Physico-Chemical and Antifungal Properties of Trypsin Inhibitor from the Seeds of Mucuna Pruriens
K. S. Chandrashekharaiah
Department of Biotechnology, PES Institute of Technology, BSK III Stage, Bangalore – 560085, Karnataka, India
DOI : http://dx.doi.org/10.13005/ojc/290329
Article Received on :
Article Accepted on :
Article Published : 28 Oct 2013
The trypsin inhibitor has been purified to apparent homogeneity from the seeds of Mucuna pruriens employing ammonium sulfate fractionation, cation exchange chromatography on CMcellulose and gel-permeation chromatography on Sephadex G-100. The purified Mucuna pruriens trypsin inhibitor (MPTI) showed a specific inhibitor activity of 474.66, fold purity of 99.51 and the yield obtained was 22.08%. The homogeneity of the purified preparation was confirmed by PAGE, IEF and SDS–PAGE. The molecular weight determined by gel-permeation chromatography and SDS–PAGE were 12 and 11.6 kDa respectively. The pI of MPTI was found to be 5.6 ± 0.05. MPTI was found to be stable at all temperatures lying between 0 and 90 ºC and pH 2.0 – 10 and hence exhibited high stability. The MPTI was inhibited both trypsin and chymotrypsin showing the double headed nature. Antifungal studies showed inhibitory activity of MPTI against Aspergillus niger and Trichoderma viridae.
KEYWORDS:Mucuna pruriens;trypsin inhibitor;purification;characterization;antifungal Properties
Download this article as:| Copy the following to cite this article: Chandrashekharaiah K. S. Physico-Chemical and Antifungal Properties of Trypsin Inhibitor from the Seeds of Mucuna Pruriens. Orient J Chem 2013;29(3). http://dx.doi.org/10.13005/ojc/290329 |
| Copy the following to cite this URL: Chandrashekharaiah K. S. Physico-Chemical and Antifungal Properties of Trypsin Inhibitor from the Seeds of Mucuna Pruriens. Orient J Chem 2013;29(3). Available from: http://www.orientjchem.org/?p=319 |
Introduction
Protease inhibitors (PIs) are proteins or peptides capable of inhibiting the catalytic activity of proteolytic enzymes and are widely distributed in plants, animals and microorganisms. Plants are the most abundant sources of PIs of which most of them studied and characterized were serine protease inhibitors (Rao and Suresh, 2007). These PIs are concentrated in seeds and tubers of plants belonging to Gramineae, Leguminosae and solanaceae families (Connors et al., 2002). Among the seed legumes, two major families of PIs, Bowman – Birk inhibitors (BBI) and Kunitz type inhibitors (KTI) have been studied extensively (Lingaraju and Gowda, 2008). A Bowman – Birk inhibitor (BBI) was first isolated from soybeans by Bowman and its biochemical properties were studied by Birk (Birk, 1985). Subsequently, many BBIs have been isolated and characterized from legumes (only Fabaceae family), gramineae and many other plants (Ikenaka and Norioka, 1985).
PIs play essentials roles in biological system including the blood coagulation system, compliment cascade, apoptosis, cell cycle and hormone processing pathways (Lingaraju and Gowda, 2008). They are also involved in the treatment of human pathologies such as inflammation, hemorrhage (Oliva et al., 2000) and cancer (Kennedy, 1998). Plant seeds are rich sources of proteinacious protease inhibitors, which impair the nutritional quality by reducing protein digestibility and absorption (Liener and Kakade, 1980). Plant PIs also play essential role in the regulation of endogenous proteinases and involved in defense mechanisms against insects, fungi and other pathogenic microorganisms (Valueva and Mosolov, 1999; Carlini and Grossi-de-sa, 2002; Kim et al., 2005; Breiteneder and Radauer, 2004). Interest in understanding the physiological importance of PIs has increased due to their involvement in regulation of many biological processes that involved preoteolytic enzymes such as intracellular protein breakdown, transcription, cell cycle, cell invasion (Kataoka, Itoh & Koono, 2002) and apoptosis (Thompson & Palmer, 1998; Fumagalli et al., 1996; Kato, 1999). More recent studies indicated that PIs have been employed as new drugs in highly active antiretroviral combination therapy (HAART), increasing life expectancy in HIV-positive patients (Asztalos et al., 2006; Yeni, 2006; Lopes et al., 2009).
