Removal of Toxicants from Leather Industrial Wastewater using Sawdust Filter Media and Ferric Oxide (Fe2O3) Coagulant
Manjushree Chowdhury*1 , Israil Hossain1, Amal Kanti Deb1 and Tapan Kumar Biswas2
, Israil Hossain1, Amal Kanti Deb1 and Tapan Kumar Biswas2
1Institute of Leather Engineering and Technology, University of Dhaka, Dhaka-1209, Bangladesh.
2Department of Chemistry, University of Rajshahi, Rajshahi-6205, Bangladesh.
Corresponding Author E-mail: israil.rafi.du@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/350213
Article Received on : 09-02-2019
Article Accepted on : 31-03-2019
Article Published : 28 Apr 2019
The leather industrial wastewaters are a major source of water pollution among all industrial sector considering both quantity and composition. This research study was focused on the current chemical and physical parameters of tannery wastewater and treatment processes through sawdust filter media and ferric oxide (Fe2O3) coagulant. The study found that tannery wastewater contained exceptionally high values of TDS, TSS, TS, BOD, COD, SO42-, Cl-, Na, Ca, chromium, lead, cadmium and arsenic and theses parameters of the composite were exceeded the standard discharge limits which are considered as toxicants for the environment. The wastewaters were filtered by saw-dust filtration processes and then treated with different dosage of Fe2O3. The experiment elucidated that the filtration technique could minimize certain amount of toxicant load from the wastewater but not efficient enough to practise the technique alone using as wastewater treatment. The chemical interaction with 100 mg/L coagulant (Fe2O3) dosage at pH~9 showed the best result of the major parameters. The study suggested that both combined physical (filtration) and chemical (Fe2O3, coagulants) treatment processes could be fruitful to reduce the toxicants load from tannery wastewater.
KEYWORDS:Ferric Oxide; Sawdust; Tannery; Treatment; Toxicant; Wastewater
Download this article as:| Copy the following to cite this article: Chowdhury M, Hossain I, Deb A. K, Biswas T. K. Removal of Toxicants from Leather Industrial Wastewater using Sawdust Filter Media and Ferric Oxide (Fe2O3) Coagulant. Orient J Chem 2019;35(2). |
| Copy the following to cite this URL: Chowdhury M, Hossain I, Deb A. K, Biswas T. K. Removal of Toxicants from Leather Industrial Wastewater using Sawdust Filter Media and Ferric Oxide (Fe2O3) Coagulant. Orient J Chem 2019;35(2). Available from: https://bit.ly/2GQVAZa |
Introduction
The leather is the second highest foreign currency earner sector in Bangladesh next to Ready Made Garments (RMG). The leather is used to making different type of products such as footwear, leather garments and leather goods etc. Among these products footwear itself consumed about 65% of the total leather. However, the tanning industries are considered as the most polluted sector in Bangladesh because of high level of chemicals including salinity, alkalinity, organic and inorganic matters, dissolved, suspended solids, total kjeldahl nitrogen (TKN), specific pollutants and different heavy metals etc.1-3 In recent time, the leather industries were relocated from Hazaribagh to the leather industrial zone in saver an attempt to save the Buriganga, the lifeline to the capital Dhaka. There are about 155 tanneries are allocated on 200 acres of land in the Savar Leather Industrial zone. They are consisting of two main tanning techniques such as chrome tanning and vegetable tanning.
In the present day, chromium salts (around 33% basic chromium sulphate) are extensively used as tanning substance. The hides and skins tanned with Cr salts have a better mechanical property, a remarkable dyeing propriety and hydrothermal property in comparison with hides and skins which is interacted with vegetable and other tanning materials. The leather takes up around 60% of applied chromium and the rest of metallic salts (40%) are usually discharged into wastewaters during the tanning period which have serious environmental impact.4 There are many techniques are available for tannery wastewater treatment to remove toxicant or pollution load but adsorption is one of the promising processes for wastewater treatment. Recently, low cost materials, including rice-husk, maple sawdust, soya cake, coal ash, peat and bone char, have been investigated as adsorbents for wastewater treatment.5 Au et. al. (2018)6 reported that saw dust was used to remove chromium from tannery effluents. Many researchers studied that in many cases after saw dust filter a chemical pretreatment was needed to activate the sorption sites.7 Batzias et al. (2013)8 reported that clay minerals are natural substances used in wastewater treatment for metal ions (Cd and Pb) removal.
