Synthesis and Application of Lanio3 Perovskite-Type Nanocatalyst with Zr for Carbon Dioxide Reforming of Methane
Nooshin Talaie1, Moayad Hossaini Sadr2*, Hamidreza Aghabozorg3 and Karim Zare4
1Department of Chemistry, Science and Research branch, Islamic Azad University, Tehran, Iran.
2Department of Chemistry, Shahid Madani University of Azarbaijan, Tabriz, Iran.
3Research Institute of Petroleum Industry, Tehran, Iran.
4Department of Pure Chemistry, Shahid Beheshti University, Tehran, Iran.
Corresponding Author E-mail: sadr@azaruniv.edu
DOI : http://dx.doi.org/10.13005/ojc/320546
La1-xZrxNiO3 perovskite nanocatalysts were synthesized by citrate sol-gel method. The synthesized samples were characterized by using X-ray diffraction (XRD), temperature programmed reduction (TPR), and inductively coupled plasma (ICP) techniques. Surface areas of the compounds were measured by BET method. Morphology study of the nanocatalysts was performed using scanning electron microscopy. The XRD patterns confirmed a well-crystallized structure in doping level up to 0.2 for La1-xZrxNiO3. The SEM images showed that the synthesized particles were in nanoscale. The TPR analysis revealed that by increasing the amount of Zr inthe preparedsamples, reduction process became difficult. The catalytic activity of the nanocatalysts was studied in dry reforming of methane (DRM) with CO2and the H2/CO ratio~1 was reported when doping level was 0.1.
KEYWORDS:Perovskite; Nanocatalyst; catalytic behavior; petrochemical technology; dry reforming of methane(DRM)
Download this article as:| Copy the following to cite this article: Talaie N, Sadr M. H, Aghabozorg H, Zare K. Synthesis and Application of Lanio3 Perovskite-Type Nanocatalyst with Zr for Carbon Dioxide Reforming of Methane. Orient J Chem 2016;32(5). |
| Copy the following to cite this URL: Talaie N, Sadr M. H, Aghabozorg H, Zare K. Synthesis and Application of Lanio3 Perovskite-Type Nanocatalyst with Zr for Carbon Dioxide Reforming of Methane. Orient J Chem 2016;32(5). Available from: http://www.orientjchem.org/?p=21817 |
Introduction
Synthesis gas (syngasH2/CO) is a mixture of H2 and CO that is an important feed used for the petrochemical industry like Fisher-Tropeshprocess. Generally, in syngas cycle with different ratio of H2/CO, variety of products is obtained.
Similarly, several groups have carried out research on the production of syngas from methane. (e.g. the partial oxidation of methane (POM), reaction 1), steam reforming of methane (SRM), reaction 2) and dry reforming of methane (DRM), reaction 3 [1].
POM: CH4+ ½ O2 →CO +2H2(ΔH298K = -22.6 kJ/mol) (1)
SRM: CH4+ H2O → CO +3H2(ΔH298K = 225.4 kJ/mol) (2)
DRM: CH4+ CO2 → 2CO +2H2(ΔH298K = 260.5 kJ/mol) (3)
Syngas economy is considered to be a good process to reduce CO2 and CH4 emission which are both greenhouse gases [2].
In recent years considerable researchers have been focused on DRM reaction to produce syngas [3-5].A lower H2/CO ratio is produced in dry reforming of methane, which favors to liquid fuel production when compared with steam reforming and partial oxidation of methane [6-7]. The reactions that take place in this process are endothermic:
CO2 + H2 ↔ H2O + CO (ΔH298K = 41 kJ/mol) (4)
Catalysts play special role in this reaction, nowadays a considerable attention is focused on the activity and stability of the catalysts.
Recently,provskite oxide structures (ABO3) have been studied widely as catalysts in dry reforming of methane reactions [2]. In the provskite-type oxides, A is (lanthanides, alkali metals and earth alkali metals) and B is (transition metals) where thermal resistance is controlled by Acations and catalytic activity is controlled with B redox cations [1]. However, many researchers have doped transition metals in A-site; and lanthanides, alkali metals and alkali earth metals in B-site [8-10].
