Electrochemical activity of glutathione at a graphite electrode modified with gold nanoparticles
D.O. Perevezentseva1, K. V. Skirdin1, E. V. Gorchakov 2, V. I. Bimatov3
1Tomsk Polytechnic University, 30 Lenin Street, Tomsk, Russia, 634050 2Stavropol State Agrarian University, 12 Zootechnical Street, Stavropol, Russia, 355017 3Tomsk State University, 36 Lenin Street, Tomsk, Russia, 634050
DOI : http://dx.doi.org/10.13005/ojc/310226
Article Received on :
Article Accepted on :
Article Published : 10 Jun 2015
The electrochemical activity of the glutathione (GSH) at the graphite electrodes modified with gold nanoparticles (AuNP-GCE) is formed under the ratio of reactants and in solutions of supporting electrolytes of 0, 1 M NaOH, 0,1 М phosphate buffer (pH 6.86), 0,1 M HCl, 0,1 M NaClO4 is studied by the method of cyclic voltammetry. The dependence of the electrochemical activity of GSH on ratio of reagents HAuCl4:Na3C6H5O7:NaBH4, used to the preparation of sols, and the modification time of the GE surface with gold nanoparticles have been studied. The “inverse” cathodic peak of GSH at AuNP-GCE formed in the reaction mixture HAuCl4:Na3C6H5O7:NaBH4=1:1:4 is observed on the cathodic branch of cyclic voltammogram at Ec = 0.05 V in 0.1 M NaOH. The mechanism of electrochemical oxidation of the GSH on the surface of AuNP-GCE in 0.1 M NaOH is proposed.
KEYWORDS:graphite electrode; gold nanoparticles; cyclic voltammetry; glutathione
Download this article as:| Copy the following to cite this article: Perevezentseva D. O, Skirdin K. V, Gorchakov E. V, Bimatov V. I. Electrochemical activity of glutathione at a graphite electrode modified with gold nanoparticles. Orient J Chem 2015;31(2). |
| Copy the following to cite this URL: Perevezentseva D. O, Skirdin K. V, Gorchakov E. V, Bimatov V. I. Electrochemical activity of glutathione at a graphite electrode modified with gold nanoparticles. Available from: http://www.orientjchem.org/?p=9165 |
Introduction
GSH (2-amino-5-{[2 – [(carboxymethyl) amino] – 1 – (merkaptometil)-2-oxoethyl] amino}-5-oksopentanoic acid) is a water-soluble tripeptide of γ-glutamilcysteinilglycine. GSH plays an important role in protecting the organism from disease, toxins, viruses, environmental hazards, radiation and the oxidative stress (free radicals). The GSH level in the cells determines the lifetime (Garaeva S.N. at all, 2009). Therefore the development of highly sensitive methods for the determination of GSH in the different biological matrices is an important task of analytical chemistry. Traditionally, the determination of GSH in a clinical investigations is carried out by chromatography (McDermott G.P., Francis P.S., 2011), optical methods (Hormozi-Nezhad M.R. at all, 2012), capillary electrophoresis (Wang X. at all, 2014), enzymic method (Tietze F., 1969). Disadvantages of these methods are the long time of sample preparation and analysis and high costs of the equipments. At the same time, electrochemical methods are essentially simpler and cheaper, than most of other methods, used in biochemistry, molecular biology, and medical diagnostics. The main limitation of the use of electroanalytical methods in biochemical analysis is a non-selectivity with respect to the substance to be determined. The components are generally found in low concentration in the presence of a wide range of substances that may obscure the results of analysis, or the presence of masking amino acids. GSH at a mercury dripping electrode is determined by polarographic method (Huska D. at all, 2007). This method is based on the formation of mercaptide of mercury on the surface of the electrode. The detection limit of GSH was 15 ∙10-3 M. However, the toxicity of mercury does not allow the use of these electrodes in laboratories. Solid electrodes are used to expand the opportunities of voltammetric analysis of GSH (Budnikov G.K. at all, 2007; Zaharchuk N.F. at all 2008). However, the use of solid electrodes also does not allow to decrease the GSH detection limit due to adsorption of the products of electrochemical oxidation of GSH on the electrode surface. The detection limit of the voltammetric determination of GSH at a platinum electrode is 1,9∙10-5 М (Zaharchuk N.F. at all 2008). Recently, chemical modified electrodes are used to a