Aromatization of 1,4-Dihydropyridines by Hydrogen Peroxide in the Presence of Nano-Fe2O3 at Room Temperature
Farhad Hatamjafari* and Ahmad Hosseinian
Department of Chemistry, Faculty of Science, Islamic Azad University-Tonekabon Branch, Tonekabon, Iran.
Article Received on :
Article Accepted on :
Article Published : 19 Oct 2016
Hydrogen peroxide readily oxidizes 1, 4-dihydropyridine derivatives in high yields in the presence of nano-Fe2O3 as catalyst at room temperature
KEYWORDS:Nano-Fe2O3; 1; 4-dihydropyridines; Hydrogen peroxide; Aromatization
Download this article as:| Copy the following to cite this article: Hatamjafari F, Hosseinian A. Aromatization of 1,4-Dihydropyridines by Hydrogen Peroxide in the Presence of Nano-Fe2O3 at Room Temperature. Orient J Chem 2013;29(1). |
| Copy the following to cite this URL: Hatamjafari F, Hosseinian A. Aromatization of 1,4-Dihydropyridines by Hydrogen Peroxide in the Presence of Nano-Fe2O3 at Room Temperature. Available from: http://www.orientjchem.org/?p=22568 |
Introduction
Chemistry of 1,4-dihydropyridine in 1882 with Arthur Hantzsch reports the ᵝ-keto ester, aldehydes and ammonia can be combined to form 1,4-Dihydropyridine is was developed. 1, 4-Dihydropyridine ring is the common feature for various pharmacological activities such as antagonists1, antidiabetics2, calcium antagonists3, antivirals4, and antitumours5. Aromatization of 1,4-dihydropyridines (1,4-DHPs) to pyridine derivatives the principal metabolic in biologically activity NADH redox processes6, an efficient route for synthesis of pyridine derivatives are oxidation of 1,4-DHPs7. Many of the reported oxidation procedures either suffer from chromium (VI) oxidants8, CrO29, SnCl410, Pb(OAc)411, K2S2O812, an so on.
Previously, we have synthesized a number of heterocyclic compounds13-18. Although many methods are capable of effecting these oxidations but most of the reported are difficult such as separate from the products, long reaction times and low yields. Therefore, we reported the development of an efficient, a facile method for the aromatization of 1, 4-DHPs by hydrogen peroxide in the presence of nano-Fe2O3 as catalyst at room temperature (Scheme 1). The hydrogen peroxide was selected as the oxidant19-20 and nano-Fe2O3 as the catalyst was cheap, environmentally friendly, simple route and easy separation at room temperature.
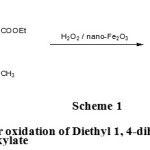 |
Scheme 1: General procedure for oxidation of Diethyl 1, 4-dihydro-2,6-dimethyl-4-phenylpyridine-3,5-dicarboxylate. |
 
Diethyl 1,4-dihydro-2,6-dimethyl-4-phenylpyridine-3,5-dicarboxylate (1 mmol) was dissolved in 10 ml acetonitrile and nano-Fe2O3 (10 mol%) was added to this solution and then 0.3 ml hydrogen peroxide 30% was added in portions over 30 min at room temperature along with stirring of the reaction mixture. The progress of the reaction was monitored by TLC. After recrystallization of the product in ethanol, 96% yield of diethyl 2, 6-dimethyl-4-phenylpyridine-3, 5-dicarboxylate was obtained.
Spectral data for diethyl 2,6-dimethyl-4-phenylpyridine-3,5-dicarboxylate
Pale yellow solid; m.p.: 60-62 ºC (Ref. [19], 60–61 ºC); FT-IR (KBr): 2988, 1718, 1575, 1212, 1145 cm-1; 1H NMR (CDCl3): δ (ppm) = 1.12 (t, 6H, J = 6.9 Hz), 2.48 (s, 6H), 4.09 (q, 4H, J = 6.9 Hz), 7.25-7.6 (m, 5H).