The present experiment was to evaluate the degradation of toxicants from leather industrial wastewater by combined physical and chemical treatments. Iron salt was used as coagulant. The aim of this experiment was to determine the comparative toxicity of tannery wastewater before and after interaction. At first, pickling and chrome tanning wastewaters were characterized and then compared to the standard permissible limits. Therefore, in this experiment; a step has been made to analyze the effectiveness of saw dust adsorption filtration and coagulant Fe2O3 to interfere the tannery wastewater of Bangladesh. There are other treatment techniques found around the world, yet this experiment, considering the socioeconomic aspect of Bangladesh, cheap cost coagulant has been selected for the treatment of industrial tannery wastewater.
Materials and Methods
Study Location
The experimental area is located at the tannery industrial estate at Savar on the bank of the river Dhaleshwari and the South Western part of Dhaka city (Figure 1), Bangladesh. The wastewater was collected from Marsons Tannery Ltd. and Al-Madina Tannery in the tannery village.
The present study was conducted during the year November 2017- June 2018 and the tannery wastewaters were collected at February 2018 located in the sampling area. These discharged wastewaters were treated with sawdust filter and chemical processes.
 |
Figure 1: Sampling location of the leather industrial zone Savar, Dhaka Bangladesh. |
Treatment Process and Characteristics of Tannery Wastewater
The collected waste waters were performed both physical and chemical treatment. Firstly in physical treatment, 2 L of tannery wastewater was taken in a sample beaker and allowed to settle for 24 hours. After settling, 1 L supernatant was transfer into another beaker and added 50 mg/L lime to minimize the solubility of cations in wastewater and approved to settle down for a day. Total suspended solids were allowed to settle again by flock formation. After completion of settling, the supernatant clear liquids were enforced through saw dust filtration media.
In chemical treatment, Fe2O3 is considered as coagulant and a definite dose of coagulants, Fe2O3 (50, 70, 100, 150, 200, 250, 400 and 500 mg/L) were accumulated to the filtered tannery wastewaters, stirred evenly for 20 minutes and then approved to settle down for 12-72 hours. Then the liquids were collected and analyzed for different physical and chemical parameters every 2 hours during the process of treatment and finally the optimization of coagulation time was studied. The experiment was conducted using coagulant namely Fe2O3 which contributes Fe3+ ions in aqueous solution. The hydrolysis of Fe3+ (in aqueous solution) was illustrated and is written as follows9-10;
Fe3+ + y H2O ↔ Fe(OH)y(3-y)+ + yH+ (1)
Fe3+ → Fe(OH)2+ → Fe(OH)2+ → Fe(OH)3 → Fe2(OH)24+ → Fe3(OH)45+ → Fe(OH)4– (2)
The hydrolysis products Fe (III) may adsorb or chemically interact with suspended or dissolved components in the tannery effluents to form complex ions [9]. Ferric oxide (Fe2O3), was typically added during wastewater treatment and readily hydrolyses to form an amorphous ferri hydrite, Fe(OH)3 as stated by Amuda et al., (2006) [11], which is given below.
Fe2O3 + 6 H2O → 2Fe(OH)3↓ + 2H3O+ (3)
The optimizations of coagulant Fe2O3 doses and pH were determined. The pH of the sample was adjusted with 1.0 M NaOH or 1.0M HCl. A schematic flow diagram of the wastewater treatment process is shown in Figure 2.
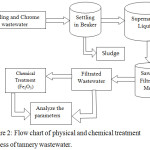 |
Figure 2: Flow chart of physical and chemical treatment process of tannery wastewater. |
All collected samples were analyzed for considering some major physical and chemical parameters. The physical and chemical parameters like pH, electrical conductivity (EC), TSS, and TDS, BOD5, COD of untreated, filtered and chemically treated wastewaters were measured. Colors were measured using Pt-Co color measurement unit. EC and pH values were found using digital pH meter (Model KP-5z, Japan) and a digital conductivity meter (Model Hanna 9033, Singapore). TDS, TSS and TS were determined gravimetrically (APHA 1998). COD value was measured using titrimetric method, (75 mL of H2SO4 (Conc.) was added to 25 mL of 0.25N K2Cr2O7 solutions then reflux it for two hours. The excess dichromate was then titrated with 0.25N (NH4)2Fe(SO4)2·6H2O solution using phenapthaline FeSO4 indicator). BOD5 was determined by 5 day BOD test method using oxitop machine. SO42-and Cl‑ ions were determined as per standard of APHA (2005). Concentrations of heavy metal ions (Cr, Cd, Pb and As) in wastewaters were estimated using AAS (Shimadzu Model AA-6800). Untreated wastewaters were used as control. Percentage of removal efficiency was calculated as follows:
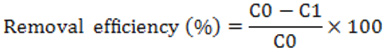
where, C0 was the concentration of control wastewaters, and C1 was the concentration of treated wastewaters.
