Investigation on Stability of Electroplated-Sulfurized CuInS2-based Photocathode Modified with an In2S3 Layer for H2 Evolution under Various pH Conditions
Gunawan1, Abdul Haris1, Hendri Widiyandari2, Wilman Septina3 and Shigeru Ikeda4
1Department of Chemistry, Faculty of Sciences and Mathematics, Diponegoro University, Semarang, Indonesia.
2Department of Physics, Faculty of Sciences and Mathematics, Diponegoro University, Semarang, Indonesia.
3Department of Chemistry, University of Zurich, Winterthurerstrasse 190, CH-8057 Zurich, Switzerland.
4Department of Chemistry, Konan University, 8-9-1 Okamoto, Higashinada, Kobe, Japan.
Corresponding Author E-mail: gunawan_undip@yahoo.com
DOI : http://dx.doi.org/10.13005/ojc/330202
Article Received on : March 31, 2017
Article Accepted on : April 17, 2017
We investigated photoelectrochemical water splitting properties of Pt-In2S3/CuInS2 photocathodes from near neutral toward basic condition (pH 6, 9, and 13). Half-cell solar-to-hydrogen (HC-STH) of Pt-In2S3/CuInS2 measured under 0.2 M NaH2PO4 was the highest when the pH of the solution was 13 (4.7% at 0.42 V vs. RHE) with a maximum photocurrent density of 16.3 mA cm-2 (at 0 V vs. RHE) and onset potential of 0.9 V vs. RHE (defined as cathodic photocurrent of 0.1 mA cm-2). However, it suffers from rapid degradation of the photocurrent over the time course when kept at 0 V vs. RHE under constant illumination. On the other hand, the photocathode showed relatively stable photocurrent when measured under near neutral condition (pH 6): HC-STH of 2.85% (at 0.28 V vs. RHE) with a maximum photocurrent density of 16.3 mA cm-2 (at 0 V vs. RHE) and onset potential of 0.72 V vs. RHE were obtained from the photocathode. Passivation using In2S3 oxygen-free can stabilize the photocathode at alkaline pH (pH 13).
KEYWORDS:Photoelectrochemical water splitting; copper indium sulfide; indium sulfide; stability
Download this article as:| Copy the following to cite this article: Gunawan G, Haris A, Widiyandari H, Septina W, Ikeda S. Investigation on Stability of Electroplated-Sulfurized CuInS2-based Photocathode Modified with an In2S3 Layer for H2 Evolution under Various pH Conditions. Orient J Chem 2017;33(2). |
| Copy the following to cite this URL: Gunawan G, Haris A, Widiyandari H, Septina W, Ikeda S. Investigation on Stability of Electroplated-Sulfurized CuInS2-based Photocathode Modified with an In2S3 Layer for H2 Evolution under Various pH Conditions. Orient J Chem 2017;33(2). Available from: http://www.orientjchem.org/?p=32415 |
Introduction
Hydrogen (H2) produced by photoelectrochemical (PEC) water splitting via direct use of sunlight irradiation is considered to be a holy grail to provide sustainable fuel that can support human life for many years to come. PEC approach by using TiO2 photoelectrode illuminated with UV light was demonstrated by Fujishima et al. as the first realization of water splitting.1 This result triggered other researchers to investigate and propose numerous varieties of semiconductors and device configurations to realize an efficient water splitting system to produce H2.2-6
Tandem water splitting system has been emerged as the most viable solution for the practical application due to the difficulty in finding a single semiconductor that is able to perform efficiently both water reduction and oxidation simultaneously. It consists of a photocathode and a photoanode where each of which is responsible for the water reduction and oxidation, respectively. Therefore, each of the electrodes allows flexibility to be investigated and optimized its potency as a promising semiconductor to achieve an efficient overall water splitting system using only sunlight (no bias).