voltammetry determination of GSH. There are carbon electrodes modified with different materials such as graphite, microspheres and nanotubes (Apetrei С. at all (2011); Ensafi A.A. at all, 2010), cobalt phtalocyanine (Luz C.S. at all, 2007), ferrocene (Raoof J.B. at all, 2009), metal nanoparticals, its oxides and hydroxides (Chee S.Y. at all, 2011; Safavi A. at all, 2009; Raoof J.B. at all, 2009) in the voltammetric analysis of GSH. The detection limit of GSH at the carbon paste electrode modified with carbon microspheres was 6,4∙10-6 М (Apetrei at all, 2011). The detection limit of GSH at the carbon paste electrode modified with carbon nanotubes is 5∙10-6 М (Apetrei С. At all, 2011). The detection limit of GSH at the glass carbon electrode modified with cobalt phtalocyanine in poly-l-lysine film was 15 nM (Luz Cassia Silva at all, 2007). The modification of carbon paste electrode with 2,7-bis (ferrocenyl ethyl) fluoren-9-on allows to determine GSH with a detection limit equal to 2,1 ∙ 10-6 M (Raoof J.B. at all, 2009). The modification of the glass carbon electrode surface with oxide nikel allows to determine the GSH with a detection limit equal to 1∙10-4 M (Chee S.Y. at all, 2011). The detection limit of GSH at carbon paste electrode modified with hydroxide cupper (II) consists 3∙10-8 M (Safavi A., 2009). The detection limit of GSH at carbon paste electrode modified with oxide titanium (IV) and ferrocene acid consists 7∙10-4 M (Raoof J.B. at all, 2009). The modification of the indicator electrode with metal nanoparticles improves detection sensitivity and reduces detection limits of amino acids. Due to their specific properties the use of nano-sized metal particles as modifiers can decrease the limit of detection of organic substances (Perevezentseva D.O. , Gorchakov E.V., 2012; Compton R.G., 2006).Therefore, modification of the surface carbon electrode with metal nanoparticles is actual task.
The purpose of this work
is to investigate the electrochemical activity of GSH at the AuNP-GCE in aqueous solution by cyclic voltammetry, and to propose a mechanism of the electrochemical oxidation of GSH.
Experimental part
Materials and methods of investigation
Gold nanoparticles were prepared by chemical reduction of HAuCl4 with NaBH4 and sodium citrate described in the work (Perevezentseva D.O., Gorchakov E.V., 2012). The morphology of the gold nanoparticles was characterized by scanning electron microscopy (JSM-5500). pH values of sol were measured with a PH-150 instrument. Cyclic voltammetric mesurements were carried out with the voltammetric analyzer TA-2 (Tomsk) with a two electrode cell having the working electrode and a Ag/AgCl/KCl reference electrode at a temperature of 20˚C. The working electrodes were either a graphite electrode (GE), a GE modified with gold film electrode (Au-GE), or a AuNP-GCE.
Preparation of the AuNP-GCE
Cleaning the surface of the carbon electrode was carried out mechanically by dense filter paper, followed by an electrochemically cleaning. Modification of the surface of the graphite electrode was carried out as follows: The electrode was placed in an electrochemical cell filled with 10 ml of 0,1 M HCl solution with the addition of 0.1 ml of gold sol prepared as described above. The gold nanoparticles were electrochemically deposited on the surface of the graphite electrode at the accumulation potential Eacc = –1,0 V during the accumulation time tacc = 30 – 300 s. Then the electrode was removed from the solution, rinsed with double distilled water and placed in the supporting electrolyte 0,1 M NaOH for the voltammetric determination of GSH.
Preparation of the GE modified with gold (Au-GE)
Cleaning the surface of the carbon electrode was carried out mechanically by dense filter paper, followed by an electrochemically cleaning. Modification of the surface of the GE was carried out as follows: The electrode was placed in electrochemical cell filled with 10 ml of 10 mg·L-1 HAuCl4 The gold were electrochemically deposited on the surface of the GE at the accumulation potential Eacc = –1,0 V during the accumulation time tacc = 60 s. Then the electrode was removed from the solution, rinsed with double distilled water and placed in supporting electrolyte 0,1 M NaOH for the voltammetric determination of GSH.
