Ag/ZnO Nanoparticles as Novel Antibacterial Agent against of Escherichia coli infection, in vitro & in vivo
1Department of Microbiology,Faculty of Basic Sciences, Islamic Azad University, Ardabil Branch, Iran 2Anti-microbial Resistance Research Center,Rasoul-e-Akram Hospital, Iran University of Medical Sciences, Tehran, Iran 3Department of Medical Nanotechnology, School of Advanced Technologies in Medicine, Tehran University of Medical Sciences, Tehran, Iran.
Corresponding Author E-mail: Jafari.ar@iums.ac.ir
DOI : http://dx.doi.org/10.13005/ojc/310322
Article Received on :
Article Accepted on :
Article Published : 25 Jul 2015
Urinary tract infection has change a basic challenge in human population. Antibacterial nanoparticles have turned over the new leaf in scientist's research.Ag/ZnO nanoparticleis synthesis in high temperature (5000C).FT-IR, XRD, SEM and TEM were used for determination of spectroscopic, structural and morphology of samples, respectively. Also the nanoparticle was digested and analyzed by ICP-AES for determining the presence of residual chemical element in the nanoparticle. Bacterial sensitivity to nanoparticle was commonly tested using by MIC and MBC tests. For achievement of in vivo tests, the renal of rats were injected and then were infected with E.coli. After that, animals were divided to 9 treatment group and the effect of three concentrations of MIC and MBC; also intermediate concentrations of MIC and MBC and each as a separate group, were studied.The particles size was less of 12nm, approximately. The MIC and MBC observed for Ag/ZnO were 32µgml-1and 512µgml-1 for E.coli, respectively.Treatment with a concentration 512µgml-1ofAg/ZnO nanoparticle in combination with Ciprofloxacin reduced severity of renal cortical thickness of pyelonephrit is rats.The microscopic pathological examination indicated that there was no any severe Inflammation of renal tubules in the groups that had been treated with 512µgml-1concentration of Ag/ZnO nanoparticlein combination with ciprofloxacin. In compared with the control groups.This study showed thatthe Ag/ZnO nanoparticle in cooperation with ciprofloxacin has great antimicrobial effect against E.coli on renal damage of Pyelonephritis.
KEYWORDS:Ag/ZnO Nanoparticle; Antibacterial; Pyelonephritis
Download this article as:| Copy the following to cite this article: Khoshkbejari M. A. P, Jafari A, Safari M. Ag/ZnO Nanoparticles as Novel Antibacterial Agent against of Escherichia coli infection, in vitro & in vivo. Orient J Chem 2015;31(3). |
| Copy the following to cite this URL: Khoshkbejari M. A. P, Jafari A, Safari M. Ag/ZnO Nanoparticles as Novel Antibacterial Agent against of Escherichia coli infection, in vitro & in vivo. Orient J Chem 2015;31(3).. Orient J Chem 2015;31(3). Available from: http://www.orientjchem.org/?p=10030 |
Introduction
Urinary tract infection is the most common nosocomial infection and also is second common infection in the human population so that each year more than 7 million patients visit physicians’ offices in the United States of America. The infection can be seen in 1 to 3 percent of school-aged girls and in 2 to 8 percent of pregnant women. Researches show that all women in their lifetime will experience a UTI turn and almost 25 percent of women in their lifetime, once are affected by symptomatic severe urinary tract infections1,2. Urinary tract infection according to infection part is divided into types of urethritis, pyelonephritis which pyelonephritis is the more dangerous type among other and has serious complications, especially in the area of childhood which is diagnosed Based on urine3,4,5 and urinary symptoms such as dysuria, urinary frequency, flank pain and fever6. According to research carried out by Wales, the most common pathogen isolated from urine, was reported E.coli7. Late Diagnosis and treatment lead to irreversible renal injury in patients with pyelonephritis and one of the most risky results of pyelonephritis is the formation of these renal scars8. This phenomenon is mainly due to inflammatory damage is caused by a bacterial infection. Since early diagnosis and antibiotic treatment in these cases is often delayed, there is the possibility of developing renal scars in more than 40 percent of those with high blood pressure, urinary protein excretion, and renal diseases9. On the other hand, due to the increasing incidence of resistance of microorganisms to beta-lactam and aminoglycoside antibiotics, which causes their efficiency to be reduced, Also consider the potential side effects of each drug, several researchers has been done to find alternative