Considering the photocathode part, Cu-chalcopyrite materials, for instance CuInSe2, CuGaSe2, CuInS2, CuGaS2 and their mixed crystals, have emerged as promising candidates to produce H2.7-11 Moreover, through bandgap engineering from 1.0 eV to 2.4 eV by varying the In/Ga and/or Se/S ratios in their mixed crystal promises a versatile flexibility to optimize the appropriate tandem design with photoanode as another part. Researchers by using different absorbers had inserted n-type thin layers on their surface to improve the photo response, which are widely employed in fabrication of p-n junctions for chalcopyrite-based solar cells.12-15 We used In2S3 by chemical bath deposition (CBD), in our previous study, as an alternative to the commonly-used CdS as the n-type layer on CuInS2 photocathode and obtained in half-cell solar-to-hydrogen (HC-STH) efficiency close to 2%.16Moreover, the photocurrent onset was shifted positively, which made this photocathode system become more favorable for the tandem PEC system with unbiased potential.
In2S3-modified CuInS2 photocathode showed higher water reduction properties; however, its behavior under different pH was not well confirmed. Consequently, we wanted to investigate the water reduction properties of In2S3-modified CuInS2 in various pH from near neutral to alkaline conditions. The photostability of the photocathode under various pH conditions also will be discussed.
Experimental Details
Fabrication of CuInS2 Thin Films
The CuInS2 thin film was synthesized by successive stack electrodeposition of Cu and In layers followed by sulfurization. Electrodeposition was carried out potentiostatically using a Hokuto Denko HSV-100 potentiostat-galvanostat under N2 atmosphere without stirring. A vertical three-electrode setup consisting of an Ag/AgCl reference electrode, a Pt wire counter electrode, and a Mo-coated glass substrate (Mo/glass) as a working electrode (area of the working electrode being 0.7 cm2) was employed. The electrolyte solution for Cu deposition consisted of 50 mM CuSO4, 150 mM trisodium citrate, and 242 mM citric acid. The solution was adjusted to pH 2.4 using H2SO4. For In deposition, the electrolyte solution consisted of 30 mM InCl3, 242 mM citric acid, and 36 mM trisodium citrate. Potentials used for Cu and In deposition were -0.2 V and -0.76 V (vs. Ag/AgCl), respectively. Electric charges of Cu and In deposition were fixed at 0.73 C and 0.84 C, respectively: this resulted in the composition ratio of Cu and In (Cu/In) in a Cu and In stacked layer of 1.3. The Cu/In stack as-deposited was then heated at 160 oC for 30 min under Ar flow (200 mL min-1), followed by sulfurization at 560 °C under H2S flow (5% H2S in Ar, 200 mL min-1) for 10 min in a glass tube furnace. Thus-obtained CuInS2 films were then etched by immersion in an aqueous KCN solution (10%) for 2 min to remove excess CuxS components.
Surface Modification With In2S3 Layer
Surface modification was performed by chemical bath deposition (CBD) method on the prepared CuInS2 film by In2S3 layers. In a typical procedure, the prepared CuInS2 was added to an aqueous solution containing 25 mM In2(SO4)3, 0.1 M CH3CSNH2 and 0.1 M CH3COOH; deposition of In2S3 was performed at 65oC for 15 min. Finally the films were washed with and dried by N2 gas. The deposition gave the formation of ca. 15 nm thick In2S3 layer on CuInS2 films (labeled In2S3/CuInS2). For revised In2S3 deposition we used 25 mM In2(SO4)3, 0.2 M CH3CSNH2 and 1 M CH3COOH in demineralized water after removing the dissolved oxygen using N2 for1 h, the similar procedure for revised In2S3 by CBD was used as above.
Surface Modification With Pt
Photoelectrodeposition in three-neck cylindrical flask containing 20 mL solution of 0.1 M Na2SO4 and 1 mM H2PtCl6 at pH 4 was performed for Pt deposition modified-CuInS2 films. Three electrodes namely Pt wire, Ag/AgCl and modified CuInS2 as counter, reference and working electrodes, respectively were inserted into the flask. By continuous illuminating the working electrode through the window the photoelectrodeposition was performed at a potential of -0.1 V for 10 min using electrochemical measurement unit (Solartron SI 1280B) equipped with a solar simulator AM-1.5 (Compact Xenon Light Source, Asahi Spectra HAL-320). Thus obtained In2S3/CuInS2 modified with Pt was labeled as Pt-In2S3/CuInS2.
Photoelectrochemical Measurements
Photoelectrochemical measurements were done using the same instruments as surface modification with Pt completed with digital function generator 0.1 mHz-2MHz DF 1906, NF Japan (at 0.3 Hz) and Shutter Controller F77 (Suruga Seiki). Solutions of 0.2 M NaH2PO4 (at pH 6, 9 and 13) were used for measurements of Pt-In2S3/CuInS2 photocathodes. The three electrode setup was immersed in each solution. The measurement was run by chopping 1.5 AM light irradiation to the working electrode under sweep potential with scan rate 10 mV/s.
