N,N-Dimethylbenzimidazolium iodide as a green catalyst for cross-coupling of aromatic aldehydeswith unactivated imines
Viwat Hahnvajana wong*,Wijitra Waengdongbung and Parinya Theramongkol
Department of Chemistry and Center for Innovation in Chemistry, Faculty of Science, KhonKaen University, KhonKaen 40002, Thailand
Corresponding Author Email: viwhah@kku.ac.th
DOI : http://dx.doi.org/10.13005/ojc/320123
Article Received on :
Article Accepted on :
Article Published : 05 Feb 2016
Cross-coupling of aromatic aldehydes with unactivated iminescatalyzed by N,N-dimethylbenzimidazolium iodide in ethanolic sodium hydroxide solution gave α-amino ketonesin satisfactory yields. Benzoin condensation and further oxidation of the resulted aroins also occurred as side reactions.
KEYWORDS:N,N-dimethylbenzimidazolium iodide; green catalyst; cross-coupling; unactivated imines; α-amino ketones
Download this article as:| Copy the following to cite this article: Wong V. H, Waengdongbung W, Theramongkol P. N,N-Dimethylbenzimidazolium iodide as a green catalyst for cross-coupling of aromatic aldehydeswith unactivated imines. Orient J Chem 2016;32(1). |
| Copy the following to cite this URL: Wong V. H, Waengdongbung W, Theramongkol P. N,N-Dimethylbenzimidazolium iodide as a green catalyst for cross-coupling of aromatic aldehydeswith unactivated imines. Orient J Chem 2016;32(1). Available from: http://www.orientjchem.org/?p=13837 |
Introduction
Acyl anion equivalents 3 resulted from treatment of aromatic aldehydes 1 with either benzimidazoliumsalt 2a or thiazolium salt 2b in the presence of base have been successfully utilized in benzoin condensation1-4 (eq.1) and Stetter reaction5-9 (eq.2), as illustrated in Scheme 1.
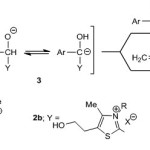 |
Scheme 1 Click here to View scheme |
In addition to aromatic aldehyde 1 and electrophilic alkene4, unactivated imines have also been employed as aroyl anion receptors.10 As shown in Scheme 2, cross-coupling of aromatic aldehydes 1 with unactivated imines 7 catalyzed by thiazolium chloride 2b and trimethylamine in ethanol at 70oC for 2 days provided exclusivelyα-amino ketones 8 in 58-85 %yields.
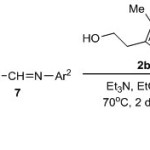 |
Scheme 2 Click here to View scheme |
As part of our continuing interest in catalytic reactivity of N,N-dimethylbenzimidazolium iodide (2a) and its reusability, we report herein the cross-coupling of aromatic aldehydes with imines catalyzed by 2a in ethanolic sodium hydroxide solution.
Experimental
Chemicals were perchased from Fluka, Aldrich and Merck chemical company and used without further purification. Melting points were determined on a Sanyo Gallenkamp melting point apparatus and compared with those of known samples. IR spectra were recorded on a Perkin Elmer Spectrum One FT-IR Spectrometer. 1H and 13C NMR spectra were obtained using a VARIAN MERCURY plus (400 MHz FT NMR).
N,N-Dimethylbenzimidazolium iodide was prepared following our previously reported reference.11
General procedure for the preparation of unactivatedimines
Aniline (9) (9.30 g, 0.10 mol) was added to a vigorously stirred aromatic aldehyde (0.10 mol). Stirring was continued for 15 minutes. The reaction mixture was poured, with rapid stirring, into 95% ethanol (33 ml) and the resulting mixture was allowed to stand, first ten minutes at room temperature and then thirty minutes in an ice bath. The pale yellow precipitates of imine were filtered and washed with cold ethanol. The filtrate was concentrated under reduced pressure and purified by using column chromatography with 20%EtOAc:hexane as eluant to give an additional amount of the imine.