Results and Discussions
The physical and chemical characteristics of industrial tannery wastewater filtered with sawdust filtration media and Fe2O3 coagulant are given tables 1 and 2.
Table 1: The characteristics of untreated and filtered industrial tannery wastewater and compared with standard.
|
Variables |
Untreated wastewater |
Saw dust filtered wastewater |
Standards permissible limits (ISI-2000/ISWBDS) |
| Appearance |
Yellowish – brown |
Black |
– |
| Color units (Pt-Co) |
(1772±2.17) |
(1455±4.49) |
15 |
| Odor |
Disagreeable |
Slightly – smell |
Smell less |
| pH |
7.6 ± 0.13 |
8.2 ±0.02 |
6-9 |
| EC (mS/cm) |
18.71±0.67 |
19.76 ±1.39 |
0.288 |
| TSS (mg/L) |
6778 ±37.45 |
5387 ±50.23 |
100 |
| TDS (mg/L) |
14020± 1.09 |
16028 ±60.88 |
2100 |
| TS (mg/L) |
20816±54.87 |
21425± 49.83 |
2200 |
| BOD5 (mg/L) |
935 ±15.61 |
1495±12.04 |
30/250 |
| COD (mg/L) |
3988 ±29.89 |
6789±31.01 |
250/400 |
| SO42- (mg/L) |
4012±14.42 |
3517 ±13.12 |
1000 |
| Cl– (mg/L) |
5017±12.84 |
1341±7.25 |
Table 2: The characteristics of Fe2O3 treated industrial tannery wastewater.
|
Variables |
Fe2O3 Coagulant dose (mg/L) |
|||||||
|
50 |
70 |
100 |
150 |
200 |
250 |
400 |
500 |
|
| Appearance |
DW |
Clear |
Clear |
Clear |
Clear |
Clear |
LB |
Br |
| Color units (Pt-Co) |
(360±) |
(280±) |
(5±) |
(25±) |
(80±) |
(120±) |
(400±) |
(530±) |
| Odor |
AGS |
AGS |
AGS |
AGS | AGS | AGS | AGS |
SS |
| pH |
10±0.5 |
9.6±0.56 |
9±0.2 |
8.5±0.25 |
8±0.26 |
7.5±0.24 |
6±0.25 |
5±0.2 |
| EC (mS/cm) |
6±0.12 |
5.6±0.21 |
1.5±0.5 |
2.5±0.4 |
4.65±1.0 |
5.6±0.35 |
6.8±0.5 |
8.8±0.5 |
| TSS (mg/L) |
400±2 |
200±1 |
0±1.5 |
100±2.5 |
200±0.2 |
600±2.2 |
1000±1.2 |
2000±5 |
| TDS(mg/L) |
5520±.06 |
4505±0.98 |
3062±2.5 |
3260±2.5 |
4075±2.6 |
4420±2.7 |
5320±3.5 |
6280±5.64 |
| TS (mg/L) |
5920±2.3 |
4705±0.69 |
3062±2.5 |
3360±0.95 |
4275±1.5 |
5120±1.6 |
6320±1.7 |
8280±1.56 |
| BOD5(mg/L) |
730±1.23 |
540±2.4 |
200±2.6 |
450±2.8 |
500±2.5 |
625±2.13 |
700±2.6 |
750±1.5 |
| COD(mg/L) |
1230±2.3 |
880±2.1 |
300±1.3 |
400±1.3 |
850±4.0 |
1050±1.6 |
1680±0.07 |
2020±0.05 |
| SO42-(mg/L) |
1550±1.5 |
1050±1.6 |
600±1.6 |
750±1.4 |
1060±.09 |
1530±1.0 |
1850±1.03 |
2350±1.5 |
| Cl—(mg/L) |
1040±1.8 |
850±0.09 |
480±1 |
580±1.5 |
880±1.63 |
1070±4 |
1250±2 |
2050±1.3 |
N.B.: DW: Dull- white color, AGS: Agreeable smell, LB: Light brownish, Br: Brownish and SS: Slightly smell
Color, Appearance and odor
The average color and appearance of untreated industrial tannery wastewater were determined to be 1772±2.17 Pt-Co units and yellowish brown color respectively. The results indicated that the industrial tannery wastewater contained highly color compounds which are 118 times higher than standard value (15 Pt-Co units). The actual color of the wastewater (1772±2.17 Pt-Co units) was decreased to 1455±4.49 Pt-Co units and appearance was found black due to filtration with sawdust filter media (Table 1). The odor was checked very bad smell from the untreated wastewater but unobjectionable odor was observed in the filtered wastewater. Similar review was found by Manu et al., 2011.12
