Synthesis and Characterization of Barium-Vanadium Oxide Nanocomposite Using a Facile thermolysis Approach
Niloufar Akbarzadeh-T , Leila Amiri-O
Department of Chemistry, University of Sistan and Baluchestan, P.O. Box 98135-674, Zahedan, Iran Corresponding Author Email: n.akbarzadeh@chem.usb.ac.ir
DOI : http://dx.doi.org/10.13005/ojc/310450
Article Received on :
Article Accepted on :
Article Published : 03 Dec 2015
In this investigation, we report synthesis of new cationic-anionic Barium-Vanadium complex with 2, 6- pyridinedicarboxylic acid ligand. This compound [Ba(H2O)8][VO2(dipic)]2(1) has been characterized using elemental analyses, FT-IR, UV–Vis and Cyclic voltammetry (CV) techniques. Also in this study we report facile synthesis of Barium-Vanadium Oxide nanoparticle under thermal decomposition, using precursor complex (1). Characterization of Barium-Vanadium Oxide nanocomposites was carried out by scanning electron microscopy (SEM), energy dispersive X-ray analysis (EDX), X-ray powder diffraction (XRD) and Fourier transform infrared (FT-IR) spectroscopy. The X-ray diffraction pattern at room temperature revealed that, highly pure and crystallized Ba3(VO4)2 with rhombohedral structure.
KEYWORDS:cationic-anionic; elemental analyses; Cyclic voltammetry; scanning electron microscopy
Download this article as:| Copy the following to cite this article: Akbarzadeh-T. N, Amiri-O. L. Synthesis and Characterization of Barium-Vanadium Oxide Nanocomposite Using a Facile thermolysis Approach. Orient J Chem 2015;31(4). |
| Copy the following to cite this URL: Akbarzadeh-T. N, Amiri-O. L. Synthesis and Characterization of Barium-Vanadium Oxide Nanocomposite Using a Facile thermolysis Approach. Orient J Chem 2015;31(4). Available from: http://www.orientjchem.org/?p=12868 |
Introduction
Oxide structures with nanometric dimensions exhibit novel physical and chemical properties, with respect to bulk oxide materials, due to the spatial confinement and the proximity of the substrate. Many them have been described during the past few years1,2 . Like most other transition metal oxides, Vanadium, can appear in different states like: V2O5, VO2, V2O3 and V2O. Nanostructured vanadium oxides have been extensively studied 3,4.
various nano compounds that capacities as catalyst with highly regarded in the field of electronics and applications 5.
They exhibit a great variety of nanostructures, ranging from 1D to 3D6,7 also V2O5 has even been chosen as a model system for the description of nanostructured materials8. Vanadium oxides find major applications in the field of lithium ion batteries9,10 . Nanostructured vanadium oxides exhibit unusual morphologies such as nanowires, nanobelts, nanorods, nanotubes and even flower-like or nano-urchin shapes11-13.
In this study we describe preparation of the Barium-Vanadium oxide nanoparticle via synthesis of inorganic precursor metal complex. The complex were characterized by use of physicochemical methods, including spectroscopic (IR, UV–visible) studies. Cyclic voltammetry (CV) method was used to investigate the electrochemical behavior of the complex. The produced nanoparticle characterized using FT-IR, XRD, SEM and EDX. The results are presented and discussed.
Experimental
Materials and Physical Measurements
The raw materials used in this work were of purities above 99%, therefore no further purification was required. All solutions were prepared with double distilled deionized water. Elemental analyses (CHN) were determined on a Heraeus rapid analyzer. Fourier transform infrared (FT-IR) spectra were recorded using JASCO 460 FT-IR spectrophotometer in a KBr matrix. UV–Vis spectra were recorded on a JASCO 7850 spectrophotometer. The redox properties of the complexes were studied by cyclic voltammetric method. Cyclic voltammogram was recorded by using a SAMA 500 Electro Analyzer. X-ray powder diffraction (XRD) measurements were performed using an X’pert diffractometer of Philips Company with graphite monochromatic Cu Kα radiation at room temperature in the 2q range of 10-60°. The sample characterized by using scanning electron microscopy (SEM) and energy dispersive X-ray (EDX) techniques (Philips XL30) with gold coating.