N-Benzylideneaniline(10a).Yellow crystals, mp 51 oC (lit.1250-52 oC), FTIR (KBr, ν, cm-1)nmax 3059, 3026, 1626, 1590, 1483, 1450, 1366, 1192, 1169, 760, 693 cm-1; 1H NMR(CDCl3, 400 MHz) δ 8.45 (1H, s, CH), 7.90 (2H, brd, J = 9.6 Hz, 2- and 6-H), 7.46 (3H, m,3¢-, 4- and 5¢-H), 7.39 (2H, t, J = 8.1 Hz, 3- and 5-H), 7.22 (3H, m, 2¢-, 4¢- and 6¢-H);13C NMR (CDCl3) d 120.9, 125.9, 128.8, 129.1, 131.4, 152.1, 160.4.
N-(4-Methylbenzylidene)aniline(10b). Yellow crystals, mp 44-45 oC (lit.12 44-45 oC), FTIR (KBr, ν, cm-1)nmax 3061, 2884, 1624, 1586, 1483, 1448, 1367, 1191, 1169, 814, 750 cm-1; 1H NMR (CDCl3, 400 MHz)δ 8.41 (1H, s, CH), 7.79 (2H, d, J = 8.1 Hz, 2- and 6-H), 7.38 (2H, t, J = 8.1 Hz, 3¢-, and 5¢-H), 7.27 (2H, d, J = 8.0 Hz, 3- and 5-H), 7.21 (3H, m, 2¢-, 4¢- and 6¢-H), 2.41 (3H, s, ArCH3);13C NMR(CDCl3)d 21.6, 120.9, 125.7, 128.8, 129.1, 129.5, 133.8, 141.8, 152.3, 160.3.
N-(4-Methoxybenzylidene)aniline(10c). Yellow crystals, mp 59-60 oC (lit.13 59-60 oC), FTIR (KBr, ν, cm-1)nmax 3004, 2839, 1604, 1576, 1511, 1485, 1422, 1370, 1308, 251, 1163, 1029, 833 cm-1; 1H NMR (CDCl3, 400 MHz)δ 8.38 (1H, s, CH), 7.85 (2H, d, J = 8.7 Hz, 2- and 6-H), 7.38 (2H, t, J = 8.2 Hz, 3¢-, and 5¢-H), 7.20 (3H, m, 2¢-, 4¢- and 6¢-H), 6.98 (2H, d, J = 8.8 Hz, 3- and 5-H), 3.86 (3H, s, ArOCH3);13C NMR(CDCl3)d 55.4, 114.2, 120.9, 125.5, 129.1, 129.3, 130.5, 152.4, 159.6, 162.3.
N-(4-Chlorobenzylidene)aniline(10d). Yellow crystals, mp 64-65 oC (lit.14 63-64oC), FTIR (KBr, ν, cm-1)nmax 3060, 2983, 2873, 1621, 1584, 1563, 1485, 1401, 1353, 1084, 828, 761, 694 cm-1; 1H NMR(CDCl3, 400 MHz)δ 8.40 (1H, s, CH), 7.83 (2H, d, J = 8.5 Hz, 2- and 6-H), 7.43 (2H, d, J = 8.5 Hz, 3-, and 5-H), 7.39 (2H, t, J = 7.7 Hz, 3¢- and 5¢-H), 7.23 (1H, t, J = 7.4 Hz, 4¢-H), 7.20 (2H, d, J = 7.3 Hz, 2¢- and 6¢-H);13C NMR (CDCl3)d 120.8, 126.1, 129.1, 129.2, 129.9, 134.7, 137.3, 151.7, 158.7.