The genus Mucuna belongs to the family Fabaceae (Leguminoceae) which contains up to 150 species of annual and perennial legumes of pan tropical distribution. Mucuna is extensively used as cover crop to control insects and weeds in agriculture. Mucuna pods are covered with reddish-orange hairs, which readily dislodge and cause intense skin irritation and itch due to presence of a chemical called Mucunain. Many varieties and accessions of the wild legume, Mucuna are in great demand in food and pharmaceutical industries. The nutritional importance of Mucuna seeds as a rich source of protein supplement in food and feed has been well documented (Siddhuraju, Becker and Makkar, 2000; Siddhuraju and Becker, 2001; Bressani, 2002, Chandrashekharaiah, Ramachandra Swamy and Siddalinga Murthy, 2011). It is used in Ayurvedic medicine. The plant and its extracts have been long used in tribal communities as a toxin antagonist for various snakebites. Research on its effects against Naja spp. (cobra) (Tan et al., 2009), Echis (Saw scaled viper) (Guerranti, 1999), Calloselasma (Malayan Pit viper) and Bangarus (Krait) (Meenatchisundaram and Michael, 2010) have shown it has potential use in the prophylactic treatment of snakebites. M. pruriens seeds have also been found to have antidepressant properties in cases of depressive neurosis when consumed (Daniel, 2006) and formulations of the seed powder have shown promise in the management and treatment of Parkinson’s disease (Katzenschlager et al., 2004). In view of the importance of PIs and seeds of Mucuna being a potential source of PIs, the present investigation was undertaken to study the biological properties of protease inhibitors form the soaked seeds of Mucuna pruriens. In the present study, purification, characterization and properties of trypsin inhibitor isolated from the soaked seeds Mucuna pruriens has been described.
Materials and Methods
Materials
Seeds of Mucuna pruriens were collected from Siddarabetta, Tumkur District and Thorekempohalli, Nelamangala Taluk, Bangalore Rural District, Karnataka, India. Trypsin, Chymotrypsin, N-benzoyl-L-arginine pnitronilide (BAPNA), N-acetyl-DL-phenylalanine α naphtyl ester (APNE), BSA, ampholites (pH 3 – 10), acrylamide and bis-acrylamide were obtained from Sigma-Aldrich chemical company, St. Louis, USA and all other chemicals used were of analytical grade.
Preparation of crude trypsin inhibitor extract
Mucuna pruriens seeds were soaked overnight in distilled water and crushed with a mortar and pestle using 100 ml (10% w/v) of chilled 0.05 M Glycine – HCl buffer, pH 3. The homogenates were filtered using muslin cloth and the filtrates were centrifuged at 10,000 rpm for 30 min at 4 °C. The supernatants were collected.
Trypsin Activity
Trypsin assay was performed according to the reported method of Shibata et al., (1986) using BAPNA as substrate. Trypsin was dissolved in 0.001 N HCl containing 20 mM CaCl2 at a concentration 200 μg per ml. The assay mixture containing 100 μl of the trypsin solution, 900 μl of 0.1 M Tris Hydrochloride buffer, pH 8 and 1ml of 5 mM BAPNA (BAPNA was dissolved in 2.5% DMSO and volume was made up to appropriate using 0.05 M Tris Hydrochloride buffer, pH 8). The reaction was terminated after 10 min using 30% acetic acid. The color developed was read at 410 nm against reagent blank. Trypsin units: 1 Trypsin unit (TU) is defined as increase in the OD of 0.01 at 410 nm.
Typsin Inhibitor Activity
Trypsin inhibitor assay was performed according to the reported method of Shibata et al., (1986) using BAPNA as substrate by estimating the remaining hydrolytic activity of trypsin. The assay mixture containing 100 μl of trypsin solution, 400 μl of 0.1 M Tris Hydrochloride buffer, pH 8 and 0.5 ml of appropriately diluted inhibitor extract was incubated for 10 min at room temperature. 1ml of 5 mM BAPNA solution was added. The reaction was terminated after 10 min using 30% acetic acid. The color developed was read at 410 nm against reagent blank. Trypsin inhibitor units: One Trypsin inhibitor unit (TIU) is defined as decrease in the OD by 0.01 at 410 nm. Protein concentration was determined according to the method of Lowry et al. (1951), using bovine serum albumin (BSA) as standard. The protein content in the eluents obtained from chromatographic columns was routinely monitored by measuring absorbance at 280 nm.
Chymotrypsin assay: The chymotrypsin activity was determined using casein as the substrate according to the method of Kakade et al. (1969a). Twenty four µg of chymotrypsin was taken in 2.0 ml of sodium phosphate buffer, pH 7.6 containing 0.15 M NaCl. The reaction was initiated by the addition of 2.0 ml of 2% casein at 37oC. The reaction was stopped after 20 minutes by the addition of 6% trichloroacetic acid (6.0 ml) and after standing for 1 hr, the suspension was filtered through whatman no. 1 filter paper. Absorbance of the filtrate was measured at 275 nm using spectrophotometer. One (chymotrypsin unit is arbitrarily defined as an increase in absorbance by 0.01 275 nm under conditions of assay.
