Synthesis, Structural, Spectroscopic and Antibacterial Studies of Schiff Base Ligands and Their Metal Complexes Containing Nitrogen and Sulphur Donor Atom
B.K. Rai* and Rachana kumari1
*Dept. of Chemistry, L.N.T. college, Muzaffarpur .R.A Bihar University, Muzaffarpur, India 1Department of Bioteechnology, Amity University, Noida-201303, India
DOI : http://dx.doi.org/10.13005/ojc/290345
Article Received on :
Article Accepted on :
Article Published : 28 Oct 2013
In continuation to our work for the preparation of Schiff base complexes of transition metal and their antimicrobial screening, we in the present communication have synthesized some new Co(II), Ni(II) and Cu(II) complexes with tridentate Schiff base ligand, 3-amino 2-ethyl quinazoline 4(3H) thiosemicarbazone (AEQT). The obtained complexes were characterized on the basis of their molar mass, elemental analyses, electronic spectra, IR, magnetic susceptibility, molar conductance as well as thermal analysis. Spectral studies show that the Schiff base behaves as tridentate and coordination takes place through primary amino group, azomethine nitrogen and thione sulphur atom. The Schiff base and their complexes have been screened for their antibacterial effect by disc diffusion method.
KEYWORDS:Schiff base;AEQT;Co(II), Ni(II) and Cu(II);complexes;antibacterial effect
Download this article as:| Copy the following to cite this article: Rai B.K, Kumari R. Synthesis, Structural, Spectroscopic and Antibacterial Studies of Schiff Base Ligands and Their Metal Complexes Containing Nitrogen and Sulphur Donor Atom . Orient J Chem 2013;29(3). doi : http://dx.doi.org/10.13005/ojc/290345 |
| Copy the following to cite this URL: Rai B.K, Kumari R. Synthesis, Structural, Spectroscopic and Antibacterial Studies of Schiff Base Ligands and Their Metal Complexes Containing Nitrogen and Sulphur Donor Atom . Orient J Chem 2013;29(3). Available from: http://www.orientjchem.org/?p=352 |
INTRODUCTION
Schiff bases1-9 are useful chelators because of their ease of preparation, structural varieties, varied denticities and subtle steric and electronic control on their framework. Recently a great deal of interest have developed in the synthesis and characterization of transition metal complexes containing Schiff bases as ligands due to their applications as catalysts for many reactions and relation to synthetic and natural oxygen carriers. The biocidal effect of Schiff bases as well as its metal complexes is of interest due to their pharmacological activities. Keeping the above facts in mind and in continuation of our recent work10-15 on Schiff bases and their metal complexes, in our present endeavour, we have chosen to synthesis of a Schiff base 3-amino 2-ethyl quinazoline 4(3H) thiosemicarbazone (AEQT) and its Co(II), Ni(II) and Cu(II) metal complexes. The ligand and its metal complexes were also screened for in vitro antibacterial activity against Staphylococcu aureus and Escherichia coli.
EXPERIMENTAL
All the chemicals and reagents used were analytical grade. The solvents were used without any purification. The metal contents were determined using standard procedures16. IR spectra of the ligand and complexes were recorded in KBr pellets with a Perkin Elmer model 577 spectrophotometer in the 4000-200 cm-1 range. The electronic spectra were recorded in DMF on a Cary 2390 spectrophotometer. Molar conductivity of complexes were measured at room temperature by using Systronics conductivity meter model 303 in DMF. Magnetic susceptibility was carried out by Gouy method at room temeprature.
Preparation of the ligand :
A suspension of 3-amino 2-ethyl quinazoline 4(3H) one was treated with thiosemicarbazide hydrochloride in molar ratio 1:1 dissolved in 10% alcoholic solution of sodium acetate. The mixture was refluxed on water bath for 3 and half h, when a crystalline colourless solid began to separate after allowing the solution to stand overnight. The solids were cooled, filtered and washed with aqueous ethanol. The compounds was recrystallized with ethanol. M.P. 163±1oC; Yield 65%.
Preparation of the complexes
1 m mol of the metal halides in ethanol (10 ml) was added to a 2 m mol Schiff base (AEQT) in ethanol (20 ml) and kept under reflux for 3 h. The crystalline coloured solid complex formed on cooling was filtered and washed with ethanol. It was then recrystallized frorm methanol and dried in oven. Yield 65-70%.
RESULTS AND DISCUSSION
The characteristic IR bands for the free ligands when compared with metal complexes provide meaningful information regarding the bonding sites of the ligands (Table-2). The Schiff base, AEQT shows strong and broad band in the region 3280 cm-1 assigned17 to nN–H vibrations. In the complexes this band shifted to 3260 cm-1 indicating coordination of N atom of primary amino group. The band at 1635 cm-1 assigned18,19 to nC=N in ligand is shifted to 1605 cm-1 showing the coordination of azomethine20 nitrogen. The linkage with N atom is further supported by the appearance of a band in the far IR region at 455-425 cm-1 assigned21 to nM–N. The IR spectra of the ligand shows a strong and broad band at 800 cm-1 assigned22 to nC=S. In the complexes this band shifted to 775 cm-1 indicating coordination of thione sulphur of thiosemicarbazone moiety. The linkage with sulphur is further supported by the appearance of a new band in the far IR region at 405-390 cm-1 assigned23 to nM–S. It may therefore be that in the present complexes the ligand is neutral tridentate bonding proposes through N atom of primary amino, azomethine N and thione sulphur atom.