antibacterial agents in the case of urinary tract infection10. Hence antibacterial properties of Nano metal oxides have been discovered as new generation of antimicrobial agent and researchers have offered the use of silver and zinc ions as superior disinfectants for from hospitals infectious microorganisms11,12,13. Although, they have believed that residual these metal ions may adversely affect human health14but scientist experiments demonstrated selectivity in the toxic nature of ZnO nanoparticle to different bacterial systems and human T lymphocytes.As far as today, researchers have focused on the production of ZnO nanoparticles using the aqueous root extracts of Zingiber officinale15.Those results suggested that ZnO nanoparticle may potentially prove useful as Nano-medicine based antimicrobial agents at selective therapeutic dosing regimens11.The mechanism of action of the silver and zinc nanoparticles is not yet fully established11,16. But nowadays, we know that the bactericidal effect of metal nanoparticlehas been attributed to their small size, photo-catalystic of activity and high surface to volume ratio, which allows them to interact closely with microbial membranes and is not merely due to the release of metal ions in solution17. While various hypotheses have been proposed to explain the mechanism of antimicrobial activity of silver nanoparticle, it is widely believed that silver nanoparticle is incorporated in the cell membrane, which causes leakage of intracellular substances and eventually causes cell death18,19. Some of the silver nanoparticle also penetrate into the cells. It is also reported that bactericidal efficiency is affected by the type of microorganism. In studies with gram negative, E.coli; Kim reported greater biocide efficiency of silver nanoparticle for E.coli20. Also recently, Jayesh assumed that combination of metal oxide nanoparticle may give rise to more complete bactericidal effect against mixed bacterial population 16. The Purpose of this study was synthesis of ZnO and Ag nanoparticlesmonometallic and Ag/ZnOnanoparticle via thermal decomposition of oxalate precursor method. The aim of this study was to investigate the antibacterial effects of Ag/ZnOnanoparticle on renal scars due to pyelonephritisrenal infection in rat models.
Materials and Methods
Synthesis of Ag/ZnONanoparticle and Characterization
Ag/ZnOnanoparticle was synthesized in Antimicrobial resistance research center of Iran University of medical science, according to Jafari and their Colleague’s protocol.Crystallinity of the nanoparticle performed using an X-ray diffract-o-meter set (XRD, Bruker D8-Advance diffract meter using Cu Kα radiation). The nanoparticle was analyzed by inductively coupled plasma-atomic emission spectroscopy (ICP-AES, LIBERTY–RL, Varian Australia Co.) FT-IR spectra was recorded on a Bruker spectrophotometer in KBr pellets. Surface morphology of product was characterized by using a scanning electronic microscopy (SEM, Cam Scan MV2300) with an accelerating voltage of 30 KVand Transmission electron microscopy (TEM)21,22.
Antibacterial tests
According to research conducted, the lowest concentration of material that inhibits the growth an organism was defined as the MIC. From the serial dilution method, was employed to determine the MIC of the nanoparticles21,22. The microbial growth was studied by turbid metric measurement, using a spectrophotometer (Nano-volume spectrophotometer, Scan drop 250, Analyticjena Co.)23. All the experiments were carried out on triplicate. MBC, that is, the lowest concentration of nanoparticle that kills 99.9% of the bacteria was also determined from the batch culture studies. To experiments for bactericidal effect, loopfull from each test tube was inoculated on Muller Hinton agar and incubated at 35°C for 24 h. The release of Ag+ and Zn2+ ions from the nanoparticle into DI water and Muller Hinton Broth medium was deliberated. And also residual Ag+ and Zn2+ concentration in the aqueous phase was definite by ICP-AES after 24 h.
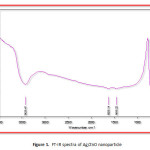 |
Figure1: FT-IR spectra of Ag/ZnO nanoparticle Click here to View figure |
Maintenance Condition of Rat model
Random bred male Wistarrats weighing approximately 130±10 g, obtained from TarbiatMoallem University of Karaj laboratory, Laboratory Animal of Reproduce Center, Karaj, Iran. The rats were evaluated prior to initiation of the study to ensure a sanitary hygienic condition and acclimation to the study environment. Clinically accepted animals were randomly assigned into nine major groups and in each group were 5 rats (5 rats/group).
