Preparation of Stereocomplex Polylactide Bioplastics from Star-shaped/Linear Polylactide Blending
Supasin Pasee, Onanong Cheerarot and Yodthong Baimark*
Biodegradable Polymers Research Unit, Department of Chemistry and Center of Excellence for Innovation in Chemistry, Faculty of Science, Mahasarakham University, Mahasarakham 44150, Thailand. Corresponding Author. E-mail: yodthong.b@msu.ac.th
DOI : http://dx.doi.org/10.13005/ojc/310333
Article Received on :
Article Accepted on :
Article Published : 15 Sep 2015
The stereocomplex polylactide (scPLA) films of star-shaped poly(L-lactide) (PLLA)/linear poly(D-lactide) (1PDLA) blends were prepared by solution blending before film casting. The influences of the PLLA/1PDLA blend ratios (75/25, 50/50 and 25/75 w/w), 1PDLA molecular weights (30,000 and 60,000 g/mol) and PLLA arm numbers (1-, 4- and 16-arm) on the crystallinity and mechanical properties of the scPLA films were investigated. The stereocomplex crystallinity (csc), stress at break and elongation at break of the scPLA films were the highest in the 50/50 (w/w) blend ratio. The csc of the scPLA films decreased and the mechanical properties slightly increased as the 1PDLA molecular weight increased. For the same blend ratio and 1PDLA molecular weight, the scPLA films prepared from the 16PLLA blending had the highest csc values.
KEYWORDS:Bioplastics; Stereocomplex polylactides; Star-shaped polymers; Thermal properties; Mechanical properties
Download this article as:| Copy the following to cite this article: Pasee S, Cheerarot O, Baimark Y. Preparation of Stereocomplex Polylactide Bioplastics from Star-shaped/Linear Polylactide Blending. Orient J Chem 2015;31(3). |
| Copy the following to cite this URL: Pasee S, Cheerarot O, Baimark Y. Preparation of Stereocomplex Polylactide Bioplastics from Star-shaped/Linear Polylactide Blending. Orient J Chem 2015;31(3). Available from: http://www.orientjchem.org/?p=10895 |
Introduction
Poly(L-lactide) (PLLA) is a biodegradable semi-crystalline polylactide (PLA) that has attracted much interest for bioplastic and biomedical applications because of its renew ability and biodegradability1-4. However, poor mechanical properties, non-heat resistance and slow crystallization of the PLLA products are major disadvantages for bioplastic applications, but they have been improved by blending with some additives5-6 and the formation of stereocomplex polylactides (scPLAs)7.
In the past few decades, scPLAs prepared from linear PLLA/linear poly(D-lactide) (PDLA) blending have been extensively investigated7. The PDLA is an optical isomer of PLLA. The homocrystallites of both PLLA and PDLA have melting temperatures (Tms) about 180 °C. The Tm of the scPLAs or PLLA/PDLA blends (Tm » 230 °C) are approximately 50 °C higher than the Tm of both the original PLLA and PDLA, due to the strong van der Waals interactions in the stereocomplex crystallites8-10. The thermal resistance and mechanical properties of the scPLA products were better than the PLLA products7, 11-12. However, scPLA with controllable properties are still required for each specific process and application.
PLAs with block13 and star-shaped14-17 structures have been used to prepare scPLAs. However, to the best of our knowledge, the influences of arm number and molecular weight of the star-shaped PLAs on the stereocomplex crystallinity and mechanical properties of the scPLAs are still not clear. Thus the purpose of this work is to investigate the effects of the blend ratio, PDLA molecular weight and arm number of the PLLA on the thermal and mechanical properties of star-shaped PLLA/linear PDLA blends. For this purpose, 1-, 4- and 16-arm PLLA were synthesized for blending with 1-arm PDL As with molecular weights of 30,000 and 60,000 g/mol.
