Synthesis, spectral and biological studies of new Mn(II) and Fe(II) complexes of 2,3-Butanedione 3-monoxime
V. H. Rajurkar*, V. N. Kamble
Department of Chemistry, D. G. Ruparel college of Arts, Science and commerce, Senapati Bapat Marg, Mahim, Mumbai, 400 016, (MS), India.
DOI : http://dx.doi.org/10.13005/ojc/310171
Article Received on :
Article Accepted on :
Article Published : 28 Mar 2015
Reaction between 2,3-Butanedione 3-monoxime, 3-aminophenol and some hydrated metal salts of MnII and FeII give complexes of the type [ML22(H2O)]. The ligand and the complexes were characterized by elemental analysis, magnetic susceptibility measurements, spectral (U.V.Vis., I.R., 1H N.M.R.) analysis and X-ray powder diffraction studies. Spectral studies indicate bidentate nature of the ligand and coordination occurs through oxime oxygen after deprotonation and nitrogen of azomethine. The spectral and magnetic studies suggest octahedral geometries for both the complexes. A triclinic system was proposed by X-ray powder diffraction study of Schiff base and metal complex. Anti-bacterial and anti-fungal activity have been studied using the agar well diffusion method and ditch diffusion method. The activity data shows that the metal complexes are more potent antibacterial and antifungal than the parent Schiff base ligand.
KEYWORDS:Schiff base; transition metal complexes; Triclinic system; Biological activity
Download this article as:| Copy the following to cite this article: Rajurkar V. H, Kamble V. N. Synthesis, spectral and biological studies of new Mn(II) and Fe(II) complexes of 2,3-Butanedione 3-monoxime. Orient J Chem 2015;31(1). |
| Copy the following to cite this URL: Rajurkar V. H, Kamble V. N. Synthesis, spectral and biological studies of new Mn(II) and Fe(II) complexes of 2,3-Butanedione 3-monoxime. Orient J Chem 2015;31(1). Available from: http://www.orientjchem.org/?p=8106 |
Introduction
Ligand, a metal surrounded by a cluster of ions or molecule, is used for preparation of complex compounds named as Schiff base, which are condensation products of primary amines and aldehydes or ketones (RCH=NR’, where R and R’ represents alkyl or aryl substituent). Such schiff base metal complexes play an important role in agriculture, pharmaceutical and industrial chemistry. Schiff base metal complexes gain much importance in some potentially useful biological activities like antibacterial activity, antifungal activity[1-2], antitumour activity[3], antiinflamatory activity [4-5], antioxidant activity [5-7], hepatotoxicity[8], and pesticidal activity[9] etc. Thiophene derivatives are well known to exhibit an array of biological activity such as antibacterial and antifungal activity [10]. Condensed thienopyrimidines exhibit interesting antibacterial activity [11-12]. Thus metal complexes with various donar groups had attracted by many workers.
In this paper we reported the study of 3-aminophenol 2,3-Butanedione 3-monoxime (HL) and their complexes with transition metal ions like MnII and FeII. The main interest in this ligand originates in its oxime group containing -N-O- donar atoms.
Materials and Methods
Ligand was prepared according to the literature method [13-14]. All the chemicals used for synthesis were analytical grade. 2,3-Butanedione 3-monoxime was obtained from Thomas Baker, 3-aminophenol obtained from Aldrich.
Method 1. Synthesis of Schiff base Ligand (HL)
2,3-Butanedione 3-monoxime (1.0gm) and 3-aminophenol (1.5gm) were dissolved separately in ethanol and mixed together and refluxed for 1-2 hrs. in water bath. After heating insoluble materials were removed by filtration and cooled. Dark brown colored HL was formed washed it with ethanol and then dried over anhydrous CaCl2. Yield of ligand was 99%. The physical and analytical data listed in table 1.
 |
Table 1 Click here to View table |
Method 2. Synthesis of Metal Complexes
The prepared ligand and metal salts in 1:1 proportion were dissolved separately in ethanol and mixed together and refluxed for 2-3 hrs. in water bath. After cooling insoluble materials were removed by filtration, colored crystalline metal complexes were formed, recrystalized and dried over anhydrous CaCl2.