Conclusions
In research we report oxidation of 1,4-DHPs by hydrogen peroxide and nano-Fe2O3 as catalyst was used to pyridine derivatives (Scheme 1), which could provide an efficient, cheap, high yield, environmentally friendly, easy separation and simple route at room temperature for the oxidation of 1,4-DHPs to pyridine derivatives.
Acknowledgements
We gratefully acknowledge the financial support from the Research Council of Tonekabon Branch Islamic Azad University.
References
- Malaise W.J. and Mathias P.C.F., Diabetologia., 28: 153 (1985).
- Poindexter G.S., Bruce M.A., Breitenbucher J.G., Higgins M.A., Sit S.Y., Romine J.L., Martin S.W., Ward S.A., McGovern R.T., Clarke W., Russell J. and Antal-Zimanyi I., Bioorg. Med. Chem., 12: 507 (2004).
- Visentin S., Rolando B., Di Stilo A., Frutterro R., Novara M., Carbone E., Roussel C., Vanthuyne N. and A. Gasco, J. Med. Chem., 47: 2688 (2004).
- Krauze A., Germane S., Eberlins O., Sturms I. and Klusa V., Duburs G., Eur. J. Med. Chem., 34: 301 (1999).
- Tsuruo T., Iida H., Nojiri M., Tsukagoshi S. and Sakurai Y., Cancer Res., 43: 2905 (1983).
- Kill R.J., and Widdowson D.A., In Bioorganic Chemistry., Academic Press New York., 4: 239 (1978).
- Filipan L. M., Litvic M. and Vinkovic V., Tetrahedron., 64: 10912 (2008).
- Zolfigol M. A., Salehi P., Ghorbani-Choghamarani A., Safaiee M. and, Shahamirian M., Synth. Commun., 37: 1817 (2007).
- Ko K. J. and Kim J. Y., Tetrahedron Lett., 40: 3207 (1999).
- Jain S. M., Kant R., Dhar K. L., Singh S. and Singh G. B., Ind. J. Chem., 29B: 277 (1990).
- Litvic´ M., Cepanec I., Filipan M., Kos K., Bartolincˇic´ A., Drusˇkovic´ V., Tibi M. M. and Vinkovic´ V., Heterocycles., 65: 23 (2005).
- Memarian H. R., Mohammadpoor-Baltork I., Sadeghi M. M. and Samani Z. S., Ind. J. Chem., 40B: 727 (2001).
- Vanden E. J., Dorazio R. and Van Haverbeke Y., Tetrahedron., 50: 2479 (1994).
- Geterotsikl K., Chem Abstr., 69: 77095 (1967).
- Mashraqui S. and Karnik M., Synthesis 713 (1998).
- Vanden E. J., Mayence A, and Maquestiau A., Tetrahedron., 48: 463 (1992).
- Azizian J., Hatamjafari F., Karimi A. R. and Shaabanzadeh M., Synthesis., 5: 765 (2006).
- Azizian J., Shaabanzadeh M., Hatamjafari F. and Mohammadizadeh M.R., Arkivoc., (xi): 47 (2006).
- Hatamjafari F., Synthetic Communications., 36: 3563 (2006).
- Azizian J., Hatamjafari F. and Karimi A. R., Journal of Heterocyclic Chemistry., 43: 1349 (2006).
- Hatamjafari F. and Montazeri N., Turkish Journal of Chemistry., 33: 797 (2009).
- Hatamjafari F., Orient. J. Chem., 28: 141 (2012).
- Hashemi M. M., Ahmadibeni Y. and Ghafuri H., Monatshefte fur Chemie 134: 107 (2003).
- Ko K.Y. and Kim J.Y., Tetrahedron Lett., 40: 3207(1999).

This work is licensed under a Creative Commons Attribution 4.0 International License.