Experimental
Materials
The L-lactide and D-lactide monomers were synthesized by polycondensation followed by thermal decomposition of L-lactic acid (88%, Purac, Thailand) and D-lactic acid (90%, Haihang Industry Co., Ltd., China), respectively. The both L- and D-lactides were purified by repeated re-crystallization four times from ethyl acetate before drying for 48 h in a vacuum oven at 55 °C. 1-dodecanol (98%, Fluka, Switzerland) with one-hydroxyl end group was purified by distillation under reduced pressure before use for preparing the linear polylactides. Pentaerythritol (99%, Aldrich, USA) and Boltorn H20 containing 4- and 16-hydroxyl end groups, respectively were dried for 48 h in a vacuum oven at 55 °C before use as the initiators for synthesis of the star-shaped polylactides. Stannous octoate (95%, Sigma, USA), Sn(Oct)2, was used without further purification. All reagents used were analytical grade.
Synthesis of Polylactides
The poly(L-lactide) (PLLA) and poly(D-lactide) (PDLA) were synthesized by ring-opening polymerization of L-lactide and D-lactide, respectively, in bulk at 165°C for 2 h under a nitrogen atmosphere using 0.01 mol% Sn(Oct)2 as a catalyst. The 1-arm linear, 4-arm star-shaped and 16-arm star-shaped polylactides were prepared using 1-dodecanol, pentaerythritol and Boltorn H20 as initiators, respectively. The resulting polylactides were purified via dissolution in chloroform before being precipitated in cool n-hexane and dried to a constant weight in a vacuum oven at 50 °C.
The 1-arm (1PLLA120K) and 4-arm (4PLLA120K) PLLAs with a theoretical number-average molecular weight of 120,000 g/mol were synthesized using 0.12 mol% 1-dodecanol and 0.12 mol% pentaerythritol, respectively. The 16-arm PLLA with a theoretical number-average molecular weight of 480,000 g/mol designed as 16PLLA480K was synthesized using a 0.03 mol% Boltorn H20. The 1-arm PDLAs with theoretical number-average molecular weights of 30,000 and 60,000 g/mol were prepared and designed as 1PDLA30K and 1PDLA60K, respectively. 1-dodecanol initiators at 0.48 and 0.24 mol% were used to synthesis the 1PDLA30K and 1PDLA60K, respectively. The molecular weight characteristics of these polylactides were summarized in Table 1.
Table 1: Molecular weight characteristics of polylactides.
|
Polylactides |
Arm number |
Theoretical Mn (g/mol) a |
Mn of arm (g/mol) b |
[h] (dL/g) c |
Mv (g/mol) d |
|
1PLLA120K 4PLLA120K 16PLLA480K 1PDLA30K 1PDLA60K |
1 4 16 1 1 |
120,000 120,000 480,000 30,000 60,000 |
120,000 30,000 30,000 30,000 60,000 |
2.50 1.54 1.90 1.02 1.50 |
103,700 – e – e 30,400 51,500 |
aCalculated from ratio of weight of lactide (g)/mole of initiator (mol).
bCalculated from ratio of theoretical Mn/arm number.
cDetermined from PLA solution in chloroform at 25 °C.
dCalculated from equation (1).
eMv of the star-shaped PLAs could not be calculated.
Preparation of Stereocomplex Polylactide Films
The stereocomplex polylactide films of 1PLLA120K/1PDLA, 4PLLA120K/1PDLA and 16PLLA480K/1PDLA blends were prepared via solution blending followed by solvent evaporation. The PLLA and PDLA were dissolved separately in chloroform at a concentration of 1 g/dL and then mixed together under vigorous stirring for 30 min. The blend solution was then poured onto a glass Petri dish followed by solvent evaporation at 40 °C in an air oven for 24 h. The film was then dried in a vacuum oven at 70 °C for 48 h to remove any residue solvent. The blend films with PLLA/1PDLA blend ratios of 75/25, 50/50 and 25/75 (w/w) were prepared. The 1PLLA, 4PLLA, 16PLLA and 1PDLA films were also prepared by the same method for comparison.