Analytical procedures
A volume 10 ml of the supporting electrolyte was placed in the electrode cell. The following supporting electrolytes were used: 0,1 M NaOH, 0,1 М phosphate buffer (pH 6.86), 0,1 M NaClO4, 0,1 M HCl. The voltammograms of the supporting electrolyte were recorded by cyclic voltammetry under the following conditions: scan rate 0.100 V∙s–1, potential range E = –1,0 to +1,0 V. Then 20 µl of a GSH solution of unknown concentration were added to the solution. The obtained solution was stirred for 1 minute and then the cyclic voltammogram of GSH was recorded. The solutions of GSH were prepared in double distilled water before voltammetric measurements.
Results and discussions
The electrochemical activity of the GSH at the Au-GE, AuNP- GCE
Gold nanoparticles have a spherical shape. The average size of gold nanoparticles is 5-15 nm (Figure 1). The size of the gold particles depends on the nature of the reducing agent and conditions of the formation and storage (Perevezentseva D.O., Gorchakov E.V., 2012).
 |
Figure1: SEM-image of gold nanoparticles |
At a GE, GSH does not show an electrochemical signal at a concentration of 1,3×10-12 M in 0.1 M NaOH (Curve 1, figure 2). GSH is electrochemical active at AuNP-GE, Au-GE. In the presence of 1,3×10-12 GSH at the AuNP-GCE two wave are observed at Еа = -0,2 V and Еа = 0,15 V on the anodic branch of the cyclic voltammogram (Curve 2, figure 2). There is “inverse” cathodic peak in the presence of 1,3×10-12 GSH at Еc = 0,05 V on the cathodic branch of the cyclic voltammogram at the AuNP-GCE (Сurve 2, figure 2). The “inverse” cathodic peak of GSH at the AuNP-GCE formed in the reaction mixture HAuCl4:Na3C6H5O7:NaBH4=1:1:4 is observed at Ec = 0.05 V in 0.1 M NaOH on the cathodic branch of cyclic voltammogram. There are not peaks on the anodic branch of this cyclic voltammogram at the Au-GE in the presence of 1,3×10-12 GSH (Сurve 3, figure 2). In the presence of 1,3×10-12 GSH at the Au-GE the cathodic peak is observed at Еc = 0,05 V on the cathodic branch of the cyclic voltammogram (Curve 3, figure 2).
 |
Figure2: Cyclic voltammogramms GSH at: GE (1), AuNP-GCE(2), Au-GE (3) in solution 0,1 М NaOH, СGSH=1,3 ·10-12 M; scan rate 100 mV∙s-1 |
To choose optimal conditions in which GSH shows the greatest electrochemical activity were investigated the following dependences: the heights of the maxima of GSH from the nature of the supporting electrolyte, the ratio of reagents HAuCl4:Na3C6H5O7:NaBH4, used to the preparation of sols, and of the modification time of the GE surface with gold nanoparticles.
The electrochemical activity of the GSH at AuNP- GCE in the solution of different electrolytes
Firstly, the electrochemical activity of GSH was investigated at the AuNP-GE in solutions of follows supporting electrolyte: 0.1 M HCl, 0,1 M NaOH, 0,1 M NaClO4, 0,1 M phosphate buffer (pH 6.86). In the presence of 1,3×10-12 M GSH at the AuNP-GE on the anodic branch of the cyclic voltammogram in 0.1 M phosphate buffer (pH 6.86) peaks are not observed (Curve 1, figure 3). The maximum is observed at Еc = 0,5 V on the cathodic branch of this cyclic voltammogram at the AuNP-GCE (Curve 1, figure 3). Two waves are observed at Еа1 = 0,0 V and Еа2 = 0,8 V on the anodic branch of the cyclic voltammogram in 0,1 M NaClO4. There is not maximum on the cathodic branch of this cyclic voltammogram (Curve 2, figure 3). Two waves are observed at Еа1 = 0,2 V и Еа2 = 0,6 V on the anodic branch of cyclic voltammogram in 0,1 М HCl (Curve 3, figure 3). The maximum at Еc = 0,1 V is observed on the cathodic branch of cyclic voltammogram (Curve 3, figure 3).
 |
Figure3: Cyclic voltammogramms GSH at AuNP-GCE in: 1) 0,1 M phosphate buffer pH 6,86, 2) 0,1 M NaClO4 3) 0,1 M HCl, 4) 0,1 М NaOH. Условия: СGSH=1,3 10-12 M; scan rate100 mV∙s-1 Click here to View figure |
The electrochemical activity of the GSH at AuNP- GCE formed in the reaction mixture HAuCl4:Na3C6H5O7:NaBH4 under the different molar ratio and the time modification of the GE surface with gold nanoparticles
The dependence of the electrochemical activity of GSH on ratio of reagents HAuCl4:Na3C6H5O7:NaBH4, used to the preparation of sols, and the modification time of the GE surface with gold nanoparticles have been further studied.