The color of 100mg/L Fe2O3 treated wastewater was checked 5± (Pt-Co) unit which was found within standard limits. Evidently, clean water was found after completion of treatment with various doses (70-250) mg/L of Fe2O3. When the coagulant (Fe2O3) doses increased above 250mg/L, then the effluents’ color turned clear to brown color appear (Table 2). The research results determined that 100mg/L of Fe2O3 dose was the most practical for the removal of color. The results indicated that the color removal capacity was found better compared to the previous reports. Color results from treated industrial wastewater were found significantly different from the untreated wastewater. The odor was found very bad smell in untreated wastewater but very preferable odor was found after treatment with various doses of Fe2O3.
pH and EC
The results indicated that the mean values of pH, the industrial untreated and treated wastewater were 7.6±1.3 and 8.2±0.02 respectively (Table 1). After completion of filtration, the wastewaters were treated with various doses (50-500mg/L) of Fe2O3. The experimental results executed that the pH of treated wastewater were decreased with Fe2O3 doses increasing (shown in Table 2). Similar finding was reported by Chowdhury et al. (2013)4; pH is vital parameter since the coagulation happens within a definite pH range for each coagulant.
The EC of indusstrial untreated tannery wastewater was executed 18.71±0.13 mS/cm which was higher than standard limit (Table 1). Results explained that EC of industrial untreated wastewater generally contained around 20 mS/cm; Madal et al. (2010)13 supporting the findings of this experiment. The results illustrated that EC of Fe2O3 treated wastewater was decreased sharply with Fe2O3 doses increasing until reached at the lowest EC value around 1.5 and then it gradually increased with concentrations of Fe2O3. The lowest value of EC was observed in 100 mg/L Fe2O3 treated effluents indicating the efficiency of the coagulant dose (Figure 3). These experimental results also found relatively better removal capacities (92%) in removing the parameters at a dose of 100mg/L Fe2O3 and pH 9.
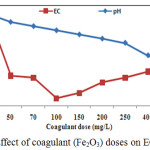 |
Figure 3: Effect of coagulant (Fe2O3) doses on EC and pH. |
TSS, TDS and TS
The mean values of TSS, TDS and TS of industrial untreated tannery wastewater were 6778±37.45, 14020±1.09 and 20816±54.87 mg/L accordingly, which were much high compared to the standard limits. After filtration with sawdust filter media, the values of TSS, TDS and TS were still very high (Table 1). The results showed that in the beginning TSS, TDS and TS decreased with increasing Fe2O3 doses until they reached at a certain lowest value and then increased with Fe2O3 (Table 2). The results also revealed that 100mg/L Fe2O3 treated wastewater showed the lowest TSS concentration which was well below the prescribed standard. 100mg/L of Fe2O3 treated wastewater showed the lowest TDS and TS values which were found to be the same i.e., 3062 mg/L, (Figure 4). The lowest values of TSS, TDS and TS were found to be in 100 mg/L of Fe2O3 treated wastewater. The results revealed that the concentration of TSS was almost “zero” (0) mg/L and the removal capacity was 100% at a coagulant (Fe2O3) dose of 100mg/L indication of the compatibility of the coagulant dose.
