Evaluation of Apoptosisrate of Pegylated nanoliposomal oxaliplatin in Breast Cancer MCF-7 and MDA-MB-231 cells
Delaram Ahmadi¹, Azim Akbarzadeh², Mahdi Arjmand³ and Amir Heidarinasab4
¹Department of Chemical Engineering, Science and Research Branch, Islamic Azad University, Tehran, Iran.
²Department of Pilot Nanobiotechnology, Pasteur Institute of Iran, Tehran, Iran.
³Department of Chemical Engineering, South Tehran Branch, Islamic Azad University, Tehran, Iran.
4Department of Chemical Engineering, Science and Research Branch, Islamic Azad University, Tehran, Iran.
Corresponding Author E-mail: azimakbarzadeh1326@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/320610
Cancer is an extremely dangerous disease among humans.breast cancer is still mostfrequently form of cancerwithin women population. Oxaliplatin isaapoptotic effectual compound in order to treatment of cancer with cytotoxic side effects. Nanocarriers with specific physicochemical properties are able to reduce side effectsof anticancer drugs.Nanoliposomes as carrierare the promising systemfor cancer treatmen due to stability,high targetingproperties, better bioavailability, slow‑releasing and low systemic toxicity. By using polyethylene glycol (PEG)canincrease blood- circulationtime of the drugwith decreased side effects and improved efficacy alsoPEGcanincreasepenetration to the the tumor tissue.In this investigation, oxaliplatin was loaded onto pegylated liposomes through reverse phase evaporation method. Profile of drug release were evaluated.Pegylatednanoliposomaloxaliplatinexhibited slow‑releasing potential in comparison to conventional formulations. The morphology of particles in each field wasanalyzedbyscanning electron microscopy (SEM).Apoptosis rate wasevaluatedin breast cancer cell lines MCF-7 and MDA-MB-231 by flow cytometry.Pegylatednanoliposomaloxaliplatinshowed a suitable impact on apoptosis of breast cancer cell lines MCF-7 and MDA-MB-231 . Thus, pegylatednanoliposomaloxaliplatin can employ with low systemic toxicity in order to treatment of breastcancer.
KEYWORDS:Breast cancer; Oxaliplatin; Liposome; Polyethyleneglycol; Nanodrugdelivery
Download this article as:| Copy the following to cite this article: Ahmadi D, Akbarzadeh A, Arjmand M, Heidarinasab A. Evaluation of Apoptosisrate of Pegylatednanoliposomaloxaliplatinin Breast Cancer MCF-7 and MDA-MB-231cells. Orient J Chem 2016;32(6). |
| Copy the following to cite this URL: Ahmadi D, Akbarzadeh A, Arjmand M, Heidarinasab A. Evaluation of Apoptosisrate of Pegylatednanoliposomaloxaliplatinin Breast Cancer MCF-7 and MDA-MB-231cells. Orient J Chem 2016;32(6). Available from: http://www.orientjchem.org/?p=26380 |
Introduction
Cancerous diseases are considered as major causes of Deaths and mortality in the world and at present, breast cancer isone of the cancers causing death in womenpopulation. There are several ways for curing breast cancer and one of them is chemotherapy [1].
One of chemotherapy drugs based on platinum for treatment of cancer isoxaliplatin (Figure1) [2]. Oxaliplatin Anticancer drug is third generation platinum (Pt).Oxaliplatin is highly potent which be employed to treat cancer.Itinhibits DNA synthesis by involving DNA cross linking mechanisms [3].
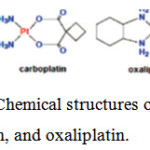 |
Figure 1: Chemical structures of cisplatin, carboplatin, and oxaliplatin. |
Liposomes are vesicles of lipid closed bilayers phospholipid systems with an aqueous cavity.liposomes can be used to vesiculize both hydrophilic and lipophilic drugs [4].Modes of liposome/cell intraction are consist of fusion and endocytosis [5,6].Polyethylene glycol attached to surface of a liposome can reduce interactions with reticuloendothelial system (RES) and increase blood – circulation time of the liposome(Figure 2)[7,8].
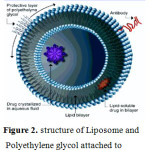 |
Figure 2: Structure of Liposome and Polyethylene glycol attached to surface of a liposome. |
Liposomes accumulate in tumor tissue due to the phenomenon of enhanced permeability and retention (EPR) effect [9]..
Purpose of this investigation was improvment therapeutic index of breast cancer.
