0.25 mm; coating thickness 0.25 μm) and a 5975Cinert MSD, with Triple-Axis detector (Agilent Technology). The analytical conditions were as follows: injector temperature, 250; oven temperature, programmed from 60°C to 240°C at 4°C/min; carrier gas helium at 1 ml/min; injection of 1 μl (10% hexane solution); and split ratio, 1:20. Constituents were identified retention index (RT) and by matching their recorded mass spectra with the Standard mass spectra from the NIST05, Wiley and flavor2 libraries data provided by the software of the GC–MS system`.
Antimicrobial assay
Microbial strains
The antimicrobial activities of essential oil were tested against four bacteria species, selected as representative of the class of Gram-positive: S. aureus (ATCC 25923), Entecococcus faecalis. Gram-negative: Escherichia coli (ATCC 25922), Pseudomonas aeruginosa (ATCC 27853). The inhibition effect on microbe growth was determined by the disc diffusion method using suspension containing colony of bacteria spread on Muller-Hinton agar (MHA) medium. Sterile 6 mm diameter filter paper discs were impregnated with 10µl of essential oil was deposited at equal distances on the surface of the inoculated agar (MHA for antibacterial assay). Cultures were then incubated at 37 °C for 24h. The diameter of inhibition zones was measured. Standard reference antibiotics (Gentamicin and Amoxicillin) were used as positive control. We used as a negative control disc impregnated with solvent (Diethyl ether) without essential oil.
The inhibition zone was measured using ruler and recorded for statistical analysis8.
Results and Discussion
The yield of volatile oil tended to increase before flowering 0.9 % and decrease once flowering 1,16 % [(w/w) based on the dry weight of the plant] of a yellow and perfumery odor oil. Forty-four constituents were characterized and listed in order of their elution in Table 1 which indicates the percentage and retention time (RT) of each component. The compounds represent 85.57% of the total oil. The major constituents were diepicedrene-1-oxide (10.6%), isoaromadendrene epoxide (7.1%), caryophyllene oxide (6.20 %) and β-eudesmol (6.17%), aromadendrene oxide (1.3 %).
The oil contains low proportion of monoterpenes (0.4 %), oxygenated monoterpenes (0.63 %), sesquiterpene (3.2 %) and high proportion of oxygenated sesquiterpenes (25.97 %)in which diepicedrene-1-oxide (10.6 %) and isoaromadendrene epoxide (7.1 %),caryophyllene oxide (6.20 %),eudesmol (6.17 %),aromadendrene oxide (1.3 %) were the major constituents. Besides there are esters (6.6 %) and fatty acid (7.80%).caryophyllene oxide is the main oxygenated sesquiterpene together with eudesmol and isoaromadendrene epoxide.
Compared to other carduncellus species such as C. tinctorius, the major constituents of the oil of flower were 1-hydroxy-3-propyl-5-(4-methyl-penten)-2-methylbenzene (25.2%), 2,5,5 trimethyl-3-propyl,tetra hydro 1-naphtol (19.8%), benzaldehyde (8.0%), caryophyllene oxide (6.5%) and lauric acid (5.1%) 9. We observed that the chemical compositions of the two oils do not show much similarity, although the plant species belong to the same genus. Of all the identified components, only caryophyllene oxide occurs inappreciable amounts in both species.
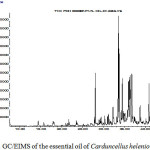 |
Figure 2: GC/EIMS of the essential oil of Carduncellus helenioides Click here to View Figure |
Table 1: Essential oil composition of Carduncellus helenioides.