Chymotrypsin inhibitor assay: The chymotrypsin inhibitor activity was determined using casein as the substrate according to the method of Kakade et al. (1969b). Enzyme solution 24 µg of chymotrypsin was preincubated with known aliquots of the inhibitor extract at 37oC for 10 min in 0.01 M sodium phosphate buffer, pH 7.6, containing 0.15 M NaCl. The residual enzyme activity was determined as described above.
Polyacrylamide gel electrophoresis (PAGE)
Non-denaturing PAGE (7.5% T, 2.7% C) was performed at pH 4.3 according to the procedure of Reisfield et al. (1962). The slab gel (7.5% separating gel and 4% spacer gel) was prepared and gel was placed into the electrophoretic chamber. The electrode chambers were filled with electrode buffer of pH 4.5 (3.12 g β alanine + 0.8 ml glacial acetic acid diluted to 600 ml with distilled water). The samples suitably diluted with 20 % sucrose containing methyl green were loaded onto each sample well and subjected to electrophoresis in cold (4 ºC) applying a current of 20-25 mA for 3 hr. SDS–PAGE (10% T, 2.7% C) was performed after denaturing the proteins with SDS and β-mercaptoethanol. Gel-electrofocussing was performed by the method of Wrigley (1969), in 8% polyacrylamide gels. The electrophoresis was performed at 4 0C for 2 h. After the run, the gels were removed and stained for esterase activity as described earlier. The gels were stained for proteins using 0.02% coomassie brilliant blue G-250 (w/v) in 3.5% (w/v) perchloric acid and destained in distilled water.
Gel localization of Trypsin Inhibitors
Visualization of trypsin and chymotrypsin inhibitor in polyacrylamide gel was performed according to Filho and Moriera (1978). After electrophoresis, native gel was incubated in 0.1 M Sodium Phosphate buffer, pH 8 containing Trypsin (40 μg/ml) for 1 hr and visualized with APNE (1 mg/ml) in DMSO and Fast blue B salt (7 mg/10 ml). The appearance of clear transparent bands against pink background indicates the presence of Trypsin inhibitor. Proteins were stained on polyacrylamide gels using 0.5 % solution of coomassie brilliant blue R-250 in 25 % methanol and 7.5 % acetic acid in water for 1 hr. The gel then was destained in 25 % methanol and 7.5 % acetic acid in water overnight. The gels were stored in 7.5 % acetic acid.
Purification
All the purification procedures were performed at 4 0C unless otherwise stated. To the crude trypsin inhibitor extract, solid ammonium sulphate was added to 0–80% saturation at 4 0C. The precipitate obtained was removed by centrifugation at 10,000 rpm for 30 min. The precipitate thus obtained was redissolved in 0.025 M sodium acetate buffer, pH 5.0 and dialyzed against the same buffer. The dialyzed fraction was loaded onto a CM-cellulose column (2.5 x 22 cm) pre-equilibrated in 0.025 M sodium acetate buffer, pH 5.0 at a flow rate of 30 ml/h. The bound proteins were eluted by stepwise increase in ionic strength using start buffer containing 0.1 and 0.3 M NaCl with a fraction volume of 10 ml. The CM-cellulose fraction III containing trypsin inhibitor activity were pooled, concentrated and applied to a Sephadex G-100 column (1.0 x 140 cm) pre-equilibrated with 0.025 M sodium phosphate buffer pH 7.0. The proteins were eluted with the same buffer and fractions of 2.0 ml were collected at a flow rate of 12 ml/h. The trypsin inhibitor activity was eluted in a single peak.
Molecular weight determination
The apparent molecular mass of the native enzymes were determined according to the method of Andrews (1970) using Sephadex G-100 (1.13 x 100 cm) pre-equilibrated with 0.025 M sodium phosphate buffer pH 7.0, at a flow rate of 10 ml/h. The column was calibrated using cytochrome-c (12.3 kDa), carbonic anhydrase (29 kDa), bovine serum albumin (BSA) (66 kDa), alcohol dehydrogenase (150 kDa) and b-amylase (200 kDa). Blue Dextran (2000 kDa) was used to determine the void volume (Vo). The molecular weight of the trypsin inhibitor was determined from the plot of log molecular weight versus Kav. The molecular weight of the purified trypsin inhibitor was also determined by SDS–PAGE from the plot of log molecular weight against relative mobility.