Preparation of [Ba(H2O)8] [VO2(dipic)]2 (1)
For the synthesis of this complex, the cationic and anionic parts were prepared separately then added together and the final product was obtained.
To Prepare cationic part: To a salt of Barium Chloride (0.122 g, 0.5 mmol) was added double distilled deionized water (10 mL) and the mixture was stirred for 10 hour at room temperature to give a clear solution.
To prepare anionic part: To a solution of NH4VO3 (0.117 g, 1 mmol) in double distilled deionized water (15 mL) was added a aqueous solution (10 mL) of (NH4)2(dipic) (0.202 g, 1 mmol) and the mixture was refluxed at 100°C for 24 hour to give a clear yellow solution.
Then a solution of [Ba(H2O)8] 2+ was added to [VO2(dipic)] and the mixture was refluxed for 5 day. White crystals of 1 were obtained by slow evaporation of solvents and kept for further characterization. Yield 69%. Anal. Calc. for C14H22BaN2O20V2: C, 21.63; H, 2.85; N, 3.60;. Found: C, 21.58; H, 2.82; N, 3.51%. FT-IR (KBr, cm-1) νmax: 2600-3000 ν(O-H), 1331, 1701 ν (-COO–), 1530–1300 ν (C=C) and ν (C=N), 956 ν (V=O), 478 ν(V-O), 460 ν(V-N), 517 ν(Ba-O)14-18 (Schem. 1) .
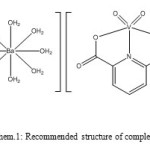 |
Schem 1: Recommended structure of complex (1) Click here to View scheme |
Electronic excitation study
The UV–visible spectra of the title complex, acquired in aqueous solution. A moderately intense band observed at 215 nm that corresponding to the charge transfer (LMCT) from oxygen atom to Vanadium ion (O → V)19.
The bands in the UV region at approximately 268 nm assigned to intra-ligand transitions (n→π* and π→π*) 20,21. V+5 complexes have d0 arrangement therefore no d-d transitions are expected. Fig.1
![Fig. 1: UV–visible absorption spectrum of [Ba(H2O)8] [VO2(dipic)]2](http://www.orientjchem.org/wp-content/uploads/2015/12/Vol31_No4_Nab_Dav_Fig1-150x150.jpg) |
Figure 1: UV–visible absorption spectrum of [Ba(H2O)8] [VO2(dipic)]2 |
Electrochemical Studies
The cyclic voltammogram of complex(1) was obtained at 25ºC under an argon atmosphere using DMF solution containing 0.1 M TBAH as supporting electrolyte and complexes concentration of 4×10-3 M at a glassy carbon working electrode. Ferrocene (Fc) was used as the internal standard, and all redox potentials are referenced to the Fc+/0 couple.
Complex (1) shows two quasi reversible reduction waves at 0.2 V and -0.9V versus the Fc/Fc+ couple attributed to reduction of V+5/ V+4 and V+4/ V+3 respectively Fig. 2. Irreversible reduction waves at negative potential -1.6 V assigned to reduction of ligand.22,23
![Fig.2: Cyclic voltammogram of [Ba(H2O)8] [VO2(dipic)]2 (1)](http://www.orientjchem.org/wp-content/uploads/2015/12/Vol31_No4_Nab_Dav_Fig2-150x150.jpg) |
Figure 2: Cyclic voltammogram of [Ba(H2O)8] [VO2(dipic)]2 (1) Click here to View figure |
Preparation of Barium-Vanadium oxide nanoparticle Ba3(VO4)2
Barium-Vanadium oxide nanoparticle was prepared from title compound (1) using the physical method. First 1.0 g of compound (1) dissolved in oleic acid solution (5mL) as a surfactant. Then calcinated at 1100˚C for 4 h in the furnace to formed Ba3(VO4)2 nanoparticle by thermal decomposition. The final product was collected and washed with distilled water and absolute ethanol several times, dried in air and kept for further characterization.