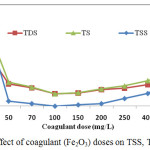 |
Figure 4: Effect of coagulant (Fe2O3) doseson TSS, TDS and TS. |
BOD5 and COD
The mean values of BOD5 and COD of industrial tannery wastewater were 935 and 3988 mg/L, successively. The experimental results described that BOD5 and COD values were found to be higher than those of standards for the ISW-BDS and ISI. The mean values of parameters BOD5 and COD for the filtered wastewater are shown in Table 1. The results revealed that the only filtration process was not good enough for BOD5 and COD removal suggesting the need for further treatment using suitable chemicals.
The results also illustrated that in the beginning BOD5, COD values decreased with increasing Fe2O3 doses till they reached at certain the lowest values and then increased with Fe2O3 doses (Table 2). The lowest values of BOD5 (200 mg/L) and COD (300 mg/L) were found in 100 mg/L Fe2O3 treated effluents which were found to be within the standard limits for ISW-BDS.
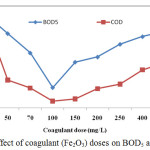 |
Figure 5: Effect of coagulant (Fe2O3) doses on BOD5 and COD. |
Sulfate (SO42-) and Chloride (Cl–)
The mean values of SO42- and Cl– in Fe2O3 treated wastewaters are shown in Table 2 which was found in relation between Fe2O3 doses and contained values of SO42- and Cl–. The analyze results revealed that 100 mg/L Fe2O3 treated effluent showed the highest removal efficiency for Cl– and SO42- and the values were found to be 480 and 600 mg/L, successively. The results indicated that the maximum SO42- and Cl– removal capacity executed were 85 and 90%, respectively and the value was below the standards limits. The study also observed that the concentration of SO42- in the untreated wastewater was 4012 mg/L and filtered wastewater was 3517 mg/L Table 1).
Concentration of Metal Alcalins/Ions
The average concentration of metals ions of the filtered and Fe2O3 treated wastewater are shown in tables 3 and 4.
Table 3: Reduction of dissolved cations from industrial tannery wastewater by filtration and compared with standard limits.
|
Variables |
Untreated wastewater |
Saw dust filtered wastewater |
Standards limits (ISI-2000)/ISWBDS |
| Na (mg/L) |
3862 ±38.02 |
3012±18.83 |
– |
| Ca (mg/L) |
774 ±4.20 |
726±3.90 |
200 |
| Cr (mg/L) |
9.89±0.68 |
6.72±0.21 |
0.1/2 |
| Pb (mg/L) |
5.28±0.17 |
5.12 ±0.17 |
0.2 |
| Cd (mg/L) |
3.47 ±0.048 |
3.32±0.19 |
0.1 |
| As (mg/L) |
1.40±0.14 |
1.25 ±0.21 |
0.1 |
Table 4: Reduction of cations of filtrated tannery wastewater by Fe2O3 treatment.
|
Variables |
Fe2O3 Coagulant dose (mg/L) |
|||||||
|
50 |
70 |
100 |
150 |
200 |
250 |
400 |
500 |
|
| Na(mg/L) |
1830±2.4 |
1020±1.3 |
460±1.63 |
860±2.5 |
950±1.8 |
1020±1.45 |
1300±1.56 |
1540±1.5 |
|
Ca((mg/L) |
280±2.7 |
200±1.8 |
140±1.8 |
240±1.02 |
360±1.4 |
450±1.2 |
650±2.06 |
750±0.41 |
| Cr(mg/L) |
5.2±0.07 |
3.5±0.5 |
0.1±0.05 |
0.65±0.06 |
0.95±0.06 |
2.5±1.3 |
3.5±.52 |
4.5±0.08 |
|
Pb(mg/L) |
2.5±0.05 |
0.67±0.6 |
0.2±0.03 |
0.45±0.09 |
0.70±0.04 |
1.5±0.5 |
2.5±0.06 |
3.5±0.21 |
|
Cd(mg/L) |
1.75±0.06 |
0.5±0.4 |
0.1±0.05 |
0.65±0.06 |
0.95±0.06 |
1.85±0.07 |
2.9±0.08 |
3.2±0.05 |
|
As(mg/L) |
0.65±0.04 |
0.35±0.05 |
0.1±0.05 |
0.2±0.04 |
0.3±0.02 |
0.5±0.05 |
0.65±0.09 |
0.95±0.06 |
The highest metal ions concentrations in the raw wastewater were found for Na and Ca and their mean values were 3862±38.02, and 774 ±4.20 mg/L, accordingly (Table 3). The research findings of the filtrated wastewater indicated that all the filtration processes were not suitable to remove heavy metal ions and Na, Ca alcalins.