Characterization of Polylactides and Stereocomplex Polylactide Films
The intrinsic viscosities, [h], of PLLA and PDLA were determined by flow-time measurements from a diluted series of solutions in chloroform at 25 °C using an Ubbelohde viscometer. The viscosity-average molecular weight (Mv) of polylactides was calculated from equation (1)18.
[η]= 5.45 × 10-4Mv0.73 (1)
The thermal transition properties of the polylactides and their stereocomplex polylactide films were investigated using a Differential Scanning Calorimeter (DSC; Perkin-Elmer Pyris Diamond) under a nitrogen flow. For DSC, 5 – 10 mg of sample was heated at 10 oC/min over a temperature range of 0 to 250 °C (the 1st heating scan) to observe the melting temperature (Tm). Then the samples were quenched to 0 °C according to the DSC instrument’s own default cooling mode before heating from 0 to 250 °C (the 2nd heating scan) to observe the glass transition (Tg) and crystallization (Tc) temperatures. The homocrystallinity (χhc) and stereocomplex crystallinity (Xsc) of the polylactides were calculated from the heats of melting of the homocrystallites (∆Hm, hc) and stereocomplex crystallites (∆Hm, sc) using equations (2) and (3), respectively.
Xhc (%) = [∆Hm, hc/93.7] ´ 100 (2)
Xsc (%) = [∆Hm, sc/142] ´ 100 (3)
where 93.7 J/g and 142 J/g are the theoretical heats of melting for 100% homocrystallinity and 100% stereocomplex crystallinity, respectively6, 19.
Fourier Transform Infrared spectroscopy (FTIR; Spectrum GX, Perkin Elmer) was used to study stereocomplex formation of polylactide blends. The scan was conducted in the range of 500 – 2000 cm-1 at a resolution of 1 cm-1. The number of scans was set to 32. Air was used as a reference.
Mechanical properties including stress at break, elongation at break and initial Young’s modulus, of the PLLA, PDLA and stereocomplex polylactide films were investigated at 25 °C and 65% relative humidity using a Lloyds LRX+ Universal Mechanical Testing Machine. Film samples with 10 ´ 80 mm sizes were tested with a gauge length of 25 mm and a crosshead speed of 20 mm/min. The mechanical properties were determined from an average of three replicate films.
The data were expressed as mean ± SD. Statistical analysis was performed using a one-way Analysis of Variance (ANOVA).
Results and Discussion
Thermal transition properties
Thermal transition properties including melting temperature (Tm), glass transition temperature (Tg) and crystallizing temperature (Tc) of the polylactides (PLAs) and stereocomplex polylactide (scPLA) films were determined from DSC thermograms, examples of which are shown in Fig. 1 for the 16PLLA480K/1PDLA blend films. The Tms of the homocrystallites (Tm, hc) and stereocomplex crystallites (Tm, sc) of the film samples were detected from the first heating scan DSC thermogram [Fig. 1(a)]. While the Tg and Tc of the films were detected from the second heating scan DSC thermogram [Fig. 1(b)].
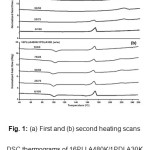 |
Figure 1: (a) First and (b) second heating scans DSC thermograms of 16PLLA480K/1PDLA30K blend films prepared with different blend ratios. Click here to View figure |
The homopolymeric films of 1PLLA120K, 4PLLA120K, 16PLLA480K, 1PDLA30K and 1PDLA60K showed a single Tm of homocrystallites in the range 171 – 175 °C. All PLLA/PDLA blend films exhibited both lower Tm of homocrystallites and higher Tm of stereocomplex crystallites, except the 50/50 w/w 16PLLA480K/1PDLA30K blend film, that only had a Tm of stereocomplex crystallites. This may be explained by the high molecular weight PLLAs (1PLLA120K, 4PLLA120K and 16PLLA480K) blending with the medium (1PDLA30K) and high (1PDLA60K) molecular weight 1PDLAs induced both homocrystallization and stereocomplex crystallization20. However, the homocrystallization of PLA was not obtained for the 50/50 w/w 16PLLA480K/1PDLA30K blend film.