Results and Discussion
The FT-IR spectrum precursor complex (1) is shown in Fig. 3. The broad absorption band at 2600-3000 in (1) is related to stretching vibrations of O-H group. Absorption bands at 1330 cm-1 and 1700 cm-1 are corresponding to stretching vibration of carboxylate group . Stretching band of V=O group is observed at 956 cm-1. Absorption bands are observed at 478, 460and 517 cm -1 in (1) are corresponding to V-O, V-N and Ba-O group respectively. As shown in Fig. 4 the intensity of these bands decreases when compound (1) is heated at 1100°C. At this temperature, there is broad band at 2500-3500 cm-1 is representative of adsorbed water on the external surface of the sample. Absorption band at 801 related to Barium-Vanadium oxide nanoparticle and providing clear evidence for the presence of the crystalline Ba3(VO4)224
 |
Figure 3: FT-IR spectrum of complex (1) before thermal decomposition Click here to View figure |
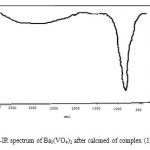 |
Figure 4: FT-IR spectrum of Ba3(VO4)2 after calcined of complex (1) in 1100 ˚C Click here to View figure |
Fig. 5 show XRD pattern for Ba3(VO4)2. It is obvious that all the diffraction peaks can be indexed to rhombohedral phase with space group R¯3n in compared with the standard XRD pattern of Ba3(VO4)2 (JCPDS card No.00-025-1192) diffraction pattern.
No peaks of impurities were detected, indicating the high purity of title product. In addition, the intense and sharp diffraction peaks suggest the obtained product is well crystallized. The broadness of the peaks indicates the small dimensions of the nanoparticle. The crystalline size of the title nanoparticle, was calculated using the Debye-Scherre formula from the major diffraction peak of the corresponding particle25. Upon the main peak, the average size of the particle calculated 2.36 nm. Maximum diffraction is in 2θ=32.06 and (015) as miller indice and The distance between crystalline planes is d=3.24Ȧ26.
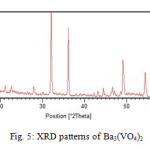 |
Figure 5: XRD pattern of Ba3(VO4)2 Click here to View figure |
The surface morphology, structure and size of the nanoparticle were carried out using SEM image. The SEM image of title nanoparticle is clear. So provided in the form of nanoparticle and almost the morphology and uniformity in the image of them, relatively spherical and porous and beneath the high porosity for use as a catalyst for much is suitable. Fig.6 show the SEM image of the prepared Ba3(VO4)2 nanoparticle.
 |
Figure 6: SEM image of Ba3(VO4)2 Click here to View figure |
To further confirm the chemical composition, EDAX spectrum was recorded. The very strong peaks related to V, Ba and O are found in the two spectrums (Fig. 7) and no impurity was detected. So this method was also verified the formation of title nanoparticle by measuring of ingredient atoms which can be observed in percent of them. Table1.
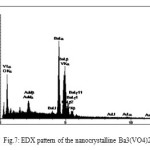 |
Figure 7: EDX pattern of the nanocrystalline Ba3(VO4)2 |
Table 1: EDX data of Ba3(VO4)2
| El | W% | A% |
| O | 18.52 | 61.88 |
| V | 11.42 | 11.98 |
| Ba | 60.52 | 23.53 |
| Au | 9.54 | 2.59 |
Conclusion
In summary, we have successfully synthesized Ba3(VO4)2 nanoparticle with the new Barium-Vanadium complex by thermal decomposition method. The size of the Ba3(VO4)2 nanoparticle was measured both by XRD and SEM and the results were in very good agreement with each other. XRD reveals the phase pure formation of nanoparticle, which is further confirmed by FT-IR. The Barium-Vanadium complex was a suitable precursor for preparation of Barium-Vanadium oxide nanoparticle with interesting morphologies. All results reveal that thermal decomposition method can be employed successfully as a simple, efficient, low cost, environmentally friendly and very promising method for the synthesis of nanoscale materials without a need for special conditions, such as long reaction times, and high pressure. This method may be extending to synthesize other metal nanoparticles oxide, nanowires, nanodisks, and even nanotubes.
