Preparation and thermal and physical properties of nano-silica modified and unmodified
Naser Gharehbash*,1 and Alireza Shakeri2
1Young Researchers and Elite Club, Gorgan Branch, Islamic Azad University, Gorgan, Iran 2Faculty of Chemistry, University College of Science, University of Tehran, Tehran, Iran Corresponding Author : E-mail: d.gha.5na@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/31.Special-Issue1.25
Article Received on :
Article Accepted on :
Article Published : 05 Sep 2015
In this study, the surface of silica nanoparticles was modified by three Silanol groups: VTMES (vinyl tries; 2-metoxy; ethoxy Silane), GAPTMS( δ-amino propyl trim ethyl Silone), and PTMS( propyl trim ethyl Silone). The purpose of this study is that the surface of the silica nanoparticle hydrophilic with organosilanol compounds derived from silica has been modified to be adapted with hydrophobic polyolefin and improved the properties of new nano-composite materials. Silica nanoparticles were modified at three levels 1, 3, and 5% by weight. To study the morphology of silica nanoparticles modified the techniques of electron microscopy (TEM) , (SEM) and thermal analysis (TGA), infrared spectroscopy (FT-IR) was used. The results showed that the subjects with higher surfaces of hydrophobic had been modified further and further reduced water absorption capacity. Silica nanoparticles enhanced the original structure, binding to polymers such as polyolefin and polypropylene is ready to prepare the nanocomposites.
KEYWORDS:Nanosilica; Silanole groups; modified; hydrophilic surface
Download this article as:| Copy the following to cite this article: Gharehbash N, Shakeri A. Preparation and thermal and physical properties of nano-silica modified and unmodified. Orient J Chem 2015;31(Special Issue1). |
| Copy the following to cite this URL: Gharehbash N, Shakeri A. Preparation and thermal and physical properties of nano-silica modified and unmodified. Orient J Chem 2015;31(Special Issue1). Available from: http://www.orientjchem.org/?p=10589 |
Introduction
Today, the polymer nano-composites have occupied the minds of most scientists. The unique properties of this material have been allocated to the attention of most craftsmen[1]. Polymer nano-composites are prepared by dispersion of nanoparticles in a polymer Natrys. The more the compatibility between the nanoparticles and the polymer is, the better the properties of nano-composites can be received[2]. Polymer nano-composites are a class of materials in which mineral nanoparticles with the plate and spherical structure have been dispersed in the polymer substrate[3-8]. Silica particles with very small particle size and high surface area have a wide use in polymer industry and surface coating. The level of these particles includes three hydroxyl groups of chemicals, hydrogen and hydroxyl groups attached to the siloxane groups. Therefore, the particles are hydrophilic, although the siloxane groups are hydrophobic alone, there may be possible the hydrophobic of silica particles using a hydrophobic material such as hexamethyldisilane[9]. Much research has been done on the use of silica particles of different polymers[10-13]. The researchers found that silica particles could improve the strength, hardness, modulus, creep resistance and adhesion within the framework of thermoplastic polyolefin and elastomer due to the surface properties of nanoparticles[14-17]. Interaction between hydrophilic silica nanoparticles mainly faces the challenge in the hydrophobic polymer level to prepare nano-composites, it means that the modified silica nanoparticles are more likely remain in the hydrophobic polymer substrate compared to the unmodified polymer. Accordingly, in view of the importance of this issue, in this paper we have tried to improve the nano-silica with three organosilanol derived from silica acid and techniques necessary to identify and demonstrate the groups bonded to the surface of silica nanoparticles. The purpose of this study is to improve the polar surface of silica nanoparticles with silica acid derivatives and create a hydrophobic coating on the surface of the nanoparticles obtained to be compatible with non-polar polymers such as polypropylene nano-composites prepared and improve these physical and mechanical properties. Materials: silica nanoparticles synthesized by the researcher of this silica project.
Materials and Methods
Material
silica gel(112220K) produced by Merk Company, calcium chloride and toluene made by Aldrich Company, silicic acid derivatives PTMS (propyltrimethoxysilane , C6H16O3S) boiling point 143°c, VTMES (viniltris (2- methoxyethoxy) silane ,C11H24O6Si) boiling point 133°c and GAPTMS (δ- aminopropyltrimethoxysilane ,C 6 H 17O3N)- boiling point 103°c, company product “Fulko”.
Equipment and Devices
vacuum, ultrasonic devices made by Philips, transmission electron microscopy (TEM) manufactured by Zeiss, Germany, the X-ray (XRD) manufactured by Philips, apparatus (TGA) manufactured by Perkin Elmer, spectrometer (FT-IR) Perkin Elmer Company.