Effect of pH and temperature
Determination of pH stability: The effect of pH on the activity of the purified Mucuna pruriens trypsin inhibitor was studied using the buffers, Glycine – HCl buffer (0.2 M, pH 2), Sodium acetate buffer (0.2 M, pH 4), Sodium citrate buffer (0.2 M, pH 5.5), Sodium phosphate buffer (0.2 M, pH 6.5), Tris – hydrochloride buffer (0.2 M, pH 8.0), Sodium borate buffer (0.2 M, pH 10). The pH stability was determined by pre-incubating the purified Mucuna pruriens trypsin inhibitor (MPTI) with above buffers for 30 min. Trypsin inhibitor assay was performed as described earlier.
Determination of temperature stability: The effect of temperature on the activity MPTI was studied at different temperatures ranging between 0 – 90 ºC. The temperature stability of purified MPTI was studied by pre-incubating, the purified MPTI at different temperatures (0 – 90 ºC) for 30 min. The incubated samples were rapidly cooled and assayed at room temperature. Trypsin inhibitor assay was performed as described earlier.
Determination of IC50: The inhibitory activity of purified Mucuna pruriens trypsin inhibitor against trypsin was determined using increasing concentrations of MPTI. Trypsin inhibitor assay was performed as described earlier and a graph of percentage residual trypsin activity versus concentration of MPTI (nM) was plotted and the IC50 value was determined.
Determination of anti-fungal activity: Potato Dextrose Agar (infusion from 200 g of potatoes + 20 g dextrose and 15 g agar in 1000 ml of distilled water) was prepared. The solution was heated to boiling to dissolve the medium completely. The media and glass wares were sterilized by autoclaving at 121 ºC at 15 psi for 15 min. The molten media was poured into two sterilized petriplates. The inoculums of Trichoderma viridae and Aspergillus niger were prepared by serial dilution. 0.1 ml of the 10-4 dilution sample was spread-plated over the solidified agar under aseptic conditions. The purified Mucuna pruriens trypsin inhibitor discs were prepared by adding 0.1 ml of the enzyme drop-wise onto a 2 cm Whatman filter paper disc and allowing to air dry. The discs were placed on the centre of the inoculated petriplate and incubated at room temperature for 3days. The diameters of the inhibition zones were measured.
Results and discussions
Protease inhibitors were purified and characterized from different sources including plants, animals, and microorganisms. There is a growing interest in the identification of novel protease inhibitors because of their involvement in many biological processes including their potent activity in preventing carcinogenesis both in vivo and in vitro systems and their use in developing pest resistance in otherwise susceptible plants (Kennedy, 1998; Jouanin et al. 1998; Schuler et al. 1998). In the present study, we described the isolation, purification and characterization of novel trypsin inhibitor from the seeds of Mucuna pruriens. MPTI was purified to homogeneity using conventional protein purification methods such as ammonium sulphate fractionation, ion exchange chromatography using CM-cellulose and gel-permeation on Sephadex G-100. The ammonium sulphate fractionation resulted with a specific activity of 8.025 and yield of 77.35 %. The ammonium sulphate fraction was subjected to ion exchange chromatography on CM Cellulose and resulted in the elution of three peaks of inhibitor activities. The elution profile of CM Cellulose chromatography is shown in the Fig.1a. Three peaks of trypsin inhibitor activities eluted were designated as fraction I, fraction II and fraction III. Fraction I was not adsorbed onto the column and eluted with the starting buffer. Fraction II was eluted by 0.1 M NaCl and fraction III by 0.3 M NaCl in starting buffer. The CM Cellulose fraction III containing appreciable level of inhibitory activity was pooled separately and dialyzed against double distilled water and then concentrated using ammonium sulphate. The concentrated CM Cellulose fraction III was subjected to gel filtration on Sephadex G-00 column. The elution profile of Sephadex G-75 chromatography is as shown in the Fig. 1b. The trypsin inhibitor activity was eluted in a single peak. The specific activity of the crude trypsin inhibitor extract towards bovine trypsin was 4.77 and 474.66 was obtained for purified trypsin inhibitor. Similarly, the fold purification of purified trypsin inhibitor after gel-filtration chromatography was 99.51 and yield obtained was 22.08% (Table 1).
 |
FIG. 1. (a) Elution profile of Trypsin inhibitor from the soaked seeds of Mucuna pruriens on CM- Cellulose using 0.025 M sodium acetate buffer pH 5.0 Click here to View figure |
 |
FIG. 1. (b) Elution profile of CM-cellulose fraction-II from the soaked seeds of Mucuna pruriens on Sephadex G-100 (1.13 x 100 cm). Click here to View figure |
Criteria of homogeneity
The homogeneity of the purified trypsin inhibitor was established by native PAGE (Fig. 2a), Isoelectric focusing (Fig. 2b) and SDS PAGE (Fig. 2c). Native PAGE of the peak fraction of sephadex G-100 showed single trypsin inhibitor (Fig. 2a (i)) corresponding to a single protein band (Fig.2a (ii)). IEF of MPTI showed a single trypsin inhibitor corresponding to a single protein band. SDS–PAGE in the presence and absence of β-mercaptoethanol showed single protein bands, suggesting the monomeric nature of the Mucuna pruriens trypsin inhibitor.