General procedure for cross-coupling of aromatic aldehydes with unactivatedimines catalyzed by N,N-dimethylbenzimidazoliumiodide(2a) in ethanolicsodium hydroxide solution
An imine (20.00 mmol) and an aromatic aldehyde (24.00 mmol) were added to a solution of N,N-dimethylbenzimidazolium iodide (2a) (1.10 g, 4.00 mmol) and NaOH (0.16 g, 4.00 mmol) in ethanol (20 ml). The reaction mixture was heated at reflux for 5 hours.The pale yellow precipitates of α-amino ketone were filtered and washed with cold ethanol. The filtrate was concentrated under reduced pressure and dissolved in dichloromethane (40 ml). The dichloromethane solution was washed with water (2×20 ml), dried (anh.Na2SO4) and evaporated to dryness. The residue was purified by using preparative thin layer chromatography with 20%EtOAc:hexane as eluantto give an additional amount of the α-amino ketone, aroin, and aril.
N,N-dimethylbenzimidazolium iodide (2a) was recovered from the aqueous layer by evaporation to dryness and recrystallization of the brown residue with ethanol.
1,2-Diphenyl-2-(phenylamino)ethanone (11a). Yellow crystals; mp 99-100 oC (lit.15 98-99 oC); FTIR (KBr, ν, cm-1)nmax 3396, 3054, 3026, 1679, 1599, 1504, 1449, 1319, 1247, 1175, 748, 693 cm-1; 1H NMR (CDCl3, 400 MHz)δ 7.80 (2H, d, J = 7.4 Hz, 2- and 6-H), 7.53 (1H, t, J = 7.5 Hz, 4-H), 7.44 (4H, m, 2¢¢-, 3-, 5- and 6¢¢-H), 7.27 (2H, t, J = 7.7 Hz, 3¢¢- and 5¢¢-H), 7.21 (1H, t, J = 7.3 Hz, 4¢¢-H), 7.13 (2H, t, J = 7.2 Hz, 3¢- and 5¢-H), 6.68 (3H, m, 2¢-,4¢- and 6¢-H), 6.02 (1H, d, J = 6.1 Hz, CH), 5.40 (1H, br d, J = 5.8 Hz, NH);13C NMR (CDCl3)d 62.8, 113.5, 117.9, 128.1, 128.1, 128.7, 128.9, 129.0, 129.2, 133.5, 135.1, 137.8, 146.2, 197.1.
2-Phenyl-2-(phenylamino)-1-p-tolylethanone (11b).Yellow crystals; mp 134-135 oC (lit.16 137oC); FTIR (KBr, ν, cm-1)nmax 3396, 3051, 3028, 1676, 1603, 1504, 1430, 1319, 1249, 1175, 747 cm-1; 1H NMR (CDCl3, 400 MHz)δ 7.91 (2H, d, J = 8.1 Hz, 2- and 6-H), 7.44 (2H, d, J = 7.7 Hz, 2¢¢- and 6¢¢-H), 7.29-7.61 (5H, m, 3-, 3¢¢-, 4¢¢-, 5- and 5¢¢-H), 7.12 (2H, t, J = 7.7 Hz, 3¢- and 5¢-H), 6.67 (3H, m, 2¢-, 4¢- and 6¢-H), 6.00 (1H, d,J= 6.3 Hz, CH), 5.41 (1H, d,J= 5.9 Hz, NH), 2.36 (1H, s, ArCH3);13C NMR (CDCl3) d 21.7, 62.6, 113.5, 113.6, 117.8, 128.0, 128.1, 129.1, 129.3, 129.4, 132.6, 138.1, 144.5, 146.3, 196.6.
1-(4-Methoxyphenyl)-2-phenyl-2-(phenylamino)ethanone (11c). White crystals; mp 149-150 oC (lit.15 143-144 oC);FTIR (KBr, ν, cm-1)nmax 3394, 3052, 1671, 1600, 1505, 1419, 1318, 1258, 1169, 1027, 749 cm-1; 1H NMR (CDCl3, 400 MHz)δ 8.01 (2H, d, J = 8.8 Hz, 2- and 6-H), 7.45 (2H, d, J = 7.6 Hz, 2¢¢- and 6¢¢-H), 7.27 (2H, t,J= 7.4 Hz, 3¢¢- and 5¢¢-H), 7.19 (1H, t, J = 7.3 Hz, 4¢¢-H), 7.12 (2H, t, J = 7.9 Hz, 3¢- and 5¢-H), 6.90 (2H, d, J = 8.8 Hz, 3- and 5-H), 6.67 (3H, m, 2¢-, 4¢- and 6¢-H), 5.98 (1H, s, CH), 5.42 (1H, br s, NH), 3.83 (3H, s, Ar-OCH3);13C NMR(CDCl3) δ55.5, 62.3, 113.5, 113.6, 113.9, 117.3, 127.8, 128.0, 129.0, 129.2, 131.3, 138.3, 146.3, 163.9, 195.4.