The crystallinities of the homocrystallites (Xhc) and stereocomplex crystallites (csc) of the PLLA/1PDLA blend films calculated from equations (2) and (3), respectively, are compared in Figs. 2 and 3, respectively. It can be seen that the chc of 4PLLA120K/1PDLA and 16PLLA480K/1PDLA blend films were lower than their PLLA and PDLA films and similar to the 1PLLA120K/1PDLA blend films (Fig. 2). The 50/50 w/w PLLA/PDLA blend films showed the lowest Xhc. The equimolar of PLLA and PDLA blending showed better stereocomplex formation than the non-equimolar blending20. It should be noted that the Xhc of the blend films slightly increased as the molecular weight of 1PDLA increased from 30K to 60K.
 |
Figure 2 Click here to View figure |
The PLLA/PDLA blend films showed the highest csc values in the 50/50 w/w blend ratio (Fig. 3). The csc values of 4PLLA120K/1PDLA blend films in all blend ratios (25/75, 50/50 and 25/75 w/w) and both 1PDLA molecular weights (1PDLA30K and 1PDLA60K) were lower than the 1PLLA120K/1PDLA blend films. This indicates that the 4-arm star-shaped structure of 4PLLA120K inhibited stereocomplex formation. However, the csc values of 16PLLA480K/1PDLA30K blend films in all blend ratios were higher than the 1PLLA120K/1PDLA blend films. The results suggest that the 1PDLA30K molecules could penetrate into the highly branched structures of 16PLLA molecules to form strong van der Waals forces. This then induces complete stereocomplex formation. But the csc values of the 16PLLA480K/1PDLA60K blend films were nearly the same as the 1PLLA120K/1PDLA blend films. This suggests that the stereocomplex formation was inhibited when the molecular weight of 1PDLA increased from 30,000 to 60,000 g/mol.
 |
Figure 3 Click here to View figure |
In addition, these PLLA/1PDLA blend films had similar Tg and Tc values in the ranges 55 – 62 °C and 93 – 106 °C, respectively. The results suggested that the PLLA arm number, blend ratio and 1PDLA molecular weight did not significantly affect the Tg and Tc values of scPLA films.
FTIR Spectroscopy
The stereocomplex formation of linear PLLA/linear PDLA blends was evaluated by FTIR spectroscopy21. The absorbance band with a wave number in the range 920 – 925 cm-1 is attributed to the coupling of C-C backbone stretching with the CH3 rocking mode, and is considered as the PLLA and PDLA homocrystallites. This absorbance band was shifted to a lower wave number (910 – 915 cm-1) when the stereocomplex crystallites of PLAs were formed.
Fig. 4 shows the FTIR spectra of 16PLLA480K/1PDLA blend films. The 16PLLA480K, 1PDLA30K and 1PDLA60K films showed C-C stretching bands of homocrystallites in the range 920 – 921 cm-1. Meanwhile the 16PLLA480K/1PDLA blend films in all blend ratios exhibited lower wave number bands of stereocomplex crystallites in the range 909 – 915 cm-1. The C-C stretching bands of the 1PLLA120K/1PDLA and 4PLLA120K/1PDLA blend films were also shifted to lower wave numbers similar to the 16PLLA480K/1PDLA blend films. The FTIR results supported that the stereocomplex formation of PLLA/PDLA blend films had occurred21.
 |
Figure 4: FTIR spectra of 16PLLA480K/1PDLA30K blend films prepared with different 1PDLA molecular weights and blend ratios. Click here to View figure |
Mechanical Properties
The mechanical properties of PLA and scPLA films were analyzed from tensile testing. The tensile stress and elongation at break of the PLA and scPLA films are illustrated in Figs. 5 and 6, respectively. The Young’s modulus of all PLA and scPLA films are values in the range 1,000 – 1,500 MPa.