Materials and Methods
Materials
MTT (3-(4,5-di-methylthiazolyl-2)-2, 5-di-phenyltetrazolium bromide), phosphatidylcholine,cholesterol, polyethylene glycol 2000 (PEG 2000),dextrose ,fluorescent iso-thiocynate (FITC), propidium iodide(PI) were purchased from sigma company (SIGMA, USA).Chloroform, Ethanol and Isopropanol purchased from Merck Company (Merck, Germany).The RPMI-1640 culture medium was purchased from Invitrogen (Invitrogen, USA) and Oxaliplatin waspreparated fromSobhanAncology Company. Breast cancer cell lines (MCF-7, MDA-MB-231) were purchased from national cell bank of Pasteur institute of Iran.
Preparation of nanoparticle spegylatedliposomaloxaliplatin
Pegylated liposomal oxaliplatin was prepared using the reverse phase evaporation technique.Phosphatidylcholineand cholesterol and polyethylene glycol 2000 (molar ratio 2:1:0.2)were dissolved in 15 mlchloroform and then 5 ml ofoxaliplatin solution(1 mg/ml)in 5% (w/v) dextrose was added toresultanmixture.Sample no oxaliplatin-containing liposomes,5% dextrose solution was dropped instead of oxaliplatin solution.The resultant solution was stirred(at 300 rpm, at roomtemperature, 1h) to gain a transparent, yellow suspension. The solvent phase was removed by rotary evaporator(0.045 mpa, 2h , 40° C)(Heidoiph, Germany)(pegylatednanoliposomaloxaliplatin and pegylatednano liposomal) .The buffer phosphate saline(pH 7.4)(30ml)wasdroppedinto resultant liposomes.Resultantsolution was stirred (150 rpm, room temperature, 24 h).The emulsion wassonicated(BandelinSonorex Digital, 60 HZ) for 10 min to reduce the size of liposomes. [14]Then resultant solution was hemogenised(7000 rpm, 5 min) (7000 rpm, 30 min)andThen was extruded through a polycarbonate membrane (200 nm poresize)(pegylatednanoliposomaloxaliplatin and pegylatednano liposomal).
Scanning Electron Microscopy (SEM)
A drop of aqueous suspension of pegylatednanoliposomaloxaliplatinwas spread on a slab and dried under vacuum. The diameter of particles in each sample(sample hemogenised at 7000 rpm, 5 min and sample hemogenised at 7000 rpm, 30 min) was analyzed by using JSM-5200 operation of scanning electron
microscope (Tokyo, Japan) at 15 kV.
In Vitro Release Study
Oxaliplatin release ratewas evaluated by membrane diffusion method. The liposome suspension equivalent to 1 mg of Oxaliplatin and pegylatednanoliposomaloxaliplatinwas poured into dialysis bags (cut off 12000Da, sigma) separately. The dialysis bags were immersed inside a container containing 25 ml of phosphate buffer, pH 7.4, and left on the magnetic stirrer (37 ºC, 120 rpm, 24h) separately. At certainintervals,1.5 ml of phosphate buffer was taken and replaced with an equal volume of phephosphate buffer. The ODs samples were separately measured by spectrophotometrat wavelength of 210nm.
Evaluation of Apoptosis Rate
Apoptose ratewasevaluated on breast cancer cell lines MCF-7 and MDA-MB-231 byflow cytometry.
Cells cultured in 6-well plates were treated withpoureoxaliplatinand pegylatednano liposomal oxaliplatin with the same amount of oxaliplatin and blank control(untreated cells were used as controls)for 72 h,
The cells were trypsinized, washed(by two washes with cold PBS) and re-suspended in 400 μl of PBS (1 × 106cells/well), and stained with Annexin V-FITC(5 μl) then incubated in the dark for 15 min. Cells were subsequently stained with PI (10 μl) and incubated in the dark for 5 minprior to evaluation by flow cytometry
(Annexin V-FITC at wavelength525nm )( PI at wavelength 631nm)
Result and discussion
Morphology of nanoparticles
Morphology of nanoparticleswas analyzedby using scanning electron microscopy (SEM) (sample hemogenised at 7000 rpm, 5 min and
samplehemogenised at 7000 rpm, 30 min)
The mean diameter of particles in sample hemogenised at 7000 rpm, 5 min was147.7nm(Figure 3). The mean diameter of particles in sample hemogenised at 7000 rpm, 30 min was 60nm(Figure 4).
 |
Figure 3: Representative scanning electron microscopy (SEM) image of nanodrug(sample hemogenised at 7000 rpm, 5 min). |
 |
Figure 4: Representative scanning electron microscopy (SEM) image of nanodrug(sample hemogenised at 7000 rpm, 30 min). |
In Vitro Release Study
The drug release profile indicated controlled release of Oxaliplatinfrom nanoparticles.
The data of released oxaliplatinof pureoxaliplatin and pegylatednanoliposomaloxaliplatin in phosphate buffer were obtained at 24h[10].