1-(4-Chlorophenyl)-2-phenyl-2-(phenylamino)ethanone (11d). Yellow crystals; mp 153-155 oC (lit.15 153-154 oC); FTIR (KBr, ν, cm-1)nmax 3399, 3054, 1683, 1602, 1588, 1504, 1454, 1400, 1317, 1248, 1093, 750 cm-1; 1H NMR(CDCl3, 400 MHz)δ 7.93 (2H, d, J = 8.6 Hz, 2- and 6-H), 7.41 (4H, m, 2¢¢-, 3-, 5- and 6¢¢-H), 7.29 (2H, t, J = 7.2 Hz, 3¢¢- and 5¢¢-H), 7.22 (1H, m, 4¢¢-H), 7.13 (2H, t, J = 7.5 Hz, 3¢- and 5¢-H), 6.68 (3H, m, 2¢-, 4¢- and 6¢-H), 5.96 (1H, d,J = 5.5 Hz, CH), 5.36 (1H, brd,J = 5.0 Hz, NH);13C NMR (CDCl3) δ 62.9, 113.5, 113.6, 118.0, 128.1, 128.3, 129.1, 129.2, 129.3, 130.2, 130.3, 133.4, 137.5, 140.0, 146.0, 195.9.
2-(Phenylamino)-1,2-dip-tolylethanone (11e). Yellow crystals; mp 117-118oC; FTIR (KBr, ν, cm-1)nmax 3396, 3021, 2916, 1674, 1604, 1504, 1430, 1410, 1375, 1316, 1246, 1170, 1000, 747 cm-1; 1H NMR(CDCl3, 400 MHz)δ 7.91 (2H, d, J = 8.1 Hz, 2- and 6-H), 7.33 (2H, d, J = 8.0 Hz, 2¢¢- and 6¢¢-H), 7.22 (2H, t, J = 8.0 Hz, 3- and 5-H), 7.10 (4H, m, 3¢-, 3¢¢-, 5¢- and 5¢¢-H), 6.66 (3H, m, 2¢-, 4¢- and 6¢-H), 5.97 (1H, d,J = 6.5 Hz, CH), 5.38 (1H, brd,J= 6.5 Hz, NH), 2.37-2.24 (6H, s, 2(Ar-CH3));13C NMR (CDCl3) δ 21.0, 21.7, 62.2, 113.4, 113.6, 117.8, 120.9, 128.0, 129.0, 129.2, 129.3, 129.4, 129.8, 132.4, 135.0, 137.8, 144.5, 146.3, 196.7.
1,2-Bis(4-methoxyphenyl)-2-(phenylamino)ethanone(11f).Yellow crystals; mp 108-109oC(lit.17 114-115oC); FTIR (KBr, ν, cm-1)nmax 3395, 2933, 1672, 1600, 1508, 1462, 1420, 1316, 1255, 1170, 1029, 750 cm-1; 1H NMR(CDCl3, 400 MHz)δ 8.00 (2H, d, J = 8.8 Hz, 2- and 6-H), 7.36 (2H, d, J = 8.7 Hz, 2¢¢- and 6¢¢-H), 7.12 (2H, t, J = 8.5 Hz, 3¢- and 5¢-H), 6.90 (2H, d, J = 8.9 Hz, 3- and 5-H), 6.81 (2H, d, J = 8.7 Hz, 3¢¢- and 5¢¢-H), 6.66 (3H, m, 2¢-, 4¢- and 6¢-H), 6.22 (1H, d,J = 6.2 Hz, CH), 5.35 (1H, brd,J = 6.1 Hz, NH), 3.84-3.72 (6H, s, 2(Ar-OCH3));13C NMR (CDCl3) δ 55.1, 55.3, 61.5, 113.4, 113.6, 113.8, 114.0, 114.4, 114.5, 117.5, 127.9, 129.2, 130.1, 131.1, 131.3, 146.3, 159.2, 163.8, 195.5.