 |
FIG. 2. (a) Native PAGE pattern of Mucuna pruriens trypsin inhibitor (i) and protein (ii)a – Crude, b- ammonium sulphate fractuion, c- ion exchange fraction, d – Gel filtration fraction(b) IEF of purified Mucuna pruriens trypsin inhibitor (i) Trypsin inhibitory activity (ii) Protein(c) SDS – PAGE pattern of (M) standard proteins (1) purified Mucuna pruriens trypsin inhibitor in the presence and (2) absence of β-mercaptoethanol. Click here to View figure |
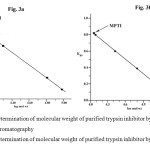 |
FIG. 3. (a) Determination of molecular weight of purified trypsin inhibitor by gel filtration chromatography (b) Determination of molecular weight of purified trypsin inhibitor by SDS – PAGE: Click here to View figure |
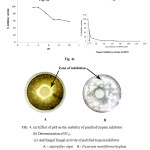 |
FIG. 4. (a) Effect of pH on the stability of purified trypsin inhibitor (b) Determination of IC50 (c) Antifungal fungal activity of purified trypsin inhibitor A – Aspergillus niger B – Fusarium moniliforme hyphae Click here to View table |
Molecular weight
The molecular weight of MPTI as determined by SDS–PAGE was found to be 11.6 kDa both in the presence and absence of β-mercaptoethanol. This clearly indicates the monomeric nature of MPTI. The monomeric nature of the purified Mucuna pruriens trypsin inhibitor (MPTI) was further confirmed by gel-filtration on Sephadex G-100 which indicated molecular weight of 12 kDa. A Bowman-Birk proteinase inhibitor from the seeds of Vigna mungo purified and characterized by Prasad et al. (2010) had a molecular weight of 8 kDa on an 18 % gel under non-reducing conditions. Scarafoni et al. (2008) purified a Bowman-Birk trypsin inhibitor from Lupinus albus seeds which consisted of a single polypeptide chain having a molecular weight of 6 kDa. Torres Castillo et al. (2009) purified and characterized a highly stable trypsin like proteinase inhibitor from the seeds of Opuntia sterptacantha which showed a low molecular mass of 6.2 kDa. The results for MPTI is also comparable with protease inhibitor from Lupinus bogotensis seeds purified and characterized by Diana Molina et al. (2010) which was a single polypeptide chain having a molecular weight of 12 kDa. Generally, the Bowman – Birk type inhibitor has a lower molecular weight compared with Kunitz type. The Kunitz type inhibitors have a molecular weight of >20 kDa with low cysteine content and a single reactive site, whereas the Bowman – Birk type inhibitors have a molecular weight of 8 – 10 kDa , as well as a high cysteine content and two reactive sites (Richardson, 1977). Two protease inhibitors of Bowman – Birk type with molecular weights of 15,000 and 10,500 kDa were found in pigeon pea (Godbole et al., 1994). From the result, Mucuna pruriens trypsin inhibitor belongs to Bowman – Birk type. However, the amino acid sequence is needed to verify the classification of this inhibitor.
Isoelectric Point
The isolelectric pH of purified MPTI was 5.6±0.05 and it showed binding affinity to a cation exchanger at pH 5.0. Maria Ligia R Macedo et al. (2000) purified and characterized a trypsin inhibitor form Dimorphandra mollis seeds. The pI values the three isoforms were found to be 5.6, 5.8 and 5.9, comparable to that of MPTI. A highly stable trypsin-like proteinase inhibitor purified and characterized by Torres Castillo et al. (2009) from the seeds of Opuntia sterptacantha, possessed acidic pI value of 4.5. Bowman-Birk proteinase inhibitors with pI values of 4.3, 4.4, 5.0, 5.3 and 6.0 were purified and characterized by Prasad et al. (2010) from the seeds of Vigna mungo.
Effect of pH and temperature
The activity of MPTI was found to be stable in the pH range 2 – 10. The inhibitory activity of MPTI was found to be almost same in temperatures ranging from 0-90 ºC. The inhibitory activity was found to be 76.77 % at 0 ºC and 73.42 % at 90 ºC. Scarafoni et al. (2008) purified a Bowman-Birk trypsin inhibitor from Lupinus albus seeds which showed no significant change in the inhibitory activity after subjecting to various pH and temperature conditions. Trypsin inhibitor from Thai mung bean was also stable between pH 2 – 10. The thermal stability of MPTI is comparable to that of LaBBI. . Torres Castillo et al. (2009) purified and characterized a highly stable trypsin-like proteinase inhibitor from the seeds of Opuntia sterptacantha which retained inhibitory activity at a temperature of 120 ºC and pH ranging from 3-7. Thermal stability of MPTI was comparable with trypsin inhibitor isolated from Thai mung bean which was stable between 0 – 100 0C (Sappasith Klomklao et al., 2011).