It was found that the tensile stress and elongation at break of the scPLA films were higher than those of their PLLA and 1PDLA films. The stronger van der Waals forces of the stereocomplex crystalline structure induced higher mechanical properties8-10. The 50/50 (w/w) PLLA/1PDLA blend films exhibited the highest tensile stress and elongation at break similar to a previous report7. This may be due to the 50/50 (w/w) PLLA/1PDLA blend films having the highest csc (see Fig. 3). It should be noted that the stress and elongation at break of the scPLA films slightly increased as the molecular weight of 1PDLA increased. It is well known that the mechanical properties of polymeric films are directly related to their molecular weight.
 |
Figure 5 |
 |
Figure 6 |
In addition, the mechanical properties of the 1PLLA120K/1PDLA, 4PLLA120K/1PDLA and 16PLLA480K/1PDLA blend films were similar for the same blend ratios and 1PDLA molecular weights. From Fig. 3, the 16PLLA480K/1PDLA blend films had higher csc values than both 1PLLA120K/1PDLA and 4PLLA120K/1PDLA blend films. This suggests that the stereocomplex formation did not have a significant effect on the mechanical properties of the PLLA/PDLA blend films.
Conclusions
The chc and csc of PLLA/1PDLA blend films depended upon the blend ratio, molecular weight of 1PDLA and arm number of PLLA. The equimolar PLLA/1PDLA blend ratio (50/50 w/w) had the lowest chc and highest csc values. The chc of scPLA films increased and csc decreased as the molecular weight of 1PDLA increased. The csc values of 4PLLA/1PDLA blend films were lower than the 1PLLA/1PDLA blend films but their chc were similar for the same blend ratio. The 16PLLA/1PDLA blend films had lower chc and higher csc than both the 1PLLA/1PDLA and 4PLLA/1PDLA blend films. The results suggested that the stereocomplexation content of 16PLLA/1PDLA blend films was higher than both the 1PLLA/1PDLA and 4PLLA/1PDLA blend films. The FTIR results confirmed the stereocomplexation of PLLA/1PDLA blend films.
The stress and elongation at break of the scPLA films strongly depended upon the blend ratio and molecular weight of 1PDLA, but not on the arm number of PLLA. The 50/50 w/w PLLA/1PDLA blend films showed the highest stress and elongation at break. The mechanical properties of the scPLA films increased with the molecular weight of 1PDLA. The effects of blend ratio, PLLA arm number and molecular weight of 1PDLA of the star-shaped PLLA/linear PDLA blends on their thermal resistance and thermo-mechanical properties will require further investigations.
Acknowledgements
This work was supported by Mahasarakham University, fiscal year 2014 and the Center of Excellence for Innovation in Chemistry (PERCH-CIC), Office of the Higher Education Commission, Ministry of Education, Thailand.
References
- Vink, E.T.H.; Rabago, K.R.; Glassner, D.A.; Gruber, P.R., Applications of life cycle assessment to NatureWorksTM polylactide (PLA) production. Polym. Degrad. Stab., 2003; 80:403-419.
- Lim, L.T.; Auras, R.; Rubino, M., Processing technologies for poly(lactic acid). Prog. Polym. Sci., 2008; 33:820-852.
- Baimark, Y.; Cheerarot, O., Effect of chain extension on thermal stability behaviors of polylactide bioplastics. Orient. J. Chem., 2015; 31:635-641.
- Vashist, A.; Sharif, A., Hydrogel: smart materials for drug delivey. Orient. J. Chem., 2013; 29:861-870.
- Saetdlou, S.; Huneault, M.A.; Li, H.; Park, C.B., Poly(lactic acid) crystallization. Prog. Polym. Sci., 2012; 37:1657-1677.