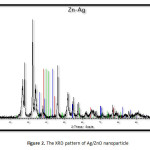 |
Figure2: The XRD pattern of Ag/ZnO nanoparticle Click here to View figure |
Environmental Condition of Rat Model
A total of 45 rats, were housed in stainless steel wire mesh cages on a bedding of wood chips (Five animals/Cage). They were kept in an ambient temperature of 22 ± 2oC, with 40-60% air moisture on a light/dark cycle of 12/12 hours and supplied rat chow (the diet) and fresh water.
Bacterial Strains
The bacterial strain of E.coli used in this project was (ATCC25922) which certainty was pathogen and disease-causing in rats and it absolutely caused APN in them.
 |
Figure3: TEM image of Ag/ZnO nanoparticle Click here to View figure |
Surgery and injection of bacterial suspension
At First, The 4:1 ratio of the two drugs Ketamine Hydrochloride and Xylazine was Prepared and the 0.1 ml of it was injected into the peritoneal of rats till they got anesthetized animals. Then the left side of the rats (Between ribs and hip bone) is sterilized with ethanol 70% (MERCK, Germany) and longitudinal incision with the length of 2 to 3 cm is created by surgical blade. After cutting the skin and muscle layers below it, incision on both sides with a special forceps kept open and with an insulin syringe, the 0.3ml of bacterial suspension with concentration of 1.5×108CFUml-1 microorganism, in two to three different regions of the apical region of renal cortex is injected. Then with the needle and suture the muscle layer and the skin layer, sutures tied and diluted povidonelodinetopical solution(DarooDarman, 10%) is used to disinfect the wound. During the course of treatment the wound disinfected every day and Washing with saline is given after surgery to prevent infection.
Urine culture
Urine was collected from mice 24 hours after surgery and In order to count the number of colonies were cultured in EMB by Pure culture.
 |
Figure4: SEM image of Ag/ZnO nanoparticle Click here to View figure |
Antibiotic Sensitivity Test
To determine the sensitivity of standard strain of E.coli to common antibiotics, Disc Diffusion agartest with Ceftriaxone (30μg),Gentamicin (10μg), Ciprofloxacin (5μg) and Cefazolin (30μg), antibiotic disks on Mueller Hinton agar medium were done. After 24 hours of incubation, the diameter of growth inhibition around each disc was measured and compared. The disc which has the maximum diameter of the inhibitory zone; it is more effective to destroy bacteria.
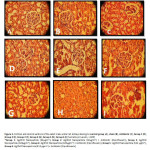 |
Figure5: Cortical and central sections of the adult male wistar rat kidneys belong to control group (A), sham (B), Antibiotic (C), Group 1 (D), Group 2 (E), Group 3 (F), Group 4 (G), Group 5 (H), Group 6 (I) (hematoxylin-eosin, × 400) *Group 1: Ag/ZnO Nanoparticle (32µgml-1), Group 2: Ag/ZnO Nanoparticle (32µgml-1) + Antibiotic (Ciprofloxacin), Group 3: Ag/ZnO Nanoparticle (128µgml-1), Group 4: Ag/ZnO Nanoparticle (128µgml-1) + Antibiotic (Ciprofloxacin), Group 5: Ag/ZnO Nanoparticle (512 µgml-1), Group 6: Ag/ZnO Nanoparticle (512 µgml-1) + Antibiotic (Ciprofloxacin) Click here to View figure
|
Treatment
Curing the Treatment groups began treatment 72 hours after surgery. Groups treated with antibiotics received the ceftriaxone at a dose of 50 mg/kg diluted with distilled water by intramuscular injection. Drug treatment groups were orally received Ag/ZnOnanoparticle as a suspension in distilled water by gavage needle.