The potentials in each PEC measurement for Pt-In2S3/CuInS2 photocathodes referred to Ag/AgCl electrode (EAg/AgCl) were converted into ERHE using the following equation:
ERHE = EAg/AgCl + 0.059 × pH + 0.199.
Half-cell solar-to-hydrogen efficiency (HC-STH) for photocathode was calculated from the data according to the following equation:
HC-STH [%] = J × (V-VH+/H2) × 100% / P,
Where J (mA cm-2) is the measured photocurrent density, V (V vs. RHE), is the bias voltage measured by the reversible hydrogen electrode (RHE) scale (ERHE, see above), V-VH+/H2 is the equilibrium redox potential of hydrogen (0 V vs. RHE) and P is the photon flux of the AM 1.5 simulated sunlight (100 mW cm-2).
Characterizations of Fabricated Photocathodes
Surface morphologies were examined using a Hitachi S-5000 FEG field emission scanning electron microscope (FE-SEM). Surface structures of Pt-CdS/CuInS2 and Pt-In2S3/CuInS2 films were examined by X-ray photoelectron spectroscopy (XPS) using a Shimadzu AXIS ULTRA X-ray photoelectron spectrometer. Elemental compositions of thus-obtained films were determined using a Hitachi TM3000 scanning electron microscope equipped with a Swift ED3000 energy dispersive X-ray spectrometer (EDX).
Results and Discussion
We investigated the performance of Pt-In2S3/CuInS2 photocathodes at various pH conditions from near neutral to alkaline condition. Figure 1 shows current-potential profiles of Pt-In2S3/CuInS2 photocathodes in 0.2 M NaH2PO4 at pH 6.0, 9.0 and 13.0 (adjusted by NaOH) measured under chopped illumination from AM 1.5 G (100 mW/cm2) solar simulator. Table 1 summarized photocurrent density (at 0 V vs. RHE), onset potential (defined as cathodic photocurrent of 0.1 mA cm-2), and half-cell solar-to-hydrogen (HC-STH) efficiency of Pt-In2S3/CuInS2 measured under 0.2 M NaH2PO4 at various pH. The onset potential was clearly shifted toward positive potential with the increase of pH. This positive shift of the potential with the increase of pH is similar to the behavior of Cu2ZnSnS4-based photoelectrode13 and flatband potential of Cu(In,Ga)S2.17 In our case, similarly, the positive shift of the onset potential could be influenced by the deeper flat-band potential of CuInS2 with the increase of pH. Photocurrent density of the photocathodes at 0 V vs. RHE were relatively similar (ca. 16 to 17 mA.cm-2). Maximum HC-STH of 4.7% (at 0.42 V vs. RHE) was obtained from Pt-In2S3/CuInS2 measured at pH 13.
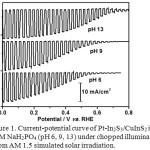 |
Figure 1. Current-potential curve of Pt-In2S3/CuInS2 in 0.2M NaH2PO4 (pH 6, 9, 13) under chopped illumination from AM 1.5 simulated solar irradiation. |
Tabel 1: Photocurrent density (at 0 V vs. RHE), onset potential (defined as cathodic photocurrent of 0.1 mA cm-2), and HC-STH of Pt-In2S3/CuInS2 measured under 0.2 M NaH2PO4 at various pH.
| pH | 6 | 9 | 13 |
| Photocurrent density/ mA cm-2 | 16.3 | 16.7 | 16.3 |
| Onset potential / V vs. RHE | 0.72 | 0.75 | 0.9 |
| HC-STH / % | 2.85 | 2.85 | 4.7 |
| (at 0.28 V) | (at 0.30 V) | (0.42 V) |
To investigate the stability of the Pt-In2S3/CuInS2 under reductive condition at various pH, the photocathode was then exposed to continuous illumination from AM 1.5 solar simulator while keeping at a bias potential of 0 V vs. RHE. Figure 2 shows current-time profiles of the photocathode at such measurement conditions. The photocurrent initially started at a relatively similar photocurrent density (ca. 16 mA cm-2), then the photocathode measured at pH 6 shows relatively stable photocurrent along the time course, while at the basic conditions the photocurrent keeps degrading and getting worse with the increase of pH: the percentage of degradation compared to initial photocurrent was 16%, 78%, and 91% for pH 6, 9, and 13, respectively.