Half-Maximal Inhibitor Concentration
A graph of percentage residual trypsin activity versus concentration of MPTI (nM) was plotted and the IC50 value was found to be 3.6 ± 0.1 nM. A Kunitz trypsin inhibitor from Entada scandens seeds purified by Lingaraju et al. (2008) was obseverd to have an IC50 value of 23.4 ± 0.001 nM which is higher when compared to that of MPTI.
Anti-Fungal Activity
Anti-fungal activity of MPTI was tested on two fungi: Aspergillus niger and Trichoderma viridae, using the disc method as described earlier. An inhibition zone was observed around both discs, each containing 25 MPTI units. The normal growth and development was suppressed by MPTI. Antifungal activity of protease inhibitors isolated from Acacia plumose was reported by Lopes et al., (2009) and they observed antifungal activity of ApTI against Aspergillus niger and Fusarium moniliforme hyphae.
Conclusion
The identification, purification and characterization of a low molecular weight trypsin inhibitor (MPTI) from seeds of Mucuna pruriens has been successfully carried out using ammonium sulphate fractionation, ion exchange chromatography and gel-filtration chromatography. This inhibitor is an acidic, 11 – 12 kDa protein. Its low molecular mass is similar to other trypsin inhibitors previously characterized from plant sources. This inhibitor showed high resistance to heat denaturation and extremes of pH. Hence it is thermo and pH stable inhibitor. Its inhibition towards both bovine trypsin and chymotrypsin and low molecular weight suggested that it is BBI type protease inhibitor. Its biological properties such as antifungal activity suggested that it is a potent agent to control unwanted proteolyic processes and as a biopesticide or insecticide in transgenic commercial and food plants
ACKNOWLEDGEMENTS
The authors wishes to gratefully acknowledge, Dr. K.N.B. Murthy, Principal, PES Institute of technology, Prof. M R Doreswamy, Founder secretary, PES group of Institutions, Bangalore and Prof. D Jawahar, CEO, PES group of Institutions, Bangalore, Karnataka, India for providing research facilities to carry out the present work.
References
- Andrews, P. (1970). Estimation of molecular size and molecular weight of biological compounds by gel filtration. Methods Biochem. Anal., 18, 1-53.
- Asztalos Bela, F., Ernst Schaefer, J., Katalin Horvath, V., Caitlin Cox, E., Sally Skinner, Jul Gerrior, Sherwood Gorbach, L., Christine Wanke, (2006). Protease inhibitor-based HAART, HDL, and CHD-risk in HIV-infected patients. Atherosclerosis, 184(1), 72- 77.
- Birk, Y. (19850. The Bowman – Birk inhibitor. Trypsin and chymotrypsin inhibitor from soybeans. Int. J. Pept. Protein Res, 25, 113 – 131.
- Breiteneder, H., Radauer, C. (2004). A classification of plant food allergens. J. Allergy Cln. Immun, 113(5), 821 – 830.
- Bressani, R. (2002). Factors influencing nutritive value in food grain legumes: Mucuna compared to other grain legumes. In: Food and Feed from Mucuna: Current Uses and the Way Forward (Editors, Flores B M, Eilittä M, Myhrman R, Carew L B and Carsky R J), Workshop, CIDICCO, CIEPCA and World Hunger Research Center, Tegucigalpa, Honduras, 164 -188.
- Carlini, C.R., Grossi – de – sa, M.F. (2002). Plant toxic proteins with insecticidal properties. A review on their potentialities as bioinsecticides. Toxicon, 40, 1515 – 1539.
- Chandrashekharaiah, K.S., Ramachandra Swamy, N.,. Siddalinga Murthy, K.R. (2011). Carboxylesterases from the seeds of an underutilized legume, Mucunapruriens; isolation, purification and characterization. Phytochemistry, 72, 2267 – 2274.
- Connors, B.J., Laun, N.P., Maynard, C.A., Powell, W.A. (2002). Molecular characterization of gene encoding a cystatin expressed in the stems of American chestnut (Castanea dentate). Planta, 215, 510 – 514.
- Daniel, M. (2006). Medicinal Plants: Chemistry and Properties. Science publishers, 5 – 23.