1,2-Bis(4-chlorophenyl)-2-(phenylamino)ethanone(11g).Yellow crystals; mp99-100oC; FTIR (KBr, ν, cm-1)nmax 3398, 3052, 1682, 1603, 1589, 1504, 1488, 1400, 1312, 1245, 1174, 1092, 747 cm-1; 1H NMR(CDCl3, 400 MHz)δ 7.90 (2H, d, J = 8.6 Hz, 2- and 6-H), 7.41 (2H, d, J = 8.5, 3- and 5-H), 7.35 (2H, d, J = 8.5, 2¢¢- and 6¢¢-H), 7.25 (2H, d, J = 8.4 Hz, 3¢¢- and 5¢¢-H), 7.13 (2H, t, J = 8.1 Hz, 3¢- and 5¢-H), 6.70 (1H, t, J = 7.4, 4¢-H), 6.63 (2H, d, J = 7.9, 2¢- and 6¢-H), 5.93 (1H, d,J = 6.2 Hz, CH), 5.36 (1H, brd,J = 6.1 Hz, NH);13C NMR (CDCl3) δ 62.1, 113.5, 113.6, 118.3, 129.2, 129.3, 129.4, 130.2, 133.1, 134.2, 136.1, 140.3, 145.7, 195.5.
Benzoin (12a).White crystals; mp 136-137oC (lit.18135oC); FTIR (KBr, ν, cm-1)nmax 3381, 3060, 2932, 1678, 1595, 1449, 1388, 1262, 1204, 1067, 977, 752 cm-1; 1H NMR(CDCl3, 400 MHz) δ 7.91 (2H, d, J = 7.4 Hz, 2- and 6-H), 7.49 (1H, t, J = 7.8 Hz, 4-H), 7.33 (2H, t, J = 7.8 Hz, 3-and 5-H), 7.30-7.25 (5H, m, ArH), 5.95 (1H, d,J= 6.1 Hz, CH); 4.55 (1H, d,J= 6.1 Hz, OH);13C NMR (CDCl3)δ 76.2, 127.8, 128.6, 128.7, 129.2, 129.1, 133.5, 133.9, 139.0, 199.0.
4,4¢-Dimethylbenzoin (12b).White crystals; mp 73-74 oC (lit.3 75-76 oC); FTIR (KBr, ν, cm-1)nmax 3458, 3030, 2921, 1673, 1606, 1511, 1409, 1391, 1277, 1077, 787 cm-1; 1H NMR(CDCl3, 400 MHz)δ 7.82 (2H, d, J = 8.1 Hz, 2- and 6-H), 7.22 (2H, d, J = 7.9 Hz, 3- and 5-H), 7.18 (2H, d, J = 8.1 Hz, 2¢- and 6¢-H), 7.11 (2H, d, J = 8.0 Hz, 3¢-H and 5¢-H), 5.90 (1H, s, CH), 2.35 (3H, s, Ar-CH3), 2.28 (3H, s, Ar-CH3); 13C NMR (CDCl3)δ 21.1, 21.7, 75.8, 127.6, 129.3, 129.4, 129.8, 131.0, 136.4, 138.4, 144.9, 198.6.