There are studies reported on the beneficial effect of noble metal addition such as many studies have been carried out with the aim to synthesize complex mixed metallic oxides with the perovskite structure.A lot of papers have reported different doping level of Ni in B-site of perovskite type oxides that are successful in catalytic activity for reforming of methane [11-14].
For examples, Goldwasser et al. have investigated the influence of Ru partial substitution by Ni and La partial substitution by Ca in LaRuO3 and LaRu0.8Ni0.2O3, respectively, in the combined reforming of methane (CRM) with CO2 and O2, in DRM processes [15]. In another work,Jahangiri et al. have reported the behavior of La1–xSmxNiO3 and LaNi1–xCoxO3 as catalyst precursors in CRM process [16&17]. In addition, they studied the performance of LaNi1-xFexO3perovskite-type oxides with various substitution degrees (x) in the CRM process [18].In the present study, La1-xZrxNiO3 perovskiteswere synthesizedwith different substitution degrees(x) and characterized with conventional methods for investigating the stability and activity of them as a catalyst in the DRM process.
Experimental
Materials and Method
In this research, La(NO3)3.6H2O,(Merck, >99/9%), ZrO(NO3)2.6H2O, (Aldrich, 99/99%), Ni(NO3)2.6H2O (Merck, >99%), citric acid (Merck, 99/5%) and ethylene glycol (99%) were used for synthesis of La1-xZrxNiO3nanocatalysts. Appropriate stoichiometric of La, Ni nitrates and Zirconium oxynitrate solution (1M) were mixed and stirred for 40 minutes, then citric acid and ethylene glycol with molar ratio of 1 were added to thissolution. The mixture was stirred for 8 hours in 80 °C. The resulting gel was dried at110 °C for 24 h and calcined in two steps: 1st at 350 °C for 0.5 h and 2nd at 800 °C for 2 h.
Equipment
The prepared sampleswere characterized by X-ray diffraction (using 3003 PTS diffractometer), with a copper anode (CuKamonochromatized radiation source, λ= 1.5405Å) to ascertainformation of the perovskite structure,phase purityand crystallite size determination. The morphology of the prepared samples was studied by scanning electron microscopy (SEM, using Philips XL30 microscope). BET method was applied for measuring the surface area of the samples using N2 at -200°C(on a Tristar 3000 apparatus) and inductivity coupled plasma (ICP) emission spectroscopy was used (on a Perkin-Elmer ICP/5500 apparatus) for quantitative analysis of the samples. Temperature programmed reduction (TPR, on a 29000 apparatus) was performed for measuring of hydrogen consumption.
Catalytic Activity Evaluation
Catalytic activity of the samples was studied under 600 to 800 °C. The reactants and products were analyzed by a gas chromatograph (Model 6890N, Agilent Technologies) provided with the thermal conductivity detector (TCD). The experimental tests for the catalyst activity study were carried out using the feed gases (CH4, CO2, N2 and H2) with ultra-high pure grade (>99.999%) in a micro-reactor equipped with mass flow controllers (Model 5850, Brooks Instrument).
The temperature of micro-reactor was measured and controlled by two thermocouples (Ni–Cr, K-Type, 0.5 mm diameter) and two PID thermo-controllers (Model JumoiTRON08). The catalyst (0.4 g for all cases) was loaded in the middle of the reactor, and the feed gases at a total flow rate of 100 ml/min (WHSV=15 l/(h.g), CH4/CO2= 1/1) under atmospheric pressure were introduced into the reactor as the defaultprior to activity measurements.Catalyst precursors were reduced in situ in a flow of 20% H2 in N2 (total flow rate of 50 ml/min) at 700 °C for 2 h to generate the metal phase. In all tests, the performances wereevaluated by conversions. The CH4 and CO2 conversions, H2 and CO yields and H2/CO ratio are defined as follow[7]:
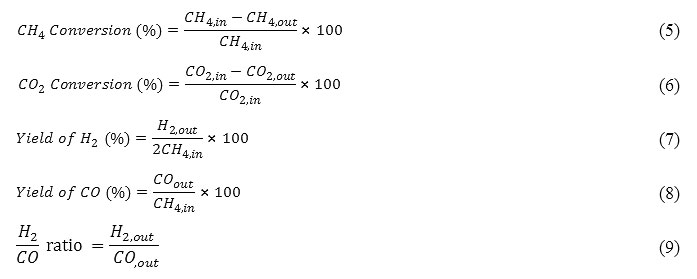
Results and Discussions
Crystalline Structure
The XRD patterns of the synthesizedsamples are shown in Fig. 1. The XRD patterns of Zr doped LaNiO3indicated that Zr doping level (x) in these samples were 0.2 with formation of well-crystallized perovskite structure without any impurities(JCPDS No: 33-710).For Zr doped LaNiO3sample, beyond x=0.2 additional peaks are presented which belongs to ZrO2, La2Zr2O7 and NiO compounds.