|
Constituents |
RT |
% |
|
| Monoterpene | |||
| Limonene | 15.725 | 0.4 | |
| Total | 0.4 | ||
| Oxygenated Monoterpenes | |||
| Linalool | 18.023 | 0.4 | |
| 1,6-Dihydrocarveol | 21.458 | 0.2 | |
| Total | 0.6 | ||
| Sesquiterpene | |||
| Caryophyllene | 29.354 | 0.4 | |
| α-Cedrene | 29.205 | 1.0 | |
| β-Cedrene | 29.51 | 0.7 | |
| β-Guaiene | 31.45 | 0.6 | |
| 3α,4β-Dihydroxy-1,5,7α(H),6β(H)-guai-10(15),11(13)-dien-6,12-olide | 38.887 | 0.5 | |
| Total | 3.2 | ||
| Oxygenated Sesquiterpenes | |||
| β-Nootkatol | 28.903 | 0.9 | |
| Caryophyllene oxide | 34.456 | 6.2 | |
| Aromadendrene oxide | 34.69 | 1.3 | |
| Isoaromadendrene epoxide | 34.171 | 7.1 | |
| Alloaromadendrene oxide | 39.439 | 0.2 | |
| β-Eudesmol | 36.33 | 6.17 | |
| Hexahydrofarnesyl acetone | 40.831 | 3.8 | |
| Hexa-hydro-farnesol | 37.931 | 0.3 | |
| Total | 25.97 | ||
| Esters | |||
| Methyl (E,Z)-2,4-decadienoate | 28.02 | 5.4 | |
| 2,4-Decadienoic acid, ethyl ester | 30.262 | 0.7 | |
| 7-Methyl-Z-tetradecen-1-ol acetate | 42.226 | 0.5 | |
| Total | 6.6 | ||
| Fatty acid | |||
| Dodecanoic acid | 33.129 | 1.0 | |
| Tetradecanoic acid | 38.688 | 2.7 | |
| Hexadecanoic acid | 43.802 | 4.1 | |
| Total | 7.8 | ||
| Others | |||
| Epoxycyclooctane | 14.404 | 0.6 | |
| 6-Nonenal | 17.904 | 0.5 | |
| Nonanal | 18.151 | 0.3 | |
| 7′-Oxaspiro[cyclopropane-1,4′-tricyclo[3.3.1.0(6,8)]nonan-2′-one] | 20.973 | 0.2 | |
| Decanal | 21.695 | 0.5 | |
| 5-Cyclodecene, 1,2-epoxy- | 23.458 | 0.6 | |
| Falcarinol | 23.692 | 0.3 | |
| 3-Methyl-2-pent-2-enyl-cyclopent-2-enone | 27.056 | 0.8 | |
| alfa-Copaene | 27.841 | 0.7 | |
| Tetracyclo[6.3.2.0(2,5).0(1,8)]tridecan-9-ol, 4,4-dimethyl- | 35.911 | 6.2 | |
| 2,13-Octadecadien-1-ol | 36.201 | 5.0 | |
| 7R,8R-8-Hydroxy-4-isopropylidene-7-methylbicyclo[5.3.1]undec-1-ene | 37.253 | 0.7 | |
| 2-Methyl-Z,Z-3,13-octadecadienol | 37.392 | 0.7 | |
| 9-Isopropyl-1-methyl-2-methylene-5-oxatricyclo[5.4.0.0(3,8)]undecane | 37.603 | 0.7 | |
| Dihydropseudoionone | 29.866 | 0.1 | |
| β-bisabolol | 30.925 | 0.9 | |
| β-Ionone | 31.14 | 1.2 | |
| Myristicine | 32.237 | 2.0 | |
| 1,7-Octadiene, 2,5-bis-(cis)-(2,2-dimethyl-3-carboxycyclopropyl)- | 41.086 | 1.5 | |
| Ethanol, 2-(9,12-octadecadienyloxy) | 41.437 | 3.3 | |
| Neocurdione | 42.673 | 3.0 | |
| Total | 29.8 | ||
 |
Figure 3: The main constituents of Carduncellus helenioides essential oil Click here to View Figure |
Table 2: Antibacterial activity of Carduncellus helenioides essential oil.
|
|
Inhibition zone (mm)
|
||||
|
Enterococcusfaecalis |
Escherichia coliATCC 25922 |
Staphylococcus aureus ATCC 25923 |
Pseudomonas aeruginosaATCC 27853. |
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| Essential oil |
10µl |
/ |
/ |
11 |
/ |
|
|
|
|
|
|
|
| Gentamicin |
(10µg/disk) |
/ |
28 |
27 |
22 |
|
|
|
|
|
|
|
| Amoxicillin |
(10µg/disk) |
32 |
/ |
/ |
/ |
|
|
|
|
|
|
|
Considering the large number of different groups of chemical compounds present in essential oil, it is most likely that their antibacterial activity is not attributable to one specific mechanism but that there are several targets in the cell10,11. The locations or mechanisms in the bacterial cell thought to be sites of action for EO components are indicated in Figure (4) 12.
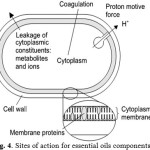 |
Figure 4: Sites of action for essential oils components Click here to View Figure |
The results regarding the antibacterial activity of the essential oil of Carduncellus helenioides are indicated in Table 2. The results obtained from disc diffusion method indicated that Staphylococcus aureus ATCC 25923 was sensitive microorganisms with diameters of inhibition of 11 mm lower than the standard Gentamicin 27mm . On the other hand, The essential oil did not display any antibacterial against Enterococcus faecalis , Escherichia coli ATCC 25922, Pseudomonas aeruginosa ATCC 27853.It is remarkable that the oil exhibited a specific activity against the gram positive bacteria S.aureus .These results are consistent with previous reports in the literature indicating that Gram-positive bacteria are more susceptible to essential oil than Gram-negative bacteria 13, 14.
This antimicrobial activity is suspected to be associated with the high percentage of caryophyllene oxide and the presence of caryophyllene, dodecanoic acid, limonene and linalool. The synergistic effects of the diverse major and minor components of the essential oil should be taken into consideration to account for the oil biological activity 15
Bougatsosa and their collaborators (2004) were tested the antibacterial activity of caryophyllene oxide,β-caryophylleneand linalool against many bacteria and they concluded that the antibacterial activity of the oil may in part be associated with the high percentage of caryophyllene oxide and linalool. Besides, the antibacterial properties of caryophyllene oxide have been reported previously 16, 17, 18.