Anisoin (12c).White crystals; mp 110-112 oC (lit.18 111oC); FTIR (KBr, ν, cm-1)nmax 3454, 3005, 2936, 2839, 1668, 1598, 1509, 1462, 1307, 1252, 1074, 1027, 828 cm-1; 1H NMR(CDCl3, 400 MHz)δ 7.89 (2H, d, J = 8.9 Hz, 2- and 6-H), 7.24 (2H, d, J = 8.7 Hz, 2¢- and 6¢-H), 6.86 (2H, d, J = 6.4 Hz, 3- and 5-H), 6.84 (2H, d, J = 6.1 Hz, 3¢- and 5¢-H), 5.84 (1H, d,J= 6.0 Hz, CH), 4.56 (1H, d,J = 6.0 Hz, OH), 3.82-3.75 (6H, s, 2(OCH3));13C NMR (CDCl3)δ 55.2, 55.5, 75.2, 113.9, 114.5, 126.3, 129.0, 131.6, 131.9, 159.6, 164.0, 197.3.
4,4¢-Dichlorobenzoin (12d).Yellow crystals; mp 89-90 oC (lit.19 88 oC); FTIR (KBr, ν, cm-1)nmax 3424, 3091, 3072, 687, 1674, 1590, 1488, 1401, 1252, 1093, 979, 812 cm-1; 1H NMR(CDCl3, 400 MHz)δ 7.81 (2H, d, J = 8.6 Hz, 2- and 6-H), 7.36 (2H, d, J = 8.6 Hz, 3- and 5-H), 7.28 (2H, d, J = 8.5 Hz, 3¢- and 5¢-H), 7.24 (2H, d, J = 8.5 Hz, 2¢- and 6¢-H), 5.88 (1H, d, J = 6.0 Hz, CH), 4.51 (1H, d, J = 6.0 Hz, OH);13C NMR (CDCl3)δ 75.5, 129.1, 129.2, 129.4, 130.4, 131.6, 134.8, 137.2, 140.7, 197.5.
Benzil (13a).Yellow crystals; mp 95-96 oC (lit.18 95 oC), FTIR (KBr, ν, cm-1)nmax 3065, 1662, 1594, 1580, 1450, 1211, 1174, 875, 719 cm-1; 1H NMR(CDCl3, 400 MHz)δ 7.98 (4H, d, J = 7.4 Hz, 2-, 2¢-, 6- and 6¢-H), 7.66 (2H, t, J = 7.5 Hz, 4- and 4¢-H), 7.52 (4H, t, J = 7.7 Hz, 3-, 3¢-, 5- and 5¢-H);13C NMR (CDCl3)δ 129.1, 130.0, 133.1, 134.9, 194.6.
4,4¢-Dimethylbenzil (13b).Yellow crystals; mp99-100oC (lit.18 103oC), FTIR (KBr, ν, cm-1)nmax 2922, 1667, 1604, 1572, 1440, 1410, 1379, 1321, 1219, 1173, 745 cm-1; 1H NMR(CDCl3, 400 MHz)δ 7.86 (4H, d, J = 8.2 Hz, 2-, 2¢-, 6- and 6¢-H), 7.04 (4H, d, J = 8.0 Hz, 3-, 3¢-, 5- and 5¢-H), 2.43 (6H, s, 2(Ar-CH3));13C NMR (CDCl3)δ 21.7, 126.5, 129.2, 130.2, 144.6, 171.4.
p-Anisil (13c).White crystals; mp 132-133 oC (lit.18133oC); FTIR (KBr, ν, cm-1)nmax2959, 1655, 1598, 1572, 1509, 1424, 1312, 1263, 1161, 1016, 832 cm-1; 1H NMR(CDCl3, 400 MHz)δ 7.87 (4H, d, J = 8.8 Hz, 2-, 2¢- and 6-, 6¢-H), 6.89 (4H, d, J = 8.9 Hz, 3-, 3¢- and 5-, 5¢-H), 3.80 (6H, s, 2(OCH3)); 13C NMR (CDCl3)δ 55.6, 114.3, 126.3, 132.4, 164.8, 193.6.
4,4¢-Dichlorobenzil (13d). Yellow crystals; mp 197-198 oC (lit.20 197-198 oC), FTIR (KBr, ν, cm-1)nmax 3094, 1661, 1587, 1425, 1316, 1210, 1173, 1094, 881, 835 cm-1; 1H NMR(CDCl3, 400 MHz)δ 7.85 (4H, d, J = 8.6 Hz, 2-, 2¢-, 6- and 6¢-H), 7.43 (4H, d, J = 8.6 Hz, 3-, 3¢-, 5- and 5¢-H);13C NMR (CDCl3)δ129.5, 131.1, 131.3, 141.8, 192.4.