 |
Figure 1: XRD patterns of La1-xZrxNiO3nanocatalysts |
For calibrating the position of the reflections, a quartz standard was used. Since all the prepared samples could be indexed with cubic symmetry of the ABO3 type, the cell parameter (a) in the unit cell of these compounds was calculated using the following equation:
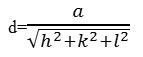
The obtained results are shown in Table 1. The results indicatethatby increasing the amountof Zr in the La1-xZrxNiO3 samples, the cell parameter decreases.
Table 1: Cell parameter (a) of the prepared samples obtained from XRD data
|
La1-xZrxNiO3 |
|
|
X |
Cell parameter (a) (Å) |
| 0 |
3.888 |
| 0.1 |
3.867 |
| 0.2 |
3.864 |
| 0.3 |
3.855 |
| 0.5 |
3.854 |
The crystallite size of the preparedsamples calculated by the Scherrer equation using the intense peak is presented in Table 2 [19].Thistableshows that the preparedparticlesare appeared in nanoscale.
Table 2: Elemental analysis*, BET surface areas and crystallite size of some calcinedLa1-xZrxNiO3perovskites (calculated by Scherrer equation)
|
La1-xZrxNiO3 |
|||||
|
X |
La (wt.%) |
Zr (wt.%) |
Ni (wt.%) |
SA (m2/g) |
D (nm) |
|
0 |
**(3.07)8.96 |
(00.0)00.0 |
(29.7) 30.2 |
3 |
50 |
|
0.1 |
65.0 (64.8) |
4.5(4.8) |
30.5(30.4) |
5 |
53 |
|
0.3 |
50.1 |
16.8 |
33.1 |
3 |
45 |
|
0.5 |
38.3 |
29.6 |
32.1 |
2 |
43 |
| * Calculated by ICP | ** Nominal values are in parentheses | ||||
Chemical Analysis and Surface Area Measurements
The chemical composition (wt.%) and surface area for some prepared catalysts are given in Table 2.As it is shown, the experimental data for Ni, Zrand La (wt.%) are close to the nominal values (reported in parentheses). BET surface areas of the catalysts are found in the range of 3-5m2/g. Since preparation of perovskite oxides by modified citrate sol-gel method, require long exposure to high temperatures, leading to low surface area solids is not surprising. Similar behavior has also been reported by Pahlevanzadeh et al. [17].
Morphology
The SEM images of the samples are shown in Fig 2. These images have shown that the synthesized particles were in nanoscale which is nearly in spherical shape. For
La1-xZrxNiO3 samples, morphology study of the particles with doping level up to 0.2 is uniform which indicate that these samples are crystallized in mono phase perovskite.
 |
Figure 2: SEM images of the La1-xZrxNiO3 (x=0 (A), x=0.1 (B), x= 0.2 (C), x=0.3 (D) and x=0.5 (E)). |
TPR profiles of the prepared samples are shown in Fig.3. TPR study of the samples displays two main steps of hydrogen consumption. The first one indicates the reduction of Ni3+ to Ni2+ with a maximum consumption of hydrogen in around 382°C [18]. The second step can be related to Ni2+ to Ni0 reduction in around 570°C [20]. Thistrend is usual for perovskite type samples and confirmsthatnickelispresent in the B-site. In addition, for Zr doped LaNiO3 sample, both steps shifted to higher temperatures which can be due to the presence of Ni-Zr-O system [21]. This result confirms the doping level of Zr which is obtained by XRD patterns.