In the presence of 1,3×10-12M GSH the “inverse” cathodic peak at the AuNP-GCE formed in the reaction mixture HAuCl4:Na3C6H5O7:NaBH4=1:1:4 only is observed at Ec = 0,05 V on the cathodic branch of cyclic voltammogram in 0,1 M NaOH (Figure 4, curve 1). There is not the “inverse” cathodic peak of GSH at the AuNP-GCE formed in the reaction mixture HAuCl4:Na3C6H5O7:NaBH4=1:1:6. The anodic peak is observed at Еа = 0,2 В on the anodic branch of cyclic voltammogram at the AuNP-GCE formed in the reaction mixture HAuCl4:Na3C6H5O7:NaBH4=1:1:6 (Curve 2, figure 4).The cathodic peak is observed at Ec = 0,1 V on the cathodic branch of cyclic voltammogram at the AuNP-GCE formed in the reaction mixture HAuCl4:Na3C6H5O7:NaBH4=1:1:6 (Curve 2, figure 4). When the ratio of reactants HAuCl4:Na3C6H5O7:NaBH4 = 1:1:10 and HAuCl4:Na3C6H5O7:NaBH4 = 1:1:40 gold particles at the GE are not electrochemically active, consequently GSH has not electrochemical activity.
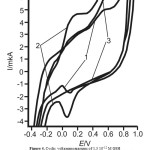 |
Figure4: Cyclic voltammogramms of 1,3 10-12 M GSH at AuNP-GCE formed in the reaction mixture HAuCl4:Na3C6H5O7:NaBH4: HAuCl4:Na3C6H5O7:NaBH4=1:1:4 (1), HAuCl4:Na3C6H5O7:NaBH4=1:1:6 (2), HAuCl4:Na3C6H5O7:NaBH4=1:1:6 (3) ω=100 mV∙s–1 Click here to View figure |
It was found that the electrochemical activity of GSH at the AuNP-GCE depends on the accumulation time of gold nanoparticles at GE. As seen from table 1, with the increase of accumulation time of gold nanoparticles from 30 to 90 s, the height of “inverse” cathodic peak of gold nanoparticles is increasing. With further increase of accumulation time from 90 to 240 s, the height of “inverse” cathodic peak of gold nanoparticles is decreasing. With further increase of accumulation time to 300 s, the height of “inverse” cathodic peak reaches its maximum value. Based on this dependence the optimal time of GE modification by gold nanoparticles is 90 s.
Table1: The magnitudes of height of “inverse” cathodic peaks of gold nanoparticles at GE on time of accumulation, supporting electrolytes 0,1 М NaOH
|
t, [s] |
30 |
60 |
90 |
120 |
150 |
180 |
210 |
240 |
270 |
300 |
|
I, [μА] |
0,2 |
0,3 |
0,4 |
0,22 |
0,2 |
0,2 |
0,18 |
0,18 |
0,5 |
0,5 |
The mechanism of the electrochemical oxidation of GSH at the AuNP-GCE
The electrochemical behavior of gold electrodeposited at the GE from the solutions and gold sols obtained by chemical reduction, it is difficult and involves the formation on the surface of the GE-Au okso- and hydroxi- compounds of metal in different oxidation states: Au(OH), Au(OH)3 or Au2O3 (Burke L, Nugent P., 1997; Burke L.D., Nugent P.F., 1998; Meretukov M.A. 2008; Perevezentseva D.O., Gorchakov E.V., 2014).
During the recording of the cathodic branche of this cyclic curve at GE modified with gold nanoparticles in 0,1 M NaOH (Curve 1, figure 4) is observed “inverse” maximum that can be described by the following scheme of the reaction equation (1):
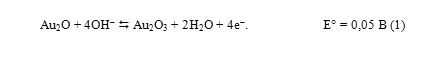
The cause of the appearance of “inverse” peak at the AuNP-GCE can be associated with a gold oxide from lower to higher oxidation states (e.g. Au2O to Au2O3), similar at mercury film to the process electrode of oxidation of Ag → Ag2O in alkaline solution (Brainina H.Z., 1988), NaClO4 (Korshunov A., Heyrovskyv M., 2009) or to the process electrode of oxidation of Cu2O → CuO (Korshunov A., Heyrovsky M., 2009), to the process OsO2 → OsO4 in H2SO4 (Kolpakova N.A., Kaminckaya O.V. at all, 1998), to the process of electroreduction of oxygen, catalyzed by the gold deposition (Korshunov A.V., Yosypchuk B. at all, 2011).