Results and Discussion
Imines 10 used in our study were prepared in very good yields, as shown in Scheme 3, by condensation between the corresponding aromatic aldehydes 1 with aniline (9) in the absence of solvent at room temperature for 15 minutes.21
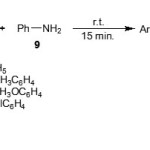 |
Scheme 3 Click here to View scheme |
We had examined cross-coupling between benzaldehyde (1a) and N-benzylideneaniline (10a) under similar conditions as reported in the literature by using 20 mol% of N,N-dimethylbenzimidazolium iodide (2a) and triethylamine in ethanol at 70oC for 2 days.10Unfortunately, this reaction gave only 4% yield of the desired 1,2-diphenyl-2-(phenylamino)ethanone (11a) together with benzoin (12a)and benzil (13a)in 28 and 26% yields, respectively. Benzoin (12a) was resulted from benzoin condensation which under went further oxidation to give benzil (13a)via the mechanism proposed by Miyashita etal.22Replacement of triethylamine with sodium hydroxide, on the other hand, was found to give α-amino ketone 11a along with benzoin (12a) and benzil (13a) in 27, 12 and 61% yields, respectively, as shown in Table 1. Increasing the reaction temperature led to a higher yield of α-amino ketone 11a, i.e.,76% yield.The two side products, 12a and 13a, were still obtained in 9 and 10% yields, respectively. Similar results were obtained despite of the shorten of the reaction time to only 5 hours, i.e.,11a, 12a and 13 a were obtained in 76, 14 and 14% yields, respectively.
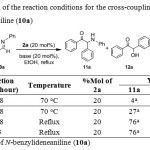 |
Table 1: Optimization of the reaction conditions for the cross-coupling of benzaldehyde (1a) with N-benzylideneaniline (10a) Click here to View table |
Among various conditions studied, cross-coupling of benzaldehyde (1a) with N-benzylideneaniline (10a) using 20 mol% of benzimidazoliumsalt2a as a catalyst in ethanolic sodium hydroxide solution at reflux for 5 hours was chosen as the optimum conditions. Although the conditions at reflux for 48 hours also gave the same yield of 1,2-diphenyl-2-(phenylamino)ethanone (11a), it took longer time and also provided comparable yields of the two side products.
Cross-coupling between a variety of substituted aromatic aldehydes 1 with imines 10 were carried out and the results are shown in Table 2. Aromatic aldehydes having a weak electron-donating group, i.e., 1b, as well as the one with a weak electron-withdrawing group, i.e., 1d, reacted with N-benzylideneaniline (10a), affording good yields of α-amino ketones 11b and 11d, respectively. Reactions between aromatic aldehydes 1b and 1d with imines 10b and 10d, derived from the same aldehydes, also provided good yields of the corresponding α-amino ketones11eand 11g, respectively. On the other hand, crossing-coupling of aromatic aldehydes 1c bearing strong electron-donating group withN-benzylideneaniline (10a) as well as N-(4-methoxybenzylidene)aniline (10c)gave poor yields of α-amino ketones11c and 11f, respectively. Corresponding aroins 12b-d and arils 13b-d were obtained from all cases as side products.
 |
Table 2: Thegenerality and scope of the cross-coupling of aromatic aldehydes with imines Click here to View table |
Replacement of an aromatic aldehyde with a half equivalent of benzoin (12a) under the optimum conditions gave 1,2-diphenyl-2-(phenylamino)ethanone (11a) together with benzoin (12a) and benzil (13a) in 76, 17 and 7% yields, respectively.