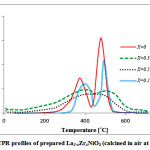 |
Figure 3: TPR profiles of prepared La1-xZrxNiO3(calcined in air at 800 °C) |
Catalytic Activity
Catalytic activity of the prepared samples in the temperature range of 600-800 °C has been studied in DRMprocess. The CH4and CO2conversions and product yields in the presence of La1-xZrxNiO3 in different temperatures are presented in Fig. 4. These figures show that CH4and CO2 conversions increase by increasing thetemperature in the presence of all catalysts. However, the CH4 conversions are lower than those of CO2. This behavior could be due to consuming of CO2 in reaction (4). In addition, both the H2and CO yields increase with increasing the reaction temperature in the presence of the prepared catalysts, while the CO yields are higher than the H2yields. This effect is more notable at higher temperatures, which are in consistent with Aghabozorg et al. study [16].
In the presence of La1-xZrxNiO3 samples with doping levels of x=0.3 and 0.5, there is no outstanding difference in the CH4and CO2conversions and product yields for each temperature. These results confirm that the mono phase pesrovskite structure shift to other metal oxide phases for doping level of x= 0.3 and 0.5 which is in consistent with XRD and TPR results.
Catalytic activity study of the prepared samples also indicate that H2/CO ratio in the presence of the catalyst with doping level of x=0 and x= 0.1 is ~1, while this ratio for upper doping level is less than 1. In other words, it can be concluded that the reactions (3) and (4) are the main occurring reactions. These results indicate that perovskite structures are favorable for producing more H2 that is in accordance with Valderrama et al. research [22].
Fig. 5 indicates the CH4 and CO2 conversions and H2 and CO yields versus time at 750 °C for La1-xZrxNiO3 samples. This figure shows that the CH4, CO2 conversions and H2, CO yields in the presence of the pure perovskite are more than those of catalysts containing more than one phase. However, these parameters for LaNiO3 are more observable than that of the samples containing Zr. The partial substitution of La3+ by Zr4+ in La1-xZrxNiO3 structure is undesirable for Ni reduction and leads to decreasing the mobility of the oxygen toward the surface of the solid[22].
Fig. 5 (H2 and CO yields) indicates that by letting time elapse, the yields in the presence of
La1-xZrxNiO3are decreased. However, since the extent of yield decrease roughly follows the same way for both CO and H2 yields, the H2/CO ratio remain constant (Fig. 6).
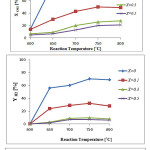 |
Figure 4: CH4 and CO2 conversions, and H2 and CO yields as a function of the reaction temperatureforLa1-xZrxNiO3samples in DRM process (CH4/CO2= 1/1, WHSV=15 l/(h.g)).
|
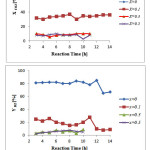 |
Figure 5: CH4 and CO2 conversions, H2 and CO yields as a function of the reaction time for La1-xZrxNiO3samples at 750 °C in DRM process (CH4/CO2= 1/1, WHSV=15 l/(h.g)). Click here to View Figure |
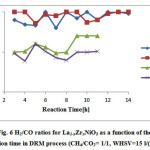 |
Figure 6: H2/CO ratios for La1-xZrxNiO3 as a function of the reaction time in DRM process (CH4/CO2= 1/1, WHSV=15 l/(h.g)).
|
Conclusions
The XRD patterns of Zr doped LaNiO3indicated that Zr doping level (x) in these samples was 0.2 with formation of well-crystallized perovskite structure in nanometer scale without any impurities.
The cell parameter calculation obtained from XRD data showed that, by increasing the amount of Zr in the La1-xZrxNiO3 samples, the cell parameter decreased.
TPR study of the prepared samples displayed two main steps of hydrogen consumption. The first one indicated the reduction of Ni3+ to Ni2+. The second step was related to Ni2+ to Ni0 reduction in around 570°C. Also TPR analysis revealed that by increasing the amount of Zr inthe preparedsamples reduction process became difficult which could be due to presence of different forms of oxides other than perovskite type in the samples.