 |
Scheme 1 Click here to View scheme |
Antibacterial activity and Antifungal activity
The synthesized ligand and complexes were screened for in vitro growth inhibitory activity against gram positive bacteria Staphylococcus aureus and gram negative bacteria Pseudomonas aeruginosa. Nutrient agar plates were made uniformly surface inoculated from the culture of the tested microorganisms. Well was made on the agar medium at the centre. Normal saline was used to prepare the inoculums of the bacteria to be used for the antibacterial study [15-16].
 |
Figure 1 Click here to View figure |
 |
Figure 2 Click here to View figure |
The synthesized ligand and complexes also screened for in vitro growth inhibitory activity against Aspergillus niger and Candida albicans. Sabouraud dextrose agar plates made uniformly and ditch made on the agar medium at the centre. Two separate striking lines of fungi were made on both the sides of ditch [16].
Mueller Hint Agar Media (HI Media) was used to subculture various strains of microorganisms. The lowest concentration of the compound which shows positive antibacterial and antifungal activity was subjected to dilution method for quantitative measurement of inhibitory (micro static) activity. The lowest concentration which completely inhibited visible microbial growth was recorded as the Minimum inhibitory concentration (MIC, µg/ml). The stock solution of test compounds (0.04mg/ml) was prepared in DMF and subsequent dilutions (80%, 60%, 40%, and 20%) of stock solutions were made in the same solvent. Under aseptic conditions, the diluted test solutions with different concentrations were added to the disc and ditch were placed on the numbered plates. Then the plates were incubated at room temperature for 24 hr. During this period, the test solution diffused and the growth of the inoculated microorganisms was affected. Antibacterial activity was indicated by the presence of clear inhibition zone around the well and antifungal activity was indicated by the presence of inhibition zone nearer to the ditch. The activity of standard ciprofloxacin and fluconazole were also recorded using the same procedure as above at the same concentrations and solvent.
 |
Figure 3 Click here to View figure |
 |
Figure 4Click here to View figure |
Results and Discussion
The complexes are stable in air but decomposed at high temperature. Easily soluble in Dimethylformamide (DMF) and dimethylsulphoxide (DMSO) .The physical and elemental analyses of the ligand and metal complexes are recorded in table 1.
 |
Figure 5 Click here to View figure |
Electronic Spectra and Magnetic moment
The solid state electronic spectra of the complexes are recorded in table 2 lists the important electronic spectral bands of the complexes.
The electronic spectrum of ligand and complexes helps to indicate the geometry. The electronic spectra of the ligand show strong absorption band at 264 nm but in complexes this band is slightly shifted to lower frequency [17] which are π → π* charge transfer transitions. The band at 425nm can be assigned to the n→ π* transitions of the azomethine group. In the spectra of all metal complexes, the bands of the azomethine chromophore n→ π* transitions are shifted to higher frequencies indicating that the imine nitrogen atom is involved in coordination to the metal ion [18]. Those very low intensity absorption bands associated with d-d transitions for MnII complex are 6A1g → 4T2g (D) (506nm), 6A1g → 4T2g (D) (624nm) and 6A1g → 4T1g (710nm) indicates octahedral geometry[19-20] and for FeII complex are 6A1g 4T1g (585) and 6A1g 4T4g (490) indicates octahedral geometry[19].
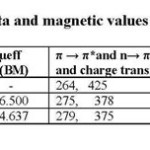 |
Table 2 Click here to View table |
The room temperature magnetic moment values also help to indicate the geometry. The magnetic moment value of MnII in the rang 5.7 to 6.0 BM is close to theoretical spin only value (μeff = 6.4 BM) for Mn2+ (d5 system) and for FeII in the rang 5.0 to 5.6 BM, is close to theoretical spin only value (μeff = 4.638 BM) for Fe2+ (d6) indicates octahedral geometry of the complexes [21].