In the presence GSH the height of “inverse” cathodic peak at E = 0,05 V is increased. The process taking place at the AuNP-GE in the presence of GSH can be referred to electrocatalytical process of GSH oxidation by scheme of the reaction equation (2):
RSSR + Au2O + 4OH– → 2RS– + Au2O3 + 2H2O +2e (2)
Conclution
In this work an electrochemical activity of GSH is investigated at the GE, Au-GE, AuNP-GCE by cyclic voltammetry. 1,3·10-12 M GSH shows no electrochemical activity at the graphite electrode. It is shown that GSH is electrochemically active on the AuNP-GCE surface and the Au-GE in the supporting electrolyte of 0,1 M NaOH. It was first shown that the “inverse” cathodic peak of GSH at the graphite electrode modified with gold nanoparticles is observed at Ec = 0,05 V on the cathodic branch of cyclic voltammogram in 0,1 M NaOH. The effect of the ratio of reagents HAuCl4:Na3C6H5O7:NaBH4, used for gold sols and time modifying gold nanoparticles of the graphite electrode and the nature of the supporting electrolyte on electrochemical activity of GSH has been studied.
It was first shown that the presence of the “inverse” cathodic peak of GSH depends on the amount of sodium borohydride was consumed for obtaining the gold nanoparticles. The “inverse” maximum of GSH is observed at the graphite electrode modified with gold nanoparticles formed in the reaction mixture HAuCl4:Na3C6H5O7:NaBH4=1:1:4. The time modifying the graphite electrode by gold nanoparticles was experimentally determined and is equal to 90 s.
The electrochemical activity of GSH at the graphite electrode modified with gold nanoparticle in the solution of some supporting electrolytes has been investigated by method cyclic voltammetry. They are as follows 0,1 M NaOH, 0,1 M NaClO4, 0,1 M HCl, 0,1 M phosphate buffer (pH 6.86).
It is found that in the presence of GSH the height of the “inverse” maximum is observed on the cathode branches of cyclic voltammogram at graphite electrode modified with gold nanoparticles in 0,1 M NaOH. The mechanism of the electrocatalytic oxidation of GSH at the graphite electrode modified with gold nanoparticles is proposed.
There are further developments to investigate the mechanism of the electrochemical oxidation and reduction of GSH. The dependence of the height of “inverse” cathodic peak on the concentration of GSH will be investigated in biological objects.
References
- Apetrei С., Apetrei I.M., De Saja J.A. and all. (2011). Carbon paste electrodes made from different carbonaceous materials: application in the study of antioxidants, Sensors 11, 328-1344.
- Kolpakova N.A., Kaminckaya O.V., Jagovkina E.V. (1998). Opredelenie osmija v mineral’nom syr’e metodom kineticheskoi inversionnoi voltamperometrii, Zavodskaja laborotorija, Diagnostika materialov, 64 (4), 9-12.
- Korshunov A., Heyrovskyv M. (2009). Dispersion of silver particles in aqueous solutions visualized by polarography/voltammetry, Electrochimica Acta, 54, 6264-6268.
- Korshunov A., Heyrovsky M. (2009). Electrochemical behavior of copper metal core/oxide shell ultra-fine particles on mercury electrodes in aqueous dispersions, Journal of Electroanalytical Chemistry, 629, 23-29.
- Korshunov A.V., Yosypchuk B., Heyrovsky M. (2011). Voltammetry of aqueous chloroauric acid hanging mercury drop electrode, Collection of Czechoslovak Chemical Communications, 76, 929-936.
- Budnikov G.K., Ziyatdinova G.K., Valitova Ya.R. (2004). Elecrtochemical glutation determination, Journal Analytical Chemistry, 59 (6), 645-648.
- Burke L, Nugent P. (1997). The electrochemistry of Gold: I the redox behavior of metal in aqueous media, Gold Bulletin, 30, 43-53.