- De Leo, F., Volpicella, M., Licciulli, F., Liuni, S., Gallerani, R., Ceci, L.R. (2002). PLANT-PIs: a database for plant protease inhibitors and their genes. Nucleic Acids Res, 30 (1): 347-348.
- Diana Molina, A., Humberto Zamora, B., Alejandro Blanco-Labra. (2010). An inhibitor from Lupinus bogotensis seeds effective against aspartic proteases from Hypothenemus hampei. Phytochemistry, 71, 923–929.
- Filho, J.X., Moreira, R.A. (1978). Visualization of proteinase inhibitors in SDS-Polyacrylamide gels. Anal. Biochem, 84, 296-303.
- Godbole, S.A., Krishna, T.C., & Bhatia, C.R. (1994). Purification and characterization of protease inhibitors from pigeon pea (Cajanus cajan (l) Millsp) seeds. Journal of the Science of food and Agriculture, 64, 87 – 93.
- Guerranti, R., Aguiyi, J.C., Leoncini, R., Pagani, R., Cinci, G., Marinello, E. (1999). Characterization of the factor responsible for the antisnake activity of Mucuna Pruriens seeds. Journal of Preventie Medicine and Hygiene, 40, 25–28.
- Ikenaka, T., Norioka, S. (1983). Bowman – Birk family serine proteinase inhibitors, in: Barrett, A.J., Salvenson, S., (Eds.), Proteinase inhibitors, Elsevier, Amsterdam, 361 – 374.
- Jouanin, L., Bonade´-Bottino, M., Girard, C., Morrot, G., Giband, M. (1998). Transgenic plants for insect resistance. Plant Sci, 131, 1–11.
- Kakade, M. L., Simons, N. R. & Liener, I. E. (1969a). The evaluation of natural vs. synthetic substrates for measuring the antitryptic activity of soybean samples. Cereal Chem., 46: 518-526.
- Kakade, M. L., Simons, N.R. & Liener, I. E. (1969b). The molecular weight of Bowman and Birk soybean protease inhibitor. Biochim. Biophys. Acta, 200: 168 – 169.
- Kataoka, H., Itoh, H., Koono, M. (2002). Emerging multifunctional aspects of cellular serine proteinase inhibitors in tumor progression and tissue regeneration. Pathol. Int, 52, 89-102.
- Kato, G.J. (1999). Human genetic diseases of proteolysis. Hum Mutat.13(2), 87-98.
- Katzenschlager, R., Evans, A., Manson, A., Patsalos, P.N., Ratnaraj, N., Watt, H.,Timmermann, L., Van Der Giessen, R., et al. (2004). Mucuna pruriens in parkinson’s disease: a double blind clinical and pharmacological study. Journal of Neurology, Neurosurgery & Psychiatry, 75 (12), 1672–1677.
- Kennedy, R. C. (1998). The Bowman-Birk inhibitor from soybeans as an anticarcinogenic agent. Am. J. Clin. Nutr, 68, 1406 -1412.
- Kim, J.Y., Park, S.C., Kim, M.H., lim, H.T., Park, Y., Hahm, K.S. (2005). Antimicrobial activity studies on a trypsin – chymotrypsin protease inhibitor obtained from potato. Biochem. Bioph. Res. Co, 330 (3), 921 – 927.
- Liener, I. E., Kakade, M. L. (1980). Proteinase inhibitors. In “ The toxic constituents of plant food stuffs”. (I.E. Liener, ed.), 2nd Edn.: 7-71, Acad. Press, New York.
- Lingaraju, M.H., Gowda, L.R. (2008). A Kunitz trypsin inhibitor of Entada scandens seeds: another member with single disulfide bridge. , 1784(5), 850-855.
- Lopes, J. L. S., Valadares, N. F., Moraes, D. I., Rosa, J. C., Araujo, H. S. S., & Beltramini, L. M. (2009). Physico-chemical and antifungal properties of protease inhibitors from Acacia plumose. Phytochemistry, 70, 871-879.
- Fumagalli, L., Rita Businaro; Stefania L Nori, Toesca, A., Pompili, E., Evangelisti, E., Giannetti, S., Flora Ippoliti, De Renzis, G. (1996). Protease inhibitors in mouse skeletal muscle: tissue-associated components of serum inhibitors and calpastatin. Cellular and molecular biology (Noisy-le-Grand, France), 42(4), 535-546.
- Lowry, O. H., Rosebrough, N. J., Farr, A. L., Randall, R. J. (1951). Protein measurement with the folin-phenol reagent. J. Biol. Chem, 193(1), 265 – 275.