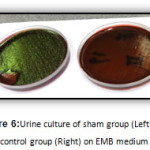 |
Figure6: Urine culture of sham group (Left) and control group (Right) on EMB medium Click here to View figure |
The experimental groups
In this study, the animals were divided into 9 groups and the effect of three concentrations of MIC and MBC tests; also intermediate concentrations of Ag/ZnO nanoparticle between them, each as a separate group, were studied. Group 1: The control group which has false surgery (Non-injected kidney infection) but the infection is not transmitted and after 72 hours, daily and for 14 days saline solution at a dose of 10 mg/kg was injected into their peritoneum. Group 2: The group which underwent surgery with transferring infection with concentration of 1.5×108 CFU/ml and the infection was transferred and After 72 hours, daily for 14 days Normal Saline solution 0.9% (Darosazi-Samen, Iran) with a dose of 10 mg/kg injected into the peritoneum of this group. Group 3: The group which underwent surgery and infection was transmitted and after 72 hours for 14 days, only received the ciprofloxacin antibiotic at a dose of 50 mg/kg by intramuscular injection. Group 4: The group which underwent surgery and infection was transmitted and after 72 hours for 14 days, orally received 1 ml Ag/ZnO nanoparticle with the concentration of 32µgml-1 (MIC). Group 5: The group which underwent surgery and infection was transmitted and after 72 hours for 14 days, orally received 1 ml Ag/ZnO nanoparticles with a concentration of 32µgml-1 (MIC) also received ciprofloxacin antibiotic concentration of 50mg/kg by intramuscular injection. Group 6: The group which underwent surgery and infection was transmitted and after 72 hours for 14 days, orally received 1 ml Ag/ZnO nanoparticles with the concentration of 128 µgml–1(Concentration between the MIC and MBC). Group 7: The group which underwent surgery and infection was transmitted and after 72 hours for 14 days, orally received 1 ml Ag/ZnO nanoparticles with the concentration of 128 µgml-1(Concentration between the MIC and MBC) also received the ciprofloxacin antibiotic at a dose of 50 mg/kg by intramuscular injection. Group 8: The group which underwent surgery and infection was transmitted and after 72 hours for 14 days, orally received 1 ml Ag/ZnO nanoparticle with the concentration of 512 µgml-1 (MBC). Group 9: The group which underwent surgery and infection was transmitted and after 72 hours for 14 days, orally received 1 ml Ag/ZnO nanoparticle with the concentration of 512µgml1 (Concentration between the MIC and MBC) also received the ciprofloxacin antibiotic at a dose of 50 mg/kg by intramuscular injection.
Histological and Pathological examination of kidney tissue
After 6 weeks, all animals were killed after anesthesia and their kidneys removed and divided longitudinally into two halves and In order to investigate the histological and pathological assessments fixed in 10% formaldehyde. Tissues Obtained are strained by Hematoxylin and Eosin (H&E) and Masson and Microscopic assessment were done by a pathologist who is unaware of their grouping. Finally, by comparing the data obtained from each of the groups, the added effect of each variable was examined.
Results
The FT-IR spectra analysis
Fig.1. shows FT-IR spectra of Ag/ZnO nanoparticle. Fig.1. conclude Ag/ZnO FT-IR spectrum. The broad band at 3426.47 cm-1 was allocated to both the νs (O-H) and νs (O-H) of hydration water. The extreme band at 1628.14 cm-1 was allocated to asymmetric and water tensional tremble δ (H-O-H). The shoulder at 1458.22 cm-1 is present in the spectrum evidence of (N-O) tremble and the closely spaced bands 625.36 cm−1 are presents in the spectrum evidence of (Ag/ZnO) tensional tremble, respectively.
The XRD spectra analysis
The XRD pattern of Ag/ZnO nanoparticle (Fig. 2.) was compared and interpreted with standard data of International Centre of Diffraction Data (ICDD). The average crystallite size (C.S) of the nanocrystale was calculated using the Debye-Scherer Equation from the major diffraction peaks. The average crystallite size of the Ag/ZnO was 12.15 nm.
The ICP-AES spectra analysis
By ICP-AES analysis, we succeed to estimation of residual ions, after digestion of nanoparticle by sonication. They indicated ions levels of 120 ppm to silver and ≤ 1 ppm to zinc oxide, in the silver/Zinc Oxide nanoparticle, respectively.
The TEM and SEM images analysis
TEM image (Fig. 3.) of nanoparticle approved that the metal particles was in the Nano range, approximately. However, SEM images (Fig. 4.) of nanoparticle was showed that Ag/ZnO metal particle was exactly in the shape of spherical and cluster.