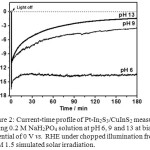 |
Figure 2: Current-time profile of Pt-In2S3/CuInS2 measured using 0.2 M NaH2PO4 solution at pH 6, 9 and 13 at bias potential of 0 V vs. RHE under chopped illumination from AM 1.5 simulated solar irradiation. |
It is clear from the above results that the alkaline condition is not favorable for the long-term stability of Pt-In2S3/CuInS2 under continuous illumination. In addition, when we repetitively measured the photocathode under cathodic swept at pH 13, both photocurrent and onset potential was gradually decreased: no clear degradation was observed upon such repetitive scan for pH 6 and 9. Furthermore from SEM images we cannot clearly see the SEM changes from initial photocathode compared with photocathodes after measured in pH 6, 9 and 13 for 3 h.
In fact, immersion of bare In2S3 deposited on FTO substrate at pH 13 for 5h resulted in dissolution of the In2S3 layer from the substrate. Accordingly, to know the further change, we treated the photocathode at pH 13 for a longer time (15 h) under continuous illumination at 0 V vs RHE, the result is depicted in Figure 3 and 4.
SEM image of Figure 3 shows the formation grape-like on the photocathode that most probably hydroxide attachments on the indium sulfide layer, as can be seen in Figure 4 the survey scan for In 3d of initial Pt-In2S3/CuInS2 using XPS analysis shows the peaks of 444.8 and 452.4 eV as representation of indium bound as In2S3, and small shoulders around 443.6 and 451.2 eV as indium hydroxide peaks. Photocathode of Pt-In2S3/CuInS2 after used there is a little bit shift to higher binding energy for survey scan of In 3d namely 445.3 and 452.9 eV main peaks and also there are increasing peak height at the shoulder with binding energy of 443.9 and 451.4 eV that representing the improvement of indium that binds OH– ions from the solution for sample after used. As we know In2S3 prepared by CBD using indium sulfate and thioacetamide will resulted almost 80% of In2S3.18 Therefore the presence of OH in its structure will tend to attract other OH– ions from the solution.
 |
Figure 3: SEM of Pt-In2S3/CuInS2 photocathode after used.
|
The stability test was done for 15 h at pH 13 in 0.2 M NaH2PO4 solution (0 V vs. RHE) under continuous illumination from AM 1.5 simulated solar irradiation.
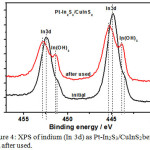 |
Figure 4: XPS of indium (In 3d) as Pt-In2S3/CuInS2 before and after used. |
The stability test was done for 15 h at pH 13 in 0.2 M NaH2PO4 solution (0 V vs. RHE) under continuous illumination from AM 1.5 simulated solar irradiation.
Figure 5 represents the current time curve of Pt-In2S3/CuInS2 of using In2S3 with oxygen-less content (revised In2S3 by CBD), oxygen was not detected (the data not shown). The figure can be seen the photocathode gave stable for 3 h continuous illumination. Moreover the current increased to 90 min and began to decrease after that and finally reduced a little bit after 160 min from the initial measurement. Compared with standard Pt-In2S3/CuInS2 the initial current diminished almost 54% (from 16.8 mA/cm2 reduced to 9 mA/cm2). The most probable reason is due to slightly blocking of incident light that reduces In2S3 transparency. Another possibility is reducing bandgap of revised In2S3 that can degrade the performance of photocathode, since reducing oxygen will lower conduction band position.19Similarly as solar cell, therefore, for In2S3 there must have been tunable oxygen to improve its performance.
