Synthesis and NMR study of N-heterocyclic carbenes (NHC) precursors derived from Tröger’s Base
Eric MUSENGIMANA and Claver FATAKANWA
National University of Rwanda, Faculty of Science, and Department of Chemistry
DOI : http://dx.doi.org/10.13005/ojc/290426
Article Received on :
Article Accepted on :
Article Published : 11 Jan 2014
Since the discovery of olefin metathesis reactions, various catalysts have been developed but those based on Ruthenium and N-heterocyclic carbenes (NHC) ligands are particularly more efficient. Herein, we report a synthesis of NHC precursors derived from the Tröger’s base and its analogues which can be used in the synthesis of the new kind of metathesis catalysts. The NHC derived from the Tröger’s base present a particular chirality that could affect stereoselectivity of catalysis, and the form “V” of these ligands implies that the aromatic substituents point totally outside the coordination sphere of the metal, where would be no steric hindrance unlike what happens with the usual NHC.
KEYWORDS:NHC (N-heterocyclic carbenes);Tröger’s base;perhalogenated adducts;thioureas adducts
Download this article as:| Copy the following to cite this article: MUSENGIMANA E, FATAKANWA C. Synthesis and NMR study of N-heterocyclic carbenes (NHC) precursors derived from Tröger’s Base. Orient J Chem 2013;29(4) |
| Copy the following to cite this URL: MUSENGIMANA E, FATAKANWA C. Synthesis and NMR study of N-heterocyclic carbenes (NHC) precursors derived from Tröger’s Base. Orient J Chem 2013;29(4). Available from: http://www.orientjchem.org/?p=1569 |
Introduction
Since their discovery in 1931, increasing attention is still paid to the olefination metathesis reactions 1. These reactions correspond to the redistribution of hydrocarbon backbone of the molecules in which carbon-carbon double bonds are rearranged 2. They have multiple and varied applications in organic synthesis, in perfume industry, in biology, in pharmaceutical chemistry, in polymers and petrochemicals industry. Therefore, considerable progress has been made for the development of olefin metathesis in organic synthesis 3, 4. Numerous studies have been devoted to the development of efficient metathesis catalysts based on ruthenium and N-heterocyclic carbenes (NHC) ligands 5, 6, 7, 8. The NHC are stable derivatives of divalent carbon. Since their discovery by Arduengo in 1991 9, they are increasingly used as ligands in organometallic chemistry. These electron-rich and sterically hindered compounds exert a beneficial influence on the activity of the metal complexes in which they are incorporated 5. Because of their structural diversity and the strength of their connections with most metals, they lead to robust and selective active species that can be used under mild conditions. They have already given rise to a wide range of catalytic systems and their development is still far from complete 10. Among the hundreds of NHC available at present, the most used are imidazole derivatives [IMes:1,3-bis(2,4,6-trimethylphenyl)-imidazol-2-ylidene and SIMes:1,3-bis(2,4,6-trmethylphenyl)-imidazolin-2-ylidene] especially in many metal complexes for olefin metathesis.
The Tröger’s base has been synthesized in 1887 by Julius Tröger, but its structure was elucidated 38 years later by Vladimir Prelog and confirmed by X-ray diffraction in 1986 11.
In the last thirty years, considerable progress has been made for the development of olefin metathesis reactions in organic synthesis 3, 4, 12; and numerous studies have been devoted to the development of efficient metathesis catalysts based on ruthenium and NHC ligands 6, 12. However, no work has been reported to the synthesis of carbenes derived from Tröger’s base or their use as ligands in organometallic catalysis. The purpose we set for this study was to synthesize carbene precursors (perhalogenated adducts and thioureas) derived from the Tröger’s base and to investigate their conversion into carbenes or into ruthenium complexes.
Experimental
Solvents used were of technical grade and commercial reagents were purchased from Acros, Sigma-Adrich, ABCR, TCI or Apollo scientific and used as received unless stated otherwise. THF was freshly distilled from sodium with benzophenone under an argon atmosphere. 1H NMR was recorded on a Bruker-300 MHz, APT, COSY, HSQC, and HMBC were recorded on a Bruker-500 MHz spectrometer. Chemical shifts are reported relative to CDCl3 (7.26, 77.16 ppm). Spectral features were designed as follows: s = singlet, d = doublet, t = triplet, q = quartet, m = multiplet. Column chromatography was carried out on silica gel (ROCC 60, 40-63 μm). TLC analyses were performed on commercial aluminum plates bearing a 0.25 mm layer of Merck Silica gel 60F254.