The antibacterial activity analysis
The results in depended of the antibacterial effects of nanoparticle against E.coli via MIC and MBC are summarized in Table 1. A greater lag phase and lower maximum absorbance (at 600 nm) were observed as the concentration of nanoparticles increased. Similar observation was reported by Sondi and Salopek-Sondi19. We had analyzed effectively of silver/zinc oxide nanoparticle against E.coli. The bactericidal effect of nanoparticle is dependent on the concentration of nanoparticle and the initial bacterial concentration24. In this study, the initial bacterial concentration was constant at 1.5×108 CFU ml-1 regardless of nanoparticle concentration and microbial strain. Our research shows that the silver/zinc oxide nanoparticle has great antibacterial effect against E.coli. In fact, the E.coli was most sensitivity against of silver/zinc oxide nanoparticle. The MIC and MBC observed in this study for silver/zinc oxide nanoparticle were 32 µgml-1 and 512 µgml-1 for E.coli, respectively.
Study of renal glomerulus pyelonephritis in mice treated with Ag/ZnO Nanoparticle Compared with other experimental groups
The results show that the number of renal glomeruli, also the kidney glomerular diameter in sham pyelonephritis rats and rats treated with concentration of 32 µgml-1 Ag/ZnO Nanoparticle (Group 4) Compared with normal rats in the control group, have a significant reduction. While the other groups, reducing the number of objects does not show a significant difference compared to control group. The results show that the thickness of the cortical kidney in pyelonephritis rats of sham group and also, rats treated with concentrations of 32 µgml-1 and 128 µgml-1Ag/ZnO nanoparticle Compared with normal rats in the control group decreased significantly. While treatment with a concentration of 512 µgml-1Ag/ZnO nanoparticle and also antibiotic treatment in separate groups and treated groups with the concentration of 512 µgml-1reduced Severity of renal cortical thickness of pyelonephritis rats in these groups compared to sham rats and reduce the number The results in depended of the antibacterial effects of nanoparticle against E.coli via MIC and MBC are summarized in Table 1. A greater lag phase and lower maximum absorbance (at 600 nm) were observed as the concentration of nanoparticles increased. Similar observation was reported by Sondi and Salopek-Sondi19. We had analyzed effectively of silver/zinc oxide nanoparticle against E.coli. The bactericidal effect of nanoparticle is dependent on the concentration of nanoparticle and the initial bacterial concentration24. In this study, the initial bacterial concentration was constant at 1.5×108 CFU ml-1 regardless of nanoparticle concentration and microbial strain. Our research shows that the silver/zinc oxide nanoparticle has great antibacterial effect against E.coli. In fact, the E.coli was most sensitivity against of silver/zinc oxide nanoparticle. The MIC and MBC observed in this study for silver/zinc oxide nanoparticle were 32 µgml-1 and 512 µgml-1 for E.coli, respectively.
of objects in this group than in the control group did not show significant differences.
Microscopic study of sections of kidney tissue
Tissue slides prepared from the experimental group, encoded and given to the expert pathologist who was unaware of the characteristics of the experimental group and order of slides so that Check the status of their tissue of various pathological factors and reports them. For this aim, complications such as Inflammation of the tissue, dilated and Dilated Tubules, Atrophic Tubules, Neutrophil Abscesses, Tubular damage, Tissue Necrosis, Apoptotic injury, PMN within the tubular, Cast within the context were pathologically scrutinized. The microscopic pathological examination was performed on sections prepared from groups of data in this study also identified areas of tissue damage that is shown in Table 2. Inflammation of renal tubules is the most importance indicators of kidney tissue pathology of pyelonephritis and the results suggest that indicate severe inflammation occurred in the sham group and there was the difference compared to the control group. However in the treated group by ciprofloxacin antibiotic and also the groups treated with different concentration of Ag/ZnO nanoparticle, especially the 128 µgml-1 and 512 µgml-1, as well as additional particles treated with antibiotics, it is quite evident.
Urine culture
The results of this study show that injection of 0.1 ml of bacterial suspension of E.coli ATCC 25922 strain which has a 0.5 McFarland standard bacterial concentration to various areas of the poles of the kidney cortex of rats causes the pyelonephritis Kidney infection model to be created in them. So that, causing infection in the kidneys of rats with urine culture in respective rats and bacterial growth in EMB medium and creating green shiny color of colonies, which is the unique characteristics of the bacteria colonies in this medium, clearly was evidenced.