Conclusion
The present study demonstrate that the most abundant constiutants of Carduncellus helenioides essential oil (EO) from M’sila are diepicedrene-1-oxide (10.6 %) and isoaromadendrene epoxide (7.1 %),caryophyllene oxide (6.20 %), eudesmol (6.17 %),aromadendrene epoxide (1.3 %).Our results demonstrated that EO of Carduncellus helenioides present antibacterial activity against Staphylococcus aureus ATCC 25923 and any antibacterial activity against Entecococcus faecalis, Escherichia coli ATCC 25922 and Pseudomonas aeruginosa ATCC 27853. However, further studies about the safety and toxicity of this oil are needed, in order to evaluate possible clinical application in therapy of infectious diseases.
Acknowledgements
The authors thank Dr. Lakhdar Djarri from Constantine University to extract the essential oil of our plant and we thank pasteur institute (m‘sila) for their kind support.
References
- Bassam Abdul Rasool Hassan , Medicinal Plants (Importance and Uses),Pharmaceut Anal. Acta. 2012.
- Tongnuanchan P and Benjakul S , Essential Oils: Extraction, Bioactivities, and Their Uses for Food Preservation, Journal of Food Science , 2014 ,79, 7.
CrossRef - Funk VA, Bayer RJ, Keeley S, Chan R, Watson L, Gemeinholzer B, Schilling E, PaneroJL, Baldwin BG, Garcia Jacas N, Susanna A and Jansen RK , Everywhere but Antarctica: using a super tree to understand the diversity and distribution of the Compositae. Biol. Skr . 2005 , 55, 343–374.
- Garnatje T, Garcia S, Vilatersana R, Valles J, Genome size variation in the genus Carthamus (AsteraceaeCardueae): systematic implications and additive changes during allopolyploidization. Ann. Bot. 2006, 97, 461– 467.
CrossRef - Turgumbayeva AA, Ustenova GO and Samir A Ross , Volatile Oil Composition of Carthamus Tinctorius L. the Flowers Grown in Kazakhstan, research Journal of Pharmaceutical Biological and Chemical Sciences, 2015.
- Quezel et Santa , La nouvelle Flore de l’Algérie et des régions désertiques méridionales , T1 et 2 , 1962.
- Shahram Amiri , Shahram Sharafzadeh, Essential Oil Components of German chamomile Cultivated in Firoozabad, , Orient J. Chem, 2014, 30 (1) ,365-367.
- Y.C.WONG*, M. Y. AHMAD-MUDZAQQIR and W.A. WAN-NURDIYANA, Extraction of Essential Oil from Cinnamon (Cinnamomum zeylanicum) ,Orient J. Chem, 2014, 30 (1), 37-47.
CrossRef - Ziarati P, Asgarpanah J and Kianifard M, The essential oil composition of Carthamus tinctorius L. flowers growing in Iran , African Journal of Biotechnology , 2012 ,11, 12921-12924.
- Skandamis PN, Koutsoumanis K, Fasseas K and Nychas GJE , Inhibition of oregano essential oil and EDTA on Escherichia coli O157:H7. Italian Journal of Food Science , 2001,13 , 65 – 75.
- Carson CF, Mee BJ and Riley TV , Mechanism of action of Melaleuca alternifolia (tea tree) oil on Staphylococcus aureus determined by time-kill, lysis, leakage and salt tolerance assays and electron microscopy, Antimicrobial Agents and Chemotherapy, 2002 , 46, 1914 –1920.
CrossRef - Burt S Essential oils: their antibacterial properties and potential applications in foods—a review, International Journal of Food Microbiology , 2004 , 94 , 223 – 253.
CrossRef - Outtara B, Simard R E, Holley RA, Piettte GJ and Begin A ,Antimicrobial activity of selected fatty acids and essential oils against six meat spoilage organisms, Int. J. Food Microbiol,1997, 37, 155-162.
CrossRef - Mangena T and Muyima NYO ,Comparative evaluation of the antimicrobial activities of essential oils of Artemisia afra, Pteroniaincana and Rosmarinus officinalis on selected bacteria and yeast strains , Lett. Appl. Microbiol. 1999, 28, 291-296.
CrossRef - Morey A and Canillac N , Food Control , 2002 ,13, 289.
CrossRef - Bougatsosa C, Ngassapab O, Runyorob DKB, and Chinoua IB ,Chemical Composition and in vitro Antimicrobial Activity of the Essential Oils of Two Helichrysum Species from Tanzania, Z. Naturforsch , 2004, 59 , 368 – 372 .
CrossRef - Magiatis P, Skaltsounis AL, Chinou I and Haroutounian SA ,Chemical composition and in vitro antimicrobial activity of the essential oils of three Greek Achillea species, Z. Naturforsch , 2002 , 57, 287-290.
CrossRef - Ulubelen A, Topcu G, Eris C, Sönmez U, Kartal M, Kurucu S and Bozok-Johansson C Terpenoids from Salvia sclarea, Phytochemistry , 1994 , 36 ,971- 974.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.