References
- Lee, K.; Wang, Y.; Cao, G.; J. Phys. Chem. 2005, 109, 16700-16704
- Wang, Y.; Takahashi, K.; Lee, K.; Cao, G.; J. Adv. Funct. Mater. 2006, 16, 1133-1144
- Tracey, A., , Willsky, G. R. Takeuchi, E. R.; Vanadium Chemistry Biochemistry Pharmacology and Practical applications, 2007, p. 217, CRC press, U.S.A.,.
- Mamedov, E.; Corberán, V. C.; J. Appl. Catal. 1995. A. 127. (1–2)
- ÇAKIR, S.; BİÇER, E.; J. Chil. Chem. Soc. 2010, 55, 236-239,
- Weckhuysen, B.M.; Keller, D.E.; J. Catal. Today 2003, 78 (1–4)
- Gambardella P et al. Nature. 2001, 416, 301.
- Schoiswohl, J.; Sock, M.; Surnev, S.; Ramsey, M. G.; J. N Surf. Sci. 2004, 555 101.
- Taracson , J.M.; Armand, M.; nature, 2001, 414, 359.
- Poizot, P.; Laruelles, S.; Grugean, L,; DuPpnt, J,M,Tarason, Powers Souces. 2001, 97-98-235.
- Larcher, D.; Masquelier, C.; Bonnin, D.; Chabre, Y.; Masson, V.; Leriche, J.B,; Tarascon, J.M., J. Electrochem,Soc. 2003. 150. A133.
- Wang, Y.; Cao, G,; J. Adv. Mater. 2008, 20, 2251-2269.
- Spahr, M.E.; Bitterli, P.; Nesper, R.; Muller, M.; Krumeich, F.; Nissen, H.U. Angew. Chem. Int. Ed. 1998, 37, 1263-1265.
- Nesper, R.; Muhr, H.J.; Nanotubes—an outstanding set of nano particles. Chimia. 1998, 52, 571-578.
- Angus, I.; Rudiger, K. R.; Meyer, J.; Sloan, J.; J. Microscopy and Microanalysis. 2005, 11, 401-409.
- ÇAKIR, S.; BİÇER, E.; J. Chil. Chem. Soc, 2010, 55, 236-239,.
- Dobley, A. K.; Ngala, S.; Yang, P.; Zavalij, Y.; Stanley, M.; J. Chem. Mater. 2001, 13, 4382- 4386.
- K . Nakamoto, Infrared and Raman Spectra of Inorganic and Coordination Compounds. PartB: Applications in Coordination, Organometallic, and Bioinorganic Chemistry, fifth ed., John Wiley & Sons Inc, New York, 1997.
- Gao, H.; Li, Q.; Dai, Y., Luo, F.; Zhang, H.;, J. Corrosion Science, 2010- 52 , 1603–1609.
- Sung, C.; J. Ceramic Processing Research, 2012, 13, 432- 436.
- Swiderski, G.; Kalinowska, M; Wojtulewski, S.; Lewandowski. W.; J. Polyhedron. 2006, 64, 24–3
- Uc¸ I. Bulut, ar,; J. Struct. Chem. 2009, 20, 825–838.
- Sayyah, S.; El-Deeb, M.; Kamal, S.; Azooz, R.; J. Appl. Poly. Sci. 2009, 112, 3695–3706
- Ghosh, T.; Bhattacharya, S.; Das, A.; Mukherjee, G.; Drew, M.; J. Inorganica Chimica Acta. ,2005. 358, 989–996,
- Klug, H. P.; Alexander, L. E.; X-ray Diffraction Procedures for Polycrystalline and Amorphous Materials, Wiley, New York, 1974.
- Parimalagandhi, K.; Vairam, S.; Orient. J. Chem., 2014, 30(4), 1957-1963.

This work is licensed under a Creative Commons Attribution 4.0 International License.