Infrared Spectra
The I.R. bands of ligand and metal complexes are listed in table 3. The I.R. spectra of the complexes shows, complexes behave as a bidentate coordinating ligand via azomethine nitrogen (C=N*) and oxygen of oxime (-NOH) group by replacement of hydrogen ion forming six membered ring around metal ion. In the ligand azomethine (C=N*) group is at 1630 cm-1 wave number but in metal complexes it shifts to lower wavenumber, this indicates that imine nitrogen atom involved in coordination to the metal ion [18, 21] while oxime (C=N) remain more or less at the same position in both ligand and metal complexes. The N-O band in ligand at 998 cm-1 but in metal complexes it shifts to higher wavenumber, this indicates that oxygen atom by replacing oxime proton involved in coordination to the metal ion [20]. A band observed in the ligand at 3296 cm-1 due to N-H group present in the imidazole ring which is rarely observed in metal complexes. Phenolic (O-H) in ligands and their metal complexes is observed in rang 3365-2609cm-1 means (O-H) of hydroxyl group does not involved in coordination or ligand formation. A broad band observed in both the complexes in rang ~3396 cm-1 due to (O-H) of the coordinated H2O. This is supported by the appearance of an additional band in the rang 950-900 cm-1 for (O-H) rocking deformation and 800-750 cm-1 for (O-H) wagging mode of coordination which is not observed in the ligand spectrum[20]. Thus H2O is coordinated in metal complexes. New bands appear in the 450-420 and 400-360 cm-1 assignable to the υ(Mn-O) and υ(Mn-N) respectively in metal complexes only [22, 23]. Thus proposed geometry is the octahedral geometry of the complexes.
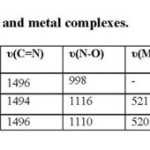 |
Table 3 Click here to View table |
1H N.M.R. Spectral analysis
The proton nuclear magnetic resonance for the Schiff base was recorded in deutarated CDCl3 as a solvent. Spectrum was critically examined and chemical shift for various protons were calculated with respect to Tetra methyl silane (TMS) as a standard. In all Schiff bases a broad peak of–OH of oxime is not observed due to rapid exchange with the solvent CDCl3.There are two signals in between 1.85 to 2.20 ppm are due to presence of methyl protons in the Schiff base. A signal of phenyl –OH for HL is observed at 9.73ppm. Similarly Multiple signals in the region δ = 7.10-8.00 ppm are assignable to aromatic protons.
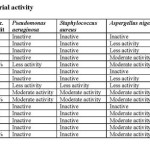 |
Table 4 Click here to View table |
X-ray Analysis
Single crystal X-ray crystallographic investigation is the most precise source of information regarding the structure of the complex, the difficulty of obtaining crystalline complexes in proper symmetric form has rendered the powder X-ray diffraction method for such study. The X-ray diffraction pattern of the complexes indicates high crystalline nature of the complexes. For HL there are only 6 reflections between 8 to 25 (2θ in deg.) with maximum at 2θ = 8.92 deg. corresponding to value of d = 9.9055A and miller indices (hkl) -101. For Mn.HL2.2H2O there are only 13 reflections between 16 to 46 (2θ in deg.) with maximum at 2θ = 20.12 deg. corresponding to value of d = 4.4099 A and miller indices (hkl) 101. Fe complex doesn’t show intense peaks in the diffractogram indicating the amorphous nature of the complex. Ligand and Mn complex shows intense peaks in the diffractogram showing crystalline in nature. The main peaks of the complex have been indexed by using computer software by trial and error method [24-26] keeping in mind characteristics of various symmetry systems could be obtained between observed and calculated 2θ and sin²θ values. The method also yielded h k l (miller indices) values. A comparison of values of 2θ and sin²θ for the complex reveals that, there is good agreement between the calculated and observed values of 2θ and sin²θ on the basis of assumption of triclinic structure [25] for the complexes. The small difference in the observed d-spacing can be attributed to difference in unit cell dimensions. The ligand HL yield values for lattice constant a = 13.5126A, b = 15.3335A and c = 6.5519A; α = 111.276D, β = 120.708D and γ = 136.572D; the unit cell volume V³ = 684.33. And Mn.HL2.2H2O complex yield values for lattice constant a = 16.9654A, b = 5.8013A and c = 4.9245A; α = 102.659D, β = 95.226D and γ = 101.200D; the unit cell volume V³ = 459.34. In conjugation with these lattice parameters in ligand and metal complex the condition such as a ≠ b ≠ c and of α ≠ β ≠ γ required for all the samples to be triclinic were tested and found to be nearly equal to it.