- Burke L.D., Nugent P.F. (1998). The electrochemistry of gold: II The electrocatalytic behavior of the metal in aqueous media, Gold Bulletin, 31, 39-50.
- Brainina H.Z. (1988). Ob obratimih pikah na polayrizacionnih krivih, Electrochemistry, 13, 678-680.
- Chee S.Y., Flegel M., Pumera M. (2011). Regulatory peptides desmopressin and glutathione voltammetric determination on nickel oxide modified electrodes, Electrochemistry Communications, 13, 963-965.
- Compton R.G. (2006). The use of nanoparticles in electroanalysis: a review, Analytical and Bioanalytical Chemistry, 384, 601-619.
- Ensafi Ali.A., Taei M., Khayamian T. (2010). Voltammetric measurement of trace amount of glutathione using multiwall carbon nanotubes as a sensor and chlorpromazine as a mediator, Journal Solid State Electrochemistry, 14, 1415-1423.
- Garaeva S.N., Redkozubova G.V., Postolati G.V. (2009). Aminokisloty v zhivom organizme, Kishinev: AN Moldovy.
- Hormozi-Nezhad M.R., Seyedhosseini E., Robatjazi H. (2012). Spectrophotometric determination of glutathione and cysteine based on aggregation of colloidal gold nanoparticles, Scientia Iranica, 19 (3), 958-963.
- Huska D., Zitka O., Adam V. and all. (2007). A sensor for investigating the interaction between biologically important heavy metals and glutathione, Czech Journal of Animal Science, 52 (2), 37-43.
- Li-Ya Niu, Ming-Yan Jia, Peng-Zhong Chen at all. (2015). Colorimetric sensors with different reactivity for the quantitative determination of cysteine, homocysteine and glutathione in a mixture, RSC Advances, 17, 13042-13045
- Luz C.S., Santos D.F. and all. (2007). Electrocatalytic determination of reduced glutathione in human erythrocytes, Analytical and Bioanalytical Chemistry, 387 (2), 1891-1894.
- McDermott G.P., Francis P.S., Holt K.J. at all. (2011). Determination of intracellular glutathione and glutathione disulfide using high performance liquid chromatography with acidic potassium permanganate chemiluminescence detection, Analyst, 136, 2578-2585.
- Meretukov M.A. (2008). Zoloto. Moscow. Ruda I metal.
- Perevezentseva D.O., Gorchakov E.V. (2014). Electrochemical response of gold nanoparticles at a graphite electrode, Advanced materials research, 1040, 297-302.
- Perevezentseva D.O., Gorchakov E.V. (2012). Voltammetric determination of cysteine at a graphite electrode modified with gold nanoparticles, Journal of Solid State Electrochemistry, 16 (7), 2450-2410.
- Raoof J.B., Ojani R., Baghayeri M. (2009) Simultaneous electrochemical determination of glutathione and tryptophan on a nano-TiO2/ferrocene carboxylic acid modified carbon paste electrode, Journal Sensors and Actuators: B: 143.. P. 261-269.
- Raoof J.B., Ojani R, Karimi-Maleh H. (2009). Electrocatalytic oxidation of glutathione at carbon paste electrode modified with 2,7-bis (ferrocenyl ethyl) fluoren-9-one: application as a voltammetric sensor, Journal of Applied Electrochemistry, 39, 1169-1175.
- Safavi A., Maleki N., Farjami E. and all. (2009). Electrochemical determination of glutathione and glutathione disulfide at a nanoscale copper hydroxide composite carbon ionic liquid electrode, Analytica Chimica Acta, 81, 7538-7543.
- Tietze F. (1969). Enzymic method for quantitative determination of nanogram amounts of total and oxidized glutathione: Applications to mammalian blood and other tissues, Analytical Biochemistry, 27, 502-522.
- Wang X, Wang J, Fu H, Liu D at all (2014). Determination of glutathione in single HepG2 cells by capillary electrophoresis with reduced graphene oxide modified microelectrode, Electrophoresis, 35(23), 3371-3378. DOI: 10.1002/elps.201400155.
- Zaharchuk N.F., Borisova N.S., Titova T.V. (2008). Study of tiolsulfid equilibrium in whole blood and its fractions by stripping voltammetric titration, Journal Analytical Chemistry, 63 (2), 189-198.

This work is licensed under a Creative Commons Attribution 4.0 International License.