The average concentration of Na, Ca, Cr, Cd, As and Pb ions in Fe2O3 (50-500 mg/L) treated effluents are illustrated in Table 4. The results observed that 100mg/L Fe2O3 treated wastewater contained the lowest Na value which was found to be 460 mg/L. The value was still far above the standards limits, suggesting the need for further treatment. The analysis results also observed that 100mg/L Fe2O3 treated effluents showed the lowest concentration of major metal ions and the values were: 140, 0.1, 0.2, 0.1 and 0.1 mg/L for Ca, Cr, Pb, Cd and As respectively (Table 4). The research revealed that 100 mg/L Fe2O3 dose found the best removal efficiency for all of major metal ions.
Influencing Parameters on Treatment Processes
Effect of Coagulant Dose
Various amount of Fe2O3 (50-500 mg/L) doses were applied to observe the optimum coagulant dose at definite pH 9 and room temperature (25±1°C) for the treatment of wastewater. The effect of coagulant (Fe2O3) dose on the removal capacity of various indicators (EC, TSS COD, SO42-, Cl– Cr, As and Cd) are illustrated in Figure 6. The figure explained that the removal efficiency of parameters initially increased with dose until it reached peak point and then it decreased. The maximum removal efficiencies for various factors were achieved 92% (EC), 92% (COD), 100% (TSS), 85% (SO42-), 99% (Cr), 93% (As), and 97% (Cd). The findings of this experiment executed that the coagulant was not adequate to form flocks or to bind the colloid particles to form bridging. At higher doses (more than100 mg/L) of the coagulant, the colloid particles become suspended due to electrostatics repulsion between colloid particles and coagulant and in low flock appearance. The present experiment observed that 100 mg/L coagulant (Fe2O3) dose indicated maximum removal capacity. A high coagulant dose in effluent treatment process increase operational costs and produce a large amount of sludge. In this study, the coagulant amount used was relatively smaller than used in the previous studies.14
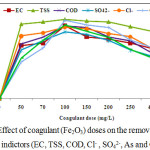 |
Figure 6: Effect of coagulant (Fe2O3) doses on the removal capacity of various indictors (EC, TSS, COD, Cl– , SO42-, As and Cr). |
Effect of pH
The study carried out in the pH range from 4 to 11.5 to determine the optimum pH value at a definite coagulant dose (100 mg/L Fe2O3) and room temperature (25±1°C). The effect of pH on the removal capacity (%) is given in Fig. 7. The maximum capacities for various factors were found to be at pH 9 and they gradually decreased on both sides of pH.
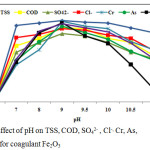 |
Figure 7: Effect of pH on TSS,COD, SO42- , Cl-, Cr, As,Cd and Pb for coagulant Fe2O3. |
The results illustrated that the highest removal capacities were 100, 92, 85, 90, 99 and 92 % for TSS,COD, SO42-, Cl-, Cr and As respectively. When a coagulant such as iron salt is added to H2O, a series of soluble hydrolysis species are to be formed. These hydrolysis species have positive or negative charges considering on the pH of water. At low pH (< 6) they are positively charged and at high pH they are negatively charged.
Effect of Time
The removal capacity of physical and chemical variables on the reaction time is illustrated in Figure 8. The experimental results of the chemical and following treatment explained those 12 hours were not enough to find maximum removal capacity for any pollutants, but after one day the removal capacities for all factors executed maximum and they remained constant after treatment period two or more day’s (Figure 8). The experimental results indicated that the time of coagulation reaction and following settling has a large effect on the removal capacity for different factors. Thus the study suggested that 24 hours reaction time would be sufficient to accomplish the hydrolysis and sedimentation processes for the coagulant.