Biological activity
Antibacterial activity
The synthesized ligand and complexes were screened for in vitro growth inhibitory activity against gram positive bacteria Staphylococcus aureus and gram negative bacteria Pseudomonas aeruginosa. A comparative study of Schiff bases and metal complexes indicate that the Mn complex exhibit higher antibacterial activity than the free ligand but as compared to standard ciprofloxacin the activity is less. Fe-complex does not show antibacterial activity. The activity of the ligand and metal complexes are shown in table 4.
Antifungal activity
The synthesized ligands and complexes also screened for in vitro growth inhibitory activity against Aspergillus niger and Candida albicans. Mn complex shows more activity than ligand. Fe-complex shows moderate activity against Aspergillus niger only and doesn’t show any activity against Candida albicans. But as compaired to standard fluconazole this antifungal activity is very less. The activities of compounds are recorded in table 4.
It has been observed from the results that the metal complexes have moderate activity than the free ligand. HL shows poor inhibitory zone up to 16mm for Pseudomonas aeruginosa at higher concentration only and its Mn-complex shows less inhibitory zone at higher concentration for Pseudomonas aeruginosa as well as Staphylococcus aureus. There is moderate activity for ligand against Aspergillus niger and very less activity against Candida albicans. In case of metal complexes there is moderate activity against Aspergillus niger and less to moderate activity against Candida albicans. Fe-complex shows moderate to higher activity against Aspergillus niger and there is no any activity against Candida albicans. It has been suggested that the complexes having antifungal activity may act either by killing the microbes or by inhibiting multiplication of the microbes by blocking their active sites of enzymes [27].This is also probably due to the greater lipophilic nature of the complexes. Such increased activity of the metal chelates can be explained on the basis of Overtone’s concept and chelation theory. According to Overtone’s concept of cell permeability the lipid membrane that surrounds the cell favours the passage of only lipid soluble materials due to which liposolubility is an important factor which controls the antimicrobial activity. On chelation, the metal ion will be reduced to a greater extent due to the overlap of the ligand orbital and partial sharing of positive charge of the metal ion with donar groups. Further, it increases the delocalization of π-electron over the whole chelate ring and enhances the lipophilicity of the complex. This lipophilicity enhances the penetration of the complex into lipid membrane and blocks the metal binding sites on enzymes of microorganisms.
Conclusion
A series of metal complexes of 3-aminophenol and 2,3-Butanedione 3-monoxime with MnII and FeII have been synthesized and characterized. All complexes exhibit octahedral geometry by involvement of azomethine nitrogen and oxime oxygen of the ligand in complex formation. Ligand and the Mn-complex crystallizes in the triclinic system. All the compounds and ligand have been screened for antibacterial and antifungal activity. Complexes have exhibited promising antibacterial activity as compared to Schiff base, hence probable applications of these metal complexes are in medicinal use.
Acknowledgements
Authors are thankful to Department of Microbiology, Walchand College of Biotechnology, Solapur for providing biological activity facility and Chemical Science Department, IIT Mumbai, for providing elemental and spectral analysis facility.
References
- Mele J., Ali M.M., Salahuddin M.S., Habib M.R., Khanam J.A., Mycobiology,, 2008, 36(1),70–73.
- Chohan Z.H., Sumrra S.H., Youssoufi M.H., Hadda T.B., Eur. J. Med.Chem. 2010, 45(7), 2739-47.