- Liu, X.; Wang, T.; Chow, L.C.; Yang, M.; Mitchell, J.W., Effects of inorganic fillers on the thermal and mechanical properties of poly(lactic acid). Int. J. Polym. Sci., 2014; 2014:Article ID 827028.
- Tsuji, H., Poly(lactide) stereocomplexes: formation, structure, properties, degradation, and applications. Macromol. Biosci., 2005; 5:569-597.
- Kelly, S.A.; Hillmyer, M.A., Melt preparation and nucleation efficiency of polylactide stereocomplex crystallites. Polymer, 2006; 47:2030-2035.
- He, Y.; Xu, Y.; Wei, J.; Fan, Z.; Li, S., Unique crystallization behavior of poly(L-lactide)/poly(D-lactide) stereocomplex depending on initial melt states. Polymer, 2008; 49:5670-5676.
- Chen, D.; Li, J.; Ren, J., Crystal and thermal properties of PLLA/PDLA blends synthesized by direct melt polycondensation. J. Polym. Environ., 2011; 19:574-581.
- Li, S.; Vert, M., Synthesis, characterization, and stereocomplex-induced gelation of block copolymers prepared by ring-opening polymerization of L(D)-lactide in the presence of poly(ethylene glycol). Macromolecules, 2003; 36:8008-8014.
- Fukushima, K.; Chang, Y.H.; Kimura, Y., Enhanced stereocomplex formation of poly(L-lactic acid) and poly(D-lactic acid) in the presence of stereoblock poly(lactic acid). Macromol. Biosci., 2007; 7:829-835.
- Tsuji, H.; Wada, T.; Sakamoto, Y.; Sugiura, Y., Stereocomplex crystallization and spherulite growth behaviour of poly(L-lactide)-b-poly(D-lactide) stereodiblock copolymers. Polymer, 2010; 51:4937-4947.
- Biela, T.; Duda, A.; Penczek, S., Enhanced melt stability of star-shaped stereocomplexes as compared with linear stereocomplexes. Macromolecules, 2006; 39:3710-3713.
- Shao, J.; Sun, J.; Bian, X.; Cui, Y.; Li, G.; Chen, X., Investigation of poly(lactide) stereocomplexes: 3-armed poly(L-lactide) blended with linear and 3-armed enantiomers. J. Phys. Chem. B, 2012; 116:9983-9991.
- Isono, T.; Kondo, Y.; Otsuka, I.; Nishiyama, Y.; Borsali, R.; Kakuchi, T.; Satoh, T., Synthesis and stereocomplex formation of star-shaped stereoblock polylactides consisting of poly(L-lactide) and poly(D-lactide) arms. Macromolecules, 2013; 46:8509-8518.
- Purnama, P.; Jung, Y.; Kim, S.H., Melt stability of 8-arms star-shaped stereocomplex polylactide with three-dimensional core structures. Polym. Degrad. Stab., 2013; 98:1097-1101.
- Hyon, S.H.; Jamshidi, K.; Ikada, Y., Synthesis of polylactides with different molecular weights. Biomaterials, 1997;18: 1503-1508.
- Liu, Y.; Shao, J.; Sun, J.; Bian, X.; Feng, L.; Xiang, S.; Sun, B.; Chen, Z.; Li, G.; Chen, X., Improved mechanical and thermal properties of PLLA by solvent blending with PDLA-b-PEG-b-PDLA. Polym. Degrad. Stab., 2014; 101:10-17.
- Tsuji, H.; Hyon, S.H.; Ikada, Y., Stereocomplex formation between enantio- meric poly(lactic acid)s. Macromolecules, 1991; 24:5651-5656.
- Ahmed, J.; Varshney, S.K.; Janvier, F., Rheological and thermal properties of stereocompled polylactide films. J. Therm. Anal. Calorim., 2014;115: 2053-2061.

This work is licensed under a Creative Commons Attribution 4.0 International License.