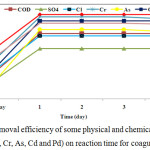 |
Figure 8: The removal efficiency of some physical and chemical variables (TSS, COD, SO42-, Cl–, Cr, As, Cd and Pd) on reaction time for coagulant Fe2O3. |
Conclusion
The leather industries in Bangladesh are considered as one of the most serious pollutants industries in terms of both environmental concern and toxic parameters. The study found that tannery wastewaters contained exceptionally high values of TDS, TSS, TS, BOD, COD, SO42- , Cl– and different heavy metals. The experimental results indicated that the theses physico-chemical parameters of the collected wastewater were exceeded the standard discharge limits. These tanning industrial wastewaters are very much harmful and toxic to surface water which poses potential threads to water ecosystem as well as human health. The study elucidated that the filtration processes could reduce certain amount of toxicants load from the industrial wastewater and its need further chemical treatment to discharge the effluents into river by maintaining standard permissible discharge limits. According to removal efficiency of toxicants load at optimum pH 9, it executed that chemical treatment with 100 mg/L coagulant (Fe2O3) does indicated the best results of the physical and chemical parameters which were much below the standard tannery wastewater discharged limits. In order to reduce the toxicants load from tannery wastewater, the study suggested that both combined physical (Filtration) and chemical (coagulation (Fe2O3)) treatment processes could be fruitful.
Acknowledgements
The authors wish to acknowledge the Centre of Advanced Research in Sciences (CARS), University of Dhaka; Central Science Laboratory and the physical Chemistry laboratory, Department of Chemistry, Rajshahi University, Bangladesh for providing the technical facilities to carry out the experiments.
Funding Source
There is no funding source.
Conflict of Interest
There is no conflict of interest.
References
- Ş. Sungur and A. Özkan, “Characterization of Wastewaters Obtained from Hatay Tanneries,” Nat. Eng. Sci., 2017, 2(2), 111–118.
- B. O. Ejechi and O. O. Akpomie, “Removal of Cr (VI) from Tannery Effluent and Aqueous Solution by Sequential Treatment with Microfungi and Basidiomycete-Degraded Sawdust,” J. Environ. Prot. (Irvine,. Calif)., 2016, 07(06), 771–777.
- E. Leghouchi, E. Laib, and M. Guerbet, “Evaluation of chromium contamination in water, sediment and vegetation caused by the tannery of Jijel (Algeria): a case study,” Environ. Monit. Assess., 2009, 153(1–4), 111–117.
- M. Chowdhury, M. G. Mostafa, T. K. Biswas, and A. K. Saha, “Treatment of leather industrial effluents by filtration and coagulation processes,” Water Resour. Ind., 2013, 3, 11–22, Sep.
- S. Kakkar, A. Malik, and S. Gupta, “Treatment of pulp and paper mill effluent using low cost adsorbents: An overview,” J. Appl. Nat. Sci., 2018, 10(2), 695–704.
- I. Au, M. E. Khan, D. P. Feka, and B. Ogoh, “Tannery Wastewater Evaluation and Remediation: Adsorption of Trivalent Chromium Using Commercial and Regenerated Adsorbents,” J Water Technol Treat Methods, 2018, 1(1), 104,
- F. A. Batzias and D. K. Sidiras, “Dye adsorption by calcium chloride treated beech sawdust in batch and fixed-bed systems.,” J. Hazard. Mater., 2004, 114(1–3), 167–74.
- X. J. Jenitta, V. Daphne, V. Gnanasalomi, and J. J. Gnanadoss, “Treatment of Leather Effluents and Waste Using Fungi,” 2013.
- A. I. Zouboulis, P. A. Moussas, and F. Vasilakou, “Polyferric sulphate: Preparation, characterisation and application in coagulation experiments,” J. Hazard. Mater., 2008, 155(3), 459–468.
- Jia-Qian Jiang and Nigel J D Graham, “Pre-polymerised inorganic coagulants and phosphorus removal by coagulation – A review,” 1998, 24(3), 239–244.
- O. S. Amuda, I. A. Amoo, and O. O. Ajayi, “Performance optimization of coagulant/flocculant in the treatment of wastewater from a beverage industry.,” J. Hazard. Mater., 2006, 129(1–3), 69–72.
- K. J. Manu, V. S. Mohana and G. K. Ganeshaiah, “Effluent Generation by the Dairy Units: Characterization and Amelioration for Irrigation,” Int. J. Res. Chem. Environ., 173–182, 2011.
- T. Mandal, D. Dasgupta, S. Mandal, and S. Datta, “Treatment of leather industry wastewater by aerobic biological and Fenton oxidation process.,” J. Hazard. Mater., 2010, 180(1–3), 204–11.

This work is licensed under a Creative Commons Attribution 4.0 International License.