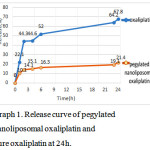 |
Graph 1: Release curve of pegylatednanoliposomaloxaliplatin and pure oxaliplatin at 24h. |
Analysis of Apoptosisrate
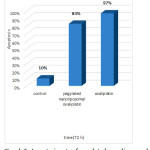
Oxaliplatin-chitosan nanoparticlesinduced more apoptosis rate when compared with freeOxaliplatin and number of apoptotic cells increased in colorectal cancer [11].
Our results exhibited that pegylatednanoliposomaloxaliplatin induced suitable apoptosis ratein breast cancer cell lines MCF-7 and MDA-MB-231 at 72h.
Pegylatednanoliposomaloxaliplatin formulation induced more cancer cell apoptosis in MDA-MB-231 cells when compared with MCF-7 cells.Results can be attributed to the greater uptake of pegylatednanoliposomaloxaliplatin by MDA-MB-231cells in compartion with MCF-7 cells.
 |
Graph 2: Apoptosis rate of pegylatednanoliposomaloxaliplatin and pure oxaliplatin was assessed for MCF-7 cell line by flow cytometry at 72h. |
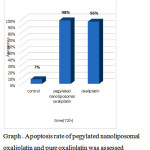 |
Graph 3: Apoptosis rate of pegylatednanoliposomaloxaliplatin and pure oxaliplatin was assessed for MDA-MB-231 cell line by flow cytometryat 72h. |
Conclusion
In this article, novel formulation of oxaliplatin used as a promising methodin order to improve the therapeutic index of breast cancer.
Profile of drug release indicated thatpegylatednanoliposomaloxaliplatinhada slower release in comparison topoureoxaliplatin formulationstherefore help to decrease thecytotoxicityof Oxaliplatin to the normal tissue. Reduction of the release rate of drug can be originated due to the presence of Poly ethylene glycol (PEG).
This studyshowed that pegylatednanoliposomaloxaliplatin induced suitable apoptosis ratein breast cancer MCF-7 and MDA-MB-231cells.
Pegylatednanoliposomaloxaliplatin formulation induced more cancer cell apoptosis in MDA-MB-231 cell line when compared with MCF-7 cell line. Results can be originated due to greater uptake of pegylatednanoliposomaloxaliplatin by MDA-MB-231cell line in compartion with MCF-7 cell line.
This investigation suggests that pegylatednanoliposomaloxaliplatin is a promising strategy for the treatment of breast cancerwith enhancement of therapeutic indexand fewer cytotoxic side effects.
References
- S Ajay ;AS Bhatnagar, Cancer Research Treat., 2007,105, 7-17.
- Kelland L. The resurgence of platinum-based cancer chemotherapy,Nat Rev Cancer,2007, 7(8):573–584.
CrossRef - Chiu SJ, Chao JI, Lee YJ, Hsu TS.Regulation of gamma-H2AXand securin contribute to apoptosis by oxaliplatin via a p38mitogen-activated protein kinase-dependent pathway in humancolorectal cancer cells. Toxicol Lett.,2008;179(2):63–70.
CrossRef - Bangham AD, Standish MM, Watkins JC. Diffusion of univalent ions across the lamellae ofswollen phospholipids.J Mol Biol.1965; 13:238–252. [PubMed: 5859039]
CrossRef - Dunnick JK, Rooke JD, Aragon S, Kriss JP: Alteration of mammalian cells byinteraction with artificial lipid vesicles. Cancer Res1976, 36:2385-2389.
- Poste G, Papahadjopoulos D: Lipid vesicles as carriers for introducingmaterials into cultured cells: influence of vesicle lipid composition onmechanism(s) of vesicle incorporation into cells. ProcNatlAcadSci USA1976, 73:1603-1607
CrossRef - Allen TM, Hansen C, Martin F, Redemann C, Yau-Young A: Liposomes containing synthetic lipid derivatives of poly(ethylene glycol) show prolonged circulation half-lives in vivo. BiochimBiophysActa1991,1066:29-36.
CrossRef - Allen C, Dos Santos N, Gallagher R, Chiu GN, Shu Y, Li WM, Johnstone SA, Janoff AS, Mayer LD, Webb MS, Bally MB: Controlling the physical behavior and biological performance of liposome formulations through use of surface grafted poly(ethylene glycol). Biosci Rep 2002, 22:225-250.
CrossRef - K. Wang, L. Liu, T. Zhang, Y.L. Zhu, F. Qiu, X.G. Wu, X.L. Wang, F.Q. Huang,International Journal of Nanomedicine2011.,6., 3207–3218.
- DelaramAhmadi, AzimAkbarzadeh, Mahdi Arjmand, Amir Heidarinasab,Bulgarian Chemical Communications,2015.,47., 147 – 151.
- A.M. Urbanska, E.D. Karagiannis, G. Guajardo, R.S. Langer, D.G. Anderson,Bio-materials.,201233.,4752–4761.

This work is licensed under a Creative Commons Attribution 4.0 International License.