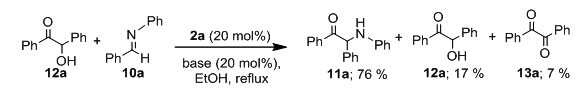
After separation of organic products,N,N-dimethylbenzimidazolium iodide (2a) could be recovered by evaporation of the aqueous solution of the benzimidazoliumsalt to dryness.Recrystallization of the residue in ethanol gave recovered purified N,N-dimethylbenzimidazolium iodide (2a).
Conclusion
N,N-Dimethylbenzimidazolium iodide (2a)in the presence of NaOH satisfactorily catalysed cross-coupling between aromatic aldehydes and unactivated imines to afford expected α-amino ketones. Aromatic aldehydesalso underwent benzoin condensation and some resulted aroins went through oxidation as evidenced by isolation of aroins and corresponding arils as minor products. Expected α-amino ketone could be obtained from treatment of benzoin with an imine. Benzimidazolium salt could be recovered and reused.
Acknowledgement
We appreciatively acknowledge the Center for Innovation in Chemistry (PERCH-CIC) and the Commission of Higher Education (CHE-RG), Ministry of Education for financial support.
References
- Iwamoto, K.;Hamaya, M.; Hashimoto, N.; Kimura, H.; Suzuki, Y.; Sato, M.Tetrahedron Lett.2006,47,7175-7177.
CrossRef - Hahnvajanawong, V.; Waengdongbung, W.; Piekkaew, S.; Phungpis, B.; Theramongkol, P.Scienceasia2013,39, 50-55.
CrossRef - Breslow,R.J. Am. Chem. Soc. 1958,80, 3719-3726.
CrossRef - Breslow, R.; McNelis, E.J. Am. Chem. Soc. 1960,82, 2394-2395.
CrossRef - Hahnvajanawong, V.;Phungpis, B.; Theramongkol, P. ACGC Chem. Res.Comm. 2009,23,26-30.
- Phungpis, B.;Hahnvajanawong, V.; Theramongkol, P. Orient. J. Chem.2014,30, 933-939.
CrossRef - Stetter, H.; Schreckenberg, M.Angew. Chem.1973, 85, 89.
CrossRef - Stetter, H.Angew. Chem.1976, 88, 695-704.
CrossRef - Stetter, H.; Kuhlmann, H. Org. React.1991, 40, 407-496.
- Li, G.-Q.; Dai, L.-X.; You, S.-L. Chem.Commun.2007,852-854.
CrossRef - Hahnvajanawong, V.;Tearavarich, R.;Theramongkol, P.ACGC Chem. Res.Comm. 2005,18, 7-10.
- Cordes, E. H.; Jencks, W.P.J. Am. Chem. Soc.1962,84, 826-831.
CrossRef - Sekiya, M.; Morimoto, T. Chem. Pharm. Bull.1975,23, 2353-2357.
CrossRef - Bennett, J. S.; Charles, K. L.; Miner, M. R.; Heuberger, C. F.; Spina, E. J.; Bartels, M. F.; Foreman, T.Green Chem. 2009,11,166-168.
CrossRef - McEwen, W. E.; Grossi, A. V.; MacDonald, R. J.; Stamegna, A. P.J. Org. Chem.1980,45,1301-1308.
CrossRef - Kozlov, N. S.ZhurnalOrganicheskoiKhimii1965,1, 1641-1642.
- Szmuszkovicz J.;Glenn,E. M.;Heinzelman,R. V.; Hester,J. B.; Youngdale, G. A.J. Med. Chem.1966,9,527-536.
CrossRef - Pouchert, C. J.; Behnke, J.The Aldrich Library of NMR Spectra, Vol 2. Milwaukee, Wisconsin. 1991.
- Buckingham, J. Dictionary of Organic Compounds 5thedition, Chapman and Hall, New York, 1982.
- Firouzabadi, H.; Mottghinejad, E.; Seddighi, M. Synthesis1989,5, 378-380.
CrossRef - Lucius, A.; Bigelow.;Eatough, H. Org. Syn. 1941,1, 80.
- Miyashita, A.; Matsuda, H.;Iijima, C.;Higashino, T.Chem. Pharm. Bull.1990, 38,1147-1152.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.