Methods
three sets of nano-silica nanoparticles were prepared in the weight of 1, 3, and 5%. 20 g of silica nanoparticles was mixed with 200 ml of each separate silica acid derivatives and then 200 ml of dry toluene was added to the mixture and with a magnetic stirrer was mixed for 1 hour. Then the products formed were centrifuged for 10 minutes. These products were washed three times with dry toluene and the sediment produced of them was dried in 180°c for 24 hours in a vacuum oven. The products obtained from silica nanoparticles have been modified.
Result and Discussion
To check unmodified nanosilica and modified nanosilica was used by transmission electron microscopy (TEM) and scaning electron microscopy(SEM) . Figure 1-a and 2-a shows TEM and of the silica nanoparticles. It can be seen that nanoparticles are uniform, having a spherical form.Figure 1-b and 2-b shows a Silanol group is well placed on the surface of silica nanoparticles.
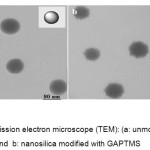 |
Figure 1: Transmission electron microscope (TEM): (a: unmodified nanosilica and b: nanosilica modified with GAPTMS Click here to View figure |
 |
Figure2: Scaning electron microscope (SEM): (a: unmodified nanosilica and b: nanosilica modified with GAPTMS Click here to View figure |
To determine the geometry and size of modified and unmodified crystalline nanosilica was used of the radiation technique X (XRD). In figure3, silica nanoparticles diffraction pattern has been shown.
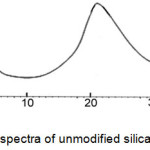 |
Figure 3: XRD spectra of unmodified silica nanoparticles |
OH groups available in nano silica are polar;on the other hands;since most polymer composites are non-polar groups;in order to adjast nano silica onto the polymers and composites; first of all the nano silica surface was modified with silanolic groups (pairing up).then the modified nano silica which has nearly taken a non-polar from can be adjusted with non-polar polymer composites.To identify the binds of OH groups of the surface of nano silica inclouding PTMS ;VTMES and GAPTMS ;(FT-IR)spectroscopy was used. Checking out the (FT-IR) spectroscopy of nano silica sample in(fig.4) shows that out of 4000 cm-1;3000 is related to hydroxyl groups .it shows the bending vibration of (Si-O-Si)at cm-1;470;the stretching vibration of (Si-O-Si)and (Si-OH)with sharp and wide peaks in 800 and 1100 cm-1.studying the spectrum of nano silica modified with derivation of silica acid shows the following vibretching C-H(1390;3460 cm-1);N-H(1650 cm-1)C=C;(3710;1720 cm-1).by analizing the spectroscopy obtained we can make sure that aminosilanole groups are bound to unsaturated vinil group and silica acid derivations.
 |
Figure 4: FT-IR spectrum of the unmodified (1) and modified nanosilica with(2) VTMES, (3) GAPTMS . Click here to View figure |
All the groups selected for modification of nano silica surface have the following two specific characteristics:
- They have a(Si-O)covalent group in their hybrid structure which shows more readiness to form Si-O-Si hybrids.
- The other side of silanolic groups selected is almost polar and these groups while paring up on Nano silica ; produce a matter which is consistent with polymer compounds (usually non-polar)and composites.the mechanism of the modifying linkage can be seen in fig 5.
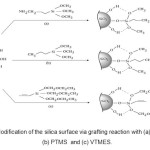 |
Figure 5: Modification of the silica surface via grafting reaction with (a) GAPTMS , (b) PTMS and (c) VTMES. Click here to View figure |
Fig.6:shows the results of differential therogravimetric analysis (TGA)of nano silica before and after the linkage .theinitial weight loss between 100 and 150 ºc is related to the loss of the absorbed moisture by nano silica particles;which is %5 for the initial nano silica ;however it is 2-3 percent for bound nano silica.since these particles are Nano – sized;they have a large contact area;so they easily absorb the moisture of their environment .as it is seen in (fig.6);the bound nano silica tends to absorb less moisture compared to unbound nano silica;which may indicate the fact that nano silica surface hydroxyle are bound by silanole groups (acid silicic derivations).thus;they have less tendency to absorb moisture.another weight loss can be seen at 310-550 °c which is related to the decomposition and destruction of the organic materials bound onto nanosilica particles. There can be seen little difference in decomposition start point due to the changes in the structure of linked (hybrid)matters.above 520 to 600 °c ;weight loss is not seen ;in other words ; above 520 ºc the compound would lose its whole organic sample and only the mineral matter is left.