Discussion
Jayesh performed extensive experiments in determination of microbial sensitivity of various bacteria to silver and copper nanoparticles, using disc diffusion method16. Regarding that the diameter of inhibition zone (DIZ), reflects the sensitivity of the organism, strains of sensitive, show larger DIZ and the resistant strains show smaller DIZ. Results of the disc diffusion with Ag nanoparticle, by Thirumurugan against strains of pathogens E.coli, indicated higher sensitivity to silver nanoparticles which is in contrast with the results of our study24. The E.coli, used in the current study, showed the least and the most sensitivity to silver and Ag/ZnO nanoparticles, respectively. Actually, the least degree of MIC in E.coli was related to combined nanoparticles of silver and zinc with concentration of 128 µgml-1. i.e, notwithstanding this nanoparticle had the most growth inhibitory effect in E.coli. Kim studied the gram negative bacteria E.coli, also reported that the antibacterial silver nanoparticle mostly affects the E.coli, which is due to the difference between cell wall of gram negative microorganism20. Limited studies performed on the antibacterial properties of ZnO nanoparticle. Reddy were amongst few authors worked on the toxicity of the ZnO nanoparticle in gram negative bacteria 11. They found that nanoparticles are able to completely inhibit the growth of E.coli. Ling Yang and jafari combined silver nanoparticles with Zn to improve antibacterial activity of Zn nanoparticle and investigate the antibacterial effect of Zinc oxide and silver nanoparticles and also comparing them with Ag/ZnO nanoparticle 25,21. They obtained interesting results. According to the findings of Kawashita and Pak-soo silver significantly increased antimicrobial activity 26,27. Actually, Ling Yang believed that photo-catalytic ability of ZnO nanoparticle plus silver nanoparticle improves and also increases its oxidation and reduction abilities, while suppressing bacteria growth28,29. However, silver ions, eventually release during sterilization and kill bacteria due to their high antibacterial activation. They theorized that silver ions release following bacteria death and colloid with other bacteria and repeat their sterilization behavior. It was also mentioned that silver covered in the surface of Zn nanoparticle has the ability to involve the electrons produced through photo-catalytic reactions of Zn nanoparticle which increases electron isolation and makes gaps in cell membrane, so increase its antimicrobial activity. Regarding studies of these authors, antibacterial property of silver and zinc oxide nanoparticles improves with their combination. Up to now, no complete and comprehensive study reported in the field of antibacterial properties of Ag/ZnO for E.colion Renal Damage in Rat Model of Pyelonephritis.Our study confirmed that the gram negative strains of bacteria, specifically E.coli; had most sensitive to silver/zinc oxide nanoparticle. In addition, out study approved thatinflammation is suppressed after completion of treatment via Ag/ZnO nanoparticle in combination with Ciprofloxacin. In fact, inhibition of tissue inflammation in the group which was treated with Ag/ZnO nanoparticle in combination with Ciprofloxacin after getting a urinary tract infection and pyelonephritis could indicate that the concentration of particles in the well can act as anti-inflammatory substance and reduced severity of inflammatory tubule which is the main characteristic of pyelonephritis kidney. Pathological studies on other indicators such as tubular dilation, Neutrophil abscesses, Atrophy, Necrosis and Tubular damage was also having the same situation. In fact, it reflects the influence of Ag/ZnOnanoparticles on renal tissue and increase protection of renal tissue against pyelonephritis infection. According to the research done, Ag/ZnO nanoparticle in cooperation with ciprofloxacin had great antimicrobial effect against E.coli (ATCC 25922) on renal damage of Pyelonephritis. Interesting point was here that in microscopic pathological examination, there was no any severe Inflammation of renal tubules in the groups that had been treated with MBC concentration of Ag/ZnO nanoparticle in cooperation with ciprofloxacin and treatment with that, reduced severity of renal cortical thickness of pyelonephritis rats.
Acknowledgement
We are indebted to research Vice Chancellor of Iran University of Medical Sciences, for supporting this research. Also, we gratefully acknowledge to University of Industrial Sahand, Tabriz, Department of Nanotechnology, for the XRD and SEM analysis. The authors would like to acknowledge to Iranian Mineral Processing Research Center (IMPRC) for ICP-AES analysis, TarbiatMoallem University and Laboratory Animal of Reproduce Center, Karaj, Iran. We gratefully acknowledge Executive Director of Iran-Nanotechnology Organization (Govt. of Iran) and pathological laboratory of Dr. Alireza Vahab-zade. The anonymous reviewers are acknowledged for providing valuable comments and insights for improving the manuscript.