- Maria Lı́gia R Macedo, Daniela Gaspar G De Matos, Olga L T Machado, Sérgio
Marangoni, José C Novello, (2000). Trypsin inhibitor from Dimorphandra mollis seeds: purification and properties. Phytochemistry, 54 (6), 553-558. - Meenatchisundaram, S., Michael, A. (2010). Antitoxin activity of Mucuna pruriens aqueous extracts against Cobra and Krait venom by in vivo and in vitro methods. International Journal of PharmTech Research, 2(1), 870 – 874.
- Oliva, M. L. V., Souza-Pinto, J. C., Batista, I. F., Araujo, M. S., Silveria, V. F., Auerswald, E. A., Menrele, R., Eckerskorn, C., Sampaio, M. U., & Sampaio, C. A. M. (2000). Leucena leucocephala serine protease performance and feed utilization. Aquaculture, 196, 105-123.
- Prasad, E.R., Dutta-Gupta, A., Padmasree, K. (2010). Purification and characterization of a Bowman-Birk proteinase inhibitor from the seeds of black gram (Vigna mungo). Phytochemistry, 71(4), 363 – 372.
- Rao, K.N., Suresh, C.G. (2007). Bowman – Birk protease inhibitor from the seeds of Vigna unguiculata forms a highly stable dimeric structure. Biochimica et Biophysica Acta, 1774, 1264 – 1273.
- Reisfield, R. A., Lewis,V. J., Williams, D. E. (1962). Disc electrophoresis of basic proteins and peptides on polyacrylamide gels. Nature, 195, 281-283.
- Richardson, M. (1977). The proteinase inhibitors of plants and microorganisms. Phytochemistry, 16, 159 – 169.
- Sappasith Klomklao, Soottawat Benzakul, Hideki Kishimura, Manat Chaijan, (2011). Extraction, purification and properties of trypsin inhibitor from Thai mung bean (Vigna radiate (L.) R. Wilczek). Food Chemistry, 129, 1348 – 1354.
- Scarafoni, Alessio · Consonni, Alessandro · Galbusera, Valerio · Negri, Armando · Tedeschi, Gabriella · Rasmussen, Patrizia · Magni, Chiara · Duranti, Marcello, (2008). Identification and characterization of a Bowman-Birk inhibitor active towards trypsin but not chymotrypsin in Lupinus albus seeds. Phytochemistry, 69(9), 1820-1825.
- Schuler, H., T., Poppy, M. G., Denholm. L. (1998). Insect-resistant transgenic plants. Trends Biotechnol, 16,168–174.
- Shibata, H., Hara, S., Ikenaka, T., Abe, J. (1986). Purification and characterization of protease inhibitors from winged bean (Psophocarpus tetragonolobus (L.) DC.) seeds. Journal of Biochemistry, 99, 1147 – 1155.
- Siddhuraju, P., Becker, K. (2001a). Preliminary evaluation of Mucuna seed meal (Mucuna pruriens var. utilis) in common carp (Cyprinus carpio L.): An assessment by growth performance and feed utilization. Aquaculture, 196, 105-123.
- Siddhuraju, P., Becker, K., Makkar, H. P. S. (2000). Studies on the nutritional composition and antinutritional factors of three different germplasm seed materials of an underutilized tropical legume. Mucuna pruriens var. utilis. Journal of Agricultural Food Chemistry, 48, 6048-6060.
- Smith, B. J. (1984). SDS Polyacrylamide gel electrophoresis of proteins. In: Walker, J. M., ed. Method in Molecular Biology, 1, Clifton: Humana Press, 41-56.
- Tan, N.H., Fung, S.Y., Sim, S.M., Marinello, E., Guerranti, R., Aguiyi, J.C. (2009). The protective effect of Mucuna pruriens seeds against snake venom poisoning. Journal of Ethnopharmacology, 123 (2), 356–358.
- Thompson, M.G., Palmer, R.M. (1998). Signalling pathways regulating protein turnover in skeletal muscle. , 10(1), 1-11.
- Torres- Castillo, J. A., Mondragon Jacobom C., Blanco Labram A. (2009). Characterization of a highly stable trypsin-like proteinase inhibitor from the seeds of Opuntia streptacantha (O. streptacantha Lemaire). Phytochemistry, 70, 1374-1381.
- Valueva, T. A., Mosolov, V. V. (1999). Protein inhibitors of proteinases in seeds: Classification, distribution, structure and properties. Russ. J. Plant Physiol, 46, 307–321.
- Wrigley, C. W. (1969). Analytical Fractionation of Proteins According to Isoelectric Point by Gel electrofocussing. Instrument applications no.29, London: Shandon Scientific Co.
- Yeni, P. (2006). Update on HAART in HIV. J. Hepatol, 44 (1), 100 – 103.

This work is licensed under a Creative Commons Attribution 4.0 International License.