CH4and CO2 conversions increased by increasing thetemperature in the presence of all catalysts. However, the CH4 conversions were lower than that of CO2.
The CH4and CO2conversions and H2and CO yields for the pure perovskite catalysts were more than those catalysts containing more than one phase.
Global warming by greenhouse gases such as carbon dioxide (CO2) and methane (CH4) is one of the most important problems in all over the world. In recent years, many researches aimed to solve this problem and promote hydrogen production technology such as CO2 reforming of methane generally called (DRM). In this study we investigate La1-xZrxNiO3nanostructures and its catalytic behavior indry reforming of methane and supposed to find out more applicable perovskitenanocatalyst in this process.
References
- Asencios Y JO, Assaf EM. Fuel Process Technol.2013, 106, 247-252.
- Goldwasser MR, Rivas ME, Pietri E, Perez-Zurita MS, Cubeiro ML, Gingembre L, Leclercq L, and Leclercq G. J MolCatal A Chem.2005,228, 325-331.
- Ferreira-Aparicio P, Guerrero-Ruiz A, and Rodriguez-Ramos I. Appl. Catal. Gen.1998,170 (1), 177-187.
- Khalesi A, Arandiyan H. R, and Parvari M. Chin. J. Catal.2008,29 (10), 960-968.
- Valderrama G, Kiennemann A, and Goldwasser M.R.J. Power Sources.2010, 195(7), 1765-1771.
- Pereniguez R, Gonzalez-Delacruz VM, Holgado J P, and Caballero A. Appl. Catal. B.2010,93(33), 346-353.
- Dejaidja A, Libs S, Kiennemann A, and Barama A. Catal. Today.2006,113(3), 194-200.
- Chendong Zuo, Doris SE, Balachandran U and Meilin Liu. Chem. Mater. 2006, 18, 4647-4650.
- Dhivya P, Prasad AK, Sridharan M. Sensor Actuat, B: Chem. 2016,222, 987-993.
- Balamurugan C, Lee D-W. Sensor Actuat, B: Chem. 2015, 221, 857-866.
- Lima SM, Assaf JM, Pena MA, Fierro JLG.Appl. Catal. A: Gen.2006, 311, 94-104.
- Gallego German S, Marin Jaime G, Batiot-Dupeyrat C, Barrault J, Mondragon F. Appl. Catal A: Gen.2009, 369, 97-103.
- Gallego German S, Mondragon F, Barrault J, Tatibouet J-M, Batiot-Dupeyrat C.Appl. Catal A: Gen.2006,311, 164-171.
- Arandian HR, Li J, Lei Ma, hashemnejad SM, Mirzaei MZ, Chen J et al. J IndEng Chem.2012,18, 2103-2114.
- Goldwasser MR, Rivas ME, Pietri E, Perez-Zurita MS, Cubeiro ML, Gingembre L, Leclercq L, and Leclercq G. Appl. Catal. Gen.2003, 225, 45-47.
- Jahangiri A, Pahlavanzadeh H, and Aghabozorg HR. Int J Hydrogen Energy. 2012,37, 9977-9984.
- Jahangiri A, Aghabozorg HR, Pahlavanzadeh H, and Towfighi J. Int J Chem React Eng. 2014,12, 1–10.
- Jahangiri A, Aghabozorg HR, and Pahlavanzadeh H. Int J Hydrogen Energy. 2013,38, 10407-10416.
- Spinicci R, Marini P, De Rossi S, Faticanti M, Porta P. J MolCatal A Chem. 2001,176, 253-265.
- Yang Eh, Young Kim N, Noh Ys , Soo Lim S, Sun Jung J, Suk Lee J, Ju Moon D.Int J Hydrogen Energy.2015,40, 11831-11839.
- Barbero J, Pena M.A, Campos-Marthin J.M, Fierro J.L.G, and Arias P.L..Catal letters. 2003, 87, 211-218.
- Valderrama G, Goldwasser MR, Navarro CU, Tatibouët JM, Barrault J, Dupeyrat CB, and Martinez F.Catal Today. 2005,107–108, 785–791.

This work is licensed under a Creative Commons Attribution 4.0 International License.