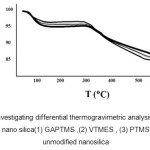 |
Figure 6: Investigating differential thermogravimetric analysis (TGA) of modified nano silica(1) GAPTMS ,(2) VTMES , (3) PTMS) and (4) unmodified nanosilica Click here to View figure |
Conclusion
Nanosilica surface was modified by three pairs of silanol (GAPTMS , PTMS and (VTMES) derived from silica acid. Studies (TEM),(SEM),(XRD),(FT-IR), and (TGA) confirm silanol compounds binding to the surface of silica nanoparticles and hydrophilic silica nanoparticles surface was modified to find a necessary compatibility with hydrophobic polymers such as polyolefin and provide nano-composite with better mechanical and physical properties.
References
- S. Pavlidou, and C.D. Papaspyrides, A Review on Polymer–layered Silicate anocomposites, Prog. Polym.Sci., 33, 1119–1198:(2008).
- Y. Choi, S. Lee, and S. Ryu, Effect of Silane Functionalization of Montmorillonite on Epoxy/Montmorillonite Nanocomposite, Polym. Bull., 63, 47–55( 2009).
- K. Soon, E.H. Jones, R.S. Rajeev, G. Menary, P.J. Martin, and C.G. Armstrong, Morphology, Barrier, and Mechanical Propertiesof Biaxially Deformed Poly(ethylene terephthalate)-MicaNanocomposites, Polym. Eng. Sci., 52, 532–54: (2012).
- A. Leszczy´nska, J. Njuguna, K. Pielichowski, and J.R. Banerjee, Polymer/Montmorillonite Nanocomposites with Improved Thermal Properties: Part I. Factors Influencing Thermal Stabilityand Mechanisms of Thermal Stability Improvement, Thermochim. Acta, 453, 75–96: (2007).
- A. Leszczy´nska, J. Njuguna, K. Pielichowski, and J.R. Banerjee, Polymer/Montmorillonite Nanocomposites with Improved Thermal Properties: Part II. Thermal Stability of Montmorillonite Nanocomposites based on Different Polymeric Matrixes,Thermochim. Acta, 454, 1–22, 2007.
- S.S. Pesetskii, S.P. Bogdanovich, and N.K. Myshkin, TribologicalBehavior of Nanocomposites Produced by the Dispersion of Nanofillers in Polymer Melts, J. Fric. Wear, 28, 457–475: (2007).
- G. Bhat, R.R. Hegde, M.G. Kamath, and B. Deshpande, NanoclayReinforced Fibers and Nonwovens, J. Eng. Fiber Fabric,3, 22-34: (2008).
- J. Njuguna, K. Pielichowski, and S. Desai, Nanofiller-Reinforced Polymer Nanocomposites, Polym. Adv. Technol., 19,947-959: (2008).
- M. Xanthos, Functional Fillers for Plastics, Wiley, Weinheim,(2005).
- S.C. Chung, W.G. Hahm, and S.S. Im, Poly(ethylene terephthalate)(PET) Nanocomposites Filled with Fumed Silicas by Melt Compounding, Macromol. Res., 10, 221-229: (2002).
- X. Zhang, X. Tian, J. Zheng, X. Yao, W. Liu, P. Cui, and Li Y.,Relationship between Microstructure and Tensile Properties of PET/Silica Nanocomposite Fibers, J. Macromol. Sci. Phys., 47,368–377: (2008).
- M.Q. Zhang, M.Z. Rong, H.B. Zhang, and K.F. Rich, Mechanical Properties of Low Nano-Silica Filled High Density Polyethylene Composites, Polym. Eng. Sci., 43, 490-500: (2003).
- O. Aso, J.I. Eguiaza´bal, and J. Naza´bal, The Influence of Surface Modification on the Structure and Properties of a Nanosilica Filled Thermoplastic Elastomer, Compos. Sci. Technol.,67, 2854–2863: (2007).
- L. Zhang, K.C. Tam, L.H. Gan, C.Y. Yue , Y.C. Lam, and X. Hu, Effect of Nano-Silica Filler on the Rheological and MorphologicalProperties of Polypropylene/Liquid-Crystalline Polymer Blends, J. Appl. Polym.Sci., 87, 1484–1492: (2003).
- M.Z. Rong, M.Q. Zhang, S.L. Pan, B. Lehmann, and K. Friedrich, Analysis of the Interfacial Interactions in Polypropylene/Silica Nanocomposites, Polym.Int., 53, 176–183: (2004).
- M. Parvinzadeh, S. Moradian, A. Rashidi, and M.E. Yazdanshenas, Surface Characterization of Polyethylene Terephthalate/Silica Nanocomposites, Appl. Surf. Sci., 256, 2792-2802: (2010).
- A. Guyard, J. Persello, J.P. Boisvert, and B. Cabane, Relationshipbetween thePolymer/Silica Interaction and Properties ofSilica Composite Materials, J. Polym. Sci., Polym.Phys., 44,1134–1146: (2006).

This work is licensed under a Creative Commons Attribution 4.0 International License.