Electronic spectra and magnetic susceptibility of the complex :
The electronic spectra24,25 and magnetic susceptibility26 value tentatively proposes octahedral geometry for Co(II), Ni(II) and Cu(II) complexes.
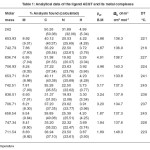 |
Table-1: Analytical data of the ligand AEQT and its metal complexes Click here to View table |
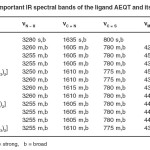 |
Table – 2: Important IR spectral bands of the ligand AEQT and its complexes Click here to View table |
![Fig.1 [M(AEQT)2] X2](http://www.orientjchem.org/wp-content/uploads/2013/11/Vol29_No3_rai_synth_fig1-150x150.jpg) |
Fig.1 [M(AEQT)2] X2M = Co(II) and Ni(II); X = Cl–, Br– and I– ; M = Cu(II); X = Cl– and Br– R = Ethyl |
Molar conductivity:
Conductivity of the complexes were measured in the solvent dimethyl formamide at the concentration 10-3 M and all the complexes were found to be electrolytic in nature of 1:2 type. The molar conductivity value of the complexes are found in the range 136.7-149.3 ohm-1 cm2 mol-1.
Antimicrobial results :
All the Co(II), Ni(II) and Cu(II) complexes as well as ligand AEQT were tested against Gram positive and Gram negative bacteria. The results indicates that all the complexes were active agaainst Gram positive bacteria while less active against E. coli which is Gram negative. The complexes were more active than the Schiff27 base which indicates that metallation increases the activity.
In vitro antibacterial activity of the ligand and the metal complexes were tested using paper disc diffusion method28.
Conclusion
On the basis of analytical and spectral data the tentative structure of Co(II) and Ni(II) complexes are proposed to be octahedral in geometry whereas distorted octahedral geometry is proposed for Cu(II) complexes. The tentative structure of the complexes may be assigned as follows in Fig.-1.
REFERENCES
- Alexander A., Chem. Rev., 95, 273 (1995).
- Vigato P. A., Tamburini and Bertolo L., Coord. Chem. Rev., 251, 1311 (2007).
- Padhye S. B. and Kalfman G. B., Coord. Chem. Rev., 03, 127 (1985).
- Dilwarth J. R., Amold P, Morales D., Wong Y. L. and Cheng Y., Modern Coord. Chem., 217 (2002).
- Quiruga A. G. and Raninger C. N., Coord. Chem., Rev., 248, 119 (2004).
- West D. X., Liberta A. E., Padhye S. B., Chikate, R. C., Sonawane P. B., Kumbhar A. S. and Yerande R. G., Coord. Chem. Rev. 123 (1993).
- Wang T. and Guoz, Curr. Med. Chem., 13, 525 (2006).
- Coires A. C. F., Antixancer Agents in Med. Chem., 7, 484 (7).
- Lobana T. S., Shama R., Bawa G., Khanna S., Coord. Chem. Rev., 253, 977 (2009).
- Rai B.K. and Anand Rahul, Asian J. Chem., 25, 583 (2013); Rai B. K. and Kumari Rachana, Asian J. Chem., 23, 4625 (2011); Rai B. K., Kumari Rachana and Thakur Amrita, Orient J. Chem., 28, 943 (2012).
- Rai B.K., Thakur Amrita and Divya, Asian J. Chem.; 25, 583(2013).
- Rai B.K. and Vidyarthi S.N., kumara Punam, Kumari Suman, Kumari Lakshmi and Singh Rajkishore, Asian J. Chem., 25, 941(2013).
- Rai B.K. and Kumar Arun, Asian J. Chem., 25, 1160(2013).
- Rai B.K., J. Indian Chem., Soc; 90, 105(2013).
- Rai B. K., Vidyarthi S. N., Sinha Puja, Singh Vineeta and Kumar Sanjiv, Orient J. Chem., 29, 271 (2013),
- Vogel A.I., A textbook of quantitative Chemical Analysis, Revised by Menaham J., Denny R.C., Barnes J.D. and Thomas M, Pearson Education, 7th Edn, London (2008).
- Cook D., Can J. Chem., 39, 2009 (1961).
- Percu G. C. and Thornton D. A., Inorg. Nucl. Chem., Lett., 7, 599 (1971).
- Kolinski R. A. and Korybut Daszkiewiez, Inorg. Chim. Acta, 14, 237 (1975).
- Meglynn S. P. and Smith J. K., J. Mol. Spectros, 6, 164 (1961).
- Babukutty P. V., Prabhakaran C. P., Anantaraman R. and C. G. R. Nair, J. Inorg. Nucl. Chem., 36, 3685 (1974).
- Banerjee P., Singh I. P., Indian J. Chem., 6, 34 (1968).
- Ferraro J. R., Low Frequency Vibration of Inorganic and Coordination Compound, Plenum Press, New York.
- Lever A.B.P., Inorganic Electronic Spectroscopy, Elsevier, Amsterdam, 395 (1968).
- Figgis B.N., Introduction to LIgand Field, Wiely Eastern Ltd., New Delhi, 279(1976).
- Carlin R.L. and Van Deynevetedt A.J., Magnetic properties of transition metal compounds springer-ver lag, New York, (1997).
- Singh H. Z., Khungar B., Tripathi U. D., Varshney A. K., Main Group Met. Chem., 24, 5 (2003).
- Mukherjee P. K., Saha K., Giri S. N., Pal M. and Saha B.P., Indian J.l Microbiology, 35 (1995).

This work is licensed under a Creative Commons Attribution 4.0 International License.