- Jesmin M., Ali M.M., Khanam .A., Thai. J. Pharm. Sci., 2010, 34, 20-31.
- Mele J., Islam M.K., Shaikh M.M., Zahan R., Int.J.A.PS.BMS, 2013, 2.(4), 272-279.
- Kalyani G.A., Purnima Ashok,1 Taranalli A.D., Ramesh C.K., Krishna V., A.H.M Swamy V., Indian J Pharmacol, 2011, 43(6), 740–741.
- Gujarathi J. R., Pawar N. S., Bendrea R. S., Der Pharma Chemica, 2013, 5(4), 120-125.
- Chidanandaa B., Venugopala Reddy K. R. *, Harishc M. N. K., Pradeepa K. M., Mruthyunjayacharia C. D., Ganeshd S. D., Prashith Kekudae T. R., Der Pharm Chemica, 2013, 5(4), 293-300.
- Ali M.M., Jesmin M., J.Natn.Sci.Foundation Sri Lanka, 2010, 38(2), 145-149.
- Ali M.M., Jesmin M., Salam S. M. A., Khanam J. A., Islam M. F., J. Sci. Res. 2009, 1 (3), 641-646.
- Swamy V. A.,. Pathak U. S, Rajasolomon V., Meena S., Ramseshu K.V., Rajesh R., Indian J Hetercycl chem., , 2004, 13, 347-355.
- Marie F. R., Hans J. S., Eoghan M.M.G., Cormac T.D., Adrain D., M & G. Declan G, J. Mol Catal A, Chem, , 2005, 231, 205-212.
- Shrivastava H. Y., Devaraj S. N. & Nain B. U., J. Inorg Biochem, , 2004, 98, 387-395.
- De R.L., Mandal M., Roy L., Mukherjee J. Indian J. Of Chem., 2008, 47A, 207-213.
- Osowole A.A. Kolawole G.A., Fagade O.E., J. of Coordination Chem. 2008, 61(7),1046-1055.
- Pandeya S.N., Raja A.S., Nath G., Indian J. of chem., 2006, 45B ,494-499.
- Raman N. Dhaveethu R., Sakthivel A., J. of chem. Sci. 2007, 119(4), 303-310.
- Samanta B., Chakraborty J., Choudhury C.R., Dey S.K. Dey D.K., Batten S.R., Jensen P. Glenn P.A., Mitra S., Struct. Chem., , 2007, 18, 33-41.
- Sonmez M., Enciyes Universities Fen Bilimleri Enstitusu Dergisi, 2008, 24,308-314.
- Abu G.M., El-Reash, Ibrahim K.M., Bekheit M.M., Tran. Metal chem, 1990,., 15,148-151.
- Sawant V.A., Gotpagar S.N., Yamgar B.A., chavan S.S., Spectrochimica Acta part A:Molecular & Biomolecular Spectroscopy, 2009, 72, 663-669.
- J. D.Lee, Concise Inorganic Chemistry, fifth edition, Blackwell Science ltd., London, (1996), 668-669.
- Bagihalli G.B., Avaji P.G., Badami P.S., Patil S.A., J. of coord. Chem,2008, ., 61(17), 2793-2806.
- Sharma V.K., Srivastava S., Srivastava A., Bioinor. chem. and appli., 2007, Artical ID-68374, 10 pages.
- Klug H. P., Le Alexander, X-ray Diffraction procedure for polycrystalline and Amorphous Materials, John Wiley, London, NY, (1974).
- Cullity B.D., Elements of X-ray Diffraction, Second edition., Addison-Wesley, (1977).
- E. Wu. School of physical science, Powdmult- an interactive powder diffraction data, interpretation and indexing program version 2.3, Flinders University of South Australia, Bedford park, Australia, S. A. 5042.
- Mohamed G.G., Omar M.M., Hindy A.M.M., Spectrochimica Acta part A 62, 2005, 1140 -1150.

This work is licensed under a Creative Commons Attribution 4.0 International License.