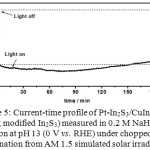 |
Figure 5: Current-time profile of Pt-In2S3/CuInS2 (using modified In2S3) measured in 0.2 M NaH2PO4 solution at pH 13 (0 V vs. RHE) under chopped illumination from AM 1.5 simulated solar irradiation. |
Conclusion
PEC water splitting of Pt-In2S3/CuInS2 photocathodes for H2 evolution showed a more enhanced photocurrent density under 0.2 M NaH2PO4 (pH 6). PEC water splitting study of Pt-In2S3/CuInS2 photocathodes reveal that HC-STH and onset potential values are quite influenced by the pH of the solutions, HC-STH and onset potential improve with increasing pH with the maximum value was 4.7% at pH 13. Long term stability test of Pt-In2S3/CuInS2 shows that the photocathode is stable at pH 6 while at basic condition it suffers from rapid degradation of the photocurrent over the time course when kept at 0 V vs. RHE. The used revised In2S3 can stabilize of Pt-In2S3/CuInS2 photocathode under long term illumination, although the current decreased compared with standard photocathode.
Acknowledgments
Financial support From Diponegoro University (No.: SP-042.01.2.400898/2016), Ministry of Research and Higher Education Republic of Indonesia is acknowledged.
References
- Fujishima, A.; Honda, K. Nature 1972, 238, 37.
CrossRef - Domen, K.; Kondo, J. N.; Hara, M.; Takata, T. Bull. Chem. Soc. Jpn. 2000, 73, 1307-1331.
CrossRef - Sato, J.; Saito, N.; Yamada, Y.; Maeda, K.; Takata, T.; Kondo, J. N.; Hara, M.; Kobayashi, H.; Domen, K.; Inoue, Y. J. Am. Chem. Soc. 2005, 127, 4150-5151.
CrossRef - Maeda, K.; Teramura, K.; Lu, D.; Takata, T.; Saito, N.; Inoue, Y.; Domen, K. Nature 2006, 440, 295.
CrossRef - Osterloh, F. E. Chem. Mater. 2008, 20, 35-54.
CrossRef - Yokoyama, D.; Minegishi, T.; Maeda, K.; Katayama, M.; Kubota, J.; Yamada, A.; Konagai, M.; Domen, K. Electrochem. Commun. 2010, 12, 851-853.
CrossRef - Ikeda, S.; Nakamura, T.; Lee, S. M.; Yagi, T.; Harada, T.; Minegishi, T.; Matsumura, M. ChemSusChem. 2011, 4, 262-268.
- Kim, J.; Minegishi, T.; Kubota, J.; Domen, K. Energy Environ. Sci. 2012, 5, 6368-6374.
CrossRef - Maeda, K.; Domen, K. J. Phys. Chem. C 2007, 111, 7851-7861.
CrossRef - Djellal, L.; Bellal, B.; Trari, M. Mater. Chem. and Phys. 2012, 137, 340-345.
CrossRef - Ikeda, S.; Nonogaki, M.; Septina, W.; Gunawan, G.; Harada, T.; Matsumura, M. Catal. Sci. Tecnol. 2013, 3, 1849-1854.
CrossRef - Moriya, M.; Minegishi, T.; Kumagai, H.; Katayama, M.; Kubota, J.; Domen, K. J. Am. Chem. Soc. 2013, 135, 3733-3735.
CrossRef - Yokoyama, D., Minegishi, T.; Jimbo, K.; Hisatomi, T.; Ma, G.; Katayama, M.; Kubota, J.; Katagiri, H.; Domen, K. Appl. Phys. Express 2010, 3, 101202.
CrossRef - Ma, G.; Minegishi, T.; Yokohama, D.; Kubota, J.; Domen, K. Chem. Phys. Lett. 2011, 501, 619-622.
CrossRef - Rovelli, L.; Tilley, S.D.; Sivula, K. ACS Appl. Mater. Interfaces 2013, 5(16), 8018-8024.
CrossRef - Gunawan; Septina, W.; Ikeda, S.; Harada, T.; Minegishi, T.; Domen, K.; Matsumura, M. Chem. Commun. 2014, 50, 8941.
- Fernández, A.; Dheree, N.; Turner, J.; Martinez, A.; Arriaga, L.; Cano, U. Sol. Energy Mater. Sol. Cells 2005, 85, 251-259.
CrossRef - Ahn, B.T.; Larina, L.; Kim, K.H.; Ahn, S. J. Pure Appl. Chem. 2008, 80(10), 2091-2102.
CrossRef - Barreau, N. Solar Energy 2009, 83, 363–371.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.