References
- Rahul, M.;Sudhir, A.;Saroj, Sh.; Sanjay, Ch.;Kusum, H. Journal of Infection and Public Health.2009, 2, 101—111
- Shirishkumar, P.;Pankajkumar, P. T.; Mala, S.;Javadekar,T. B.;Vipul, P. Ch.National Journal of Community Medicine.2012, 3, 138-141
- Heller, E.D.;Drabkin,N. British Veterinary J.1977, 133, 572-578
- Duguid, J. P.; Cleggs.;Wilson,I. J. Medical Microbiol.1979, 12, 213-217
- Bauer, A.W.;Kirby,W.M.M.;Sheris,J.C.;Truck,M. American J. Clinical Pathol.1966, 142, 225-230
- Reddy,K.;Koteeswaran,Y. A. Indian Veterinary J.1994, 71, 959-963
- Yoder, H.W.Avian Diseases.1989, 33, 502-505
- Roy, P.;Edwin,P.G.;Purushothaman,V. Indian Veterinary J.2004, 81, 1317-1320
- Susantha, M.G.;Riddell,C.;Andrew,A.P.;Allan,B.J. Canadian Journal of Veterinary Res.2001, 65, 1-6
- Reddy,K.;Koteeswaran,Y. A.;Dorairajan,N. Indian Veterinary J.1994, 71, 209-212
- Reddy, k.; Bell, J.;Wingett, D.; Hanley, C.;Punnoosea, A.Appl. Phys.Lett. 2007, 24, 90-93
- Lin, YE.;Vidic, R.D.; Stout, J. E.;Mccartney, C. A.; Yu, VL.Water Res.1998, 32, 1997–2000
- Lin,YE.;Vidic, RD.; Stout, JE.; Yu, VL.Water Res. 1996, 30, 1905–1913
- Blanc,DS.;Carrara, P.;Zanetti, G.;Francioli, P.J. Hosp. Infect.2005, 60, 69–72
- ANAND RAJ, L.F.A.;JAYALAKSHMY, E.Oriental Journal of Chemistry.2015, 31, 51-56
- Jayesh, P.; Arup K. Ch.; Siddhartha, P.;Duttagupta,;Suparna, M.Nanomedicine.2007, 3, 95–101
- Morones, J. R.;Elechiguerra, J.L.; Camacho, A.; Holt, K.;Kouri, J.B.; Ramirez, J.T.Nanotechnology.2005, 16, 2346–2353
- Cho, K.; Park, J.; Osaka, T.; Park, S.Electrochim. Acta.2005, 51, 956–960
- Sondi, I.;Salopek, S. B.J. Colloid Interf. Sci.2004, 275, 177–182
- Kim, J. S.;Kuk, E.; Yu, K. N.; Kim, J. H.; Park, S. J.; Lee, H. F.; Kim, S. H.; Park, Y. K.; Hwang, C. Y.; Kim, Y. K.; Lee, Y. S.;Jeong, D. H.; Cho, M. H. Nanomedicine.2007, 3, 95–101
- Jafari, A.;Ghane, M.;Arastoo, sh.African Journal of Microbiology Research.2011, 30, 5465-5473
- JAFARI, A.; GHANE, M.; SARABI, M.; SIYAVOSHIFAR, F.Oriental Journal of Chemistry.2011, 27, 811-822
- Case, C. L.; Johnson, T. R.California, Benjamin Cummings Pub Inc.1984, 126–129
- Dabbagh, M. A.;Moghimipour, E.;Ameri, A.;Sayfoddin, N.Iranian.2008, 7, 21-28
- Siva, K. V.;Nagaraja, B. M.;Shashikala, V.;Padmasri, A. H.;Madhavendra, S. S.;Raju, B. D.J MolCatal A Chem.2004, 223-313
- Williams, D. N.;Ehrman, SH.;Holoman, TRP.J Nanobiotechnol.2006, 4-3
- Kawashita, M.;Tsuneyama, S.; Miyaji, F.Biomaterials.2000, 21, 393-398
- Pak, S. J.; Jang, Y.Journal of Colloid and Interface Science.2003, 261, 238-243
- YANG, L.; MAO, J.; ZHANG, X.; XUE, T.; HOU, T.; WANG, L.;Mingjing, T.U.Nanoscience.2006, 11, 44-48

This work is licensed under a Creative Commons Attribution 4.0 International License.