Tröger ‘s base and its analogues (1a-d): -2,8-dimethyl-6H,12H-5,11-methanodibenzo[b ,f][1,5]diazocine, -2,8-dimethoxy-6H,12H-5,11-methanodibenzo[b, f][1,5]diazocine, -2,8-dichloro-6H,12H-5,11-methanodibenzo[b,f][1,5]diazocine, 6H,12H-5,11-methanodibenzo [b,f][1,5]diazocine, were synthesized according to the methods described in the litterature 13, 14, 15, 11. The removal of methylene bridge to the above Tröger’s base leading to the corresponding diazocines (2a-d): -2,8-Dimethyl-5,6,11,12-tetrahydrodibenzo [b, f] [1,5] diazocine,-2,8-Dimethoxy-5,6,11,12-tetrahydrodibenzo [b, f] [1,5] diazocine, -2,8-dichloro-5,6,11,12-tetrahydrodibenzo [b, f] [1,5] diazocine, -5,6,11,12-tetrahydrodibenzo [b, f] [1,5] diazocine were synthesized according to the methods described in the literature 16.
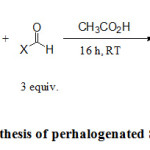
Synthesis of C6F5-adduct of Tröger’s base bearing the chloro, methyl and methoxy groups (3a-3c)
In a GPC bottle, glacial acetic acid (1 mL); 2,8-Dichloro-5,6,11,12-tetrahydrodibenzo [b, f] [1,5] diazocine (1 mmol) or 2,8-Dimethyl-5,6,11,12-tetrahydrodibenzo [b, f] [1,5] diazocine ( 1 mmol) or 2,8-Dimethoxy-5,6,11,12-tetrahydrodibenzo [b, f] [1,5] diazocine ( 1 mmol) and C6F5CHO (1.5 mmol) were introduced. The reaction mixture was stirred at room temperature until a precipitate appears. It was filtered and washed with cold methanol (-20 ° C). The resulting white solids were extracted with dichloromethane. After evaporation under reduced pressure, the solid was analyzed by NMR.
Methyl derivative (3a): 5h, white solid, yield: 0.32 g; 76%; 1H NMR (400 MHz, CDCl3) δ 7.1-6.9 (m, 2H), 6.8-6.7 (m, 2H), 6.5 (s, 1H), 6.2 (s, 1H), 5.5 (s, 1H), 4.75 (d, 2JHH = 16.6 Hz, 1H), 4.4 (d, 2JHH = 16.6 Hz, 1H), 4.2 (d, 2JHH = 16Hz, 1H), 4.15 (d, 2JHH = 16.2 Hz, 1H), 2.1 (s, 6H).
Methoxy derivative (3b): 48h, white solid, yield: 0.35 g; 78%; 1H NMR (400 MHz, CDCl3) δ 7.2-7.0 (m, 2H), 6.8-6.7 (m, 2H), 6.47 (s, 1H), 6.33 (s, 1H), 5.65 (s, 1H), 4.85 (d, 2JHH = 16.7 Hz, 1H), 4.31 (d, 2JHH = 16.7 Hz, 1H), 4.15 (d, 2JHH = 17.3 Hz, 1H), 4.05 (d, 2JHH = 17.4 Hz, 1H), 3.70 (s, 6H).
Chloro derivative (3c): 5 min, white solid, yield: 0.2 g, 45%, 1H NMR (400 MHz, CDCl3) δ 7.15-6.68 (m, 8H), 5.55 (s, 1H), 4.77 (d, 2JHH = 16.8 Hz, 1H), 4.23 (d, 2JHH = 16.7 Hz, 1H), 4.10 (d, 2JHH = 17.4 Hz, 1H), 3.97 (d, 2JHH = 17.4 Hz, 1H).
Synthesis of C6F5-adduct of the unsubstituted Tröger’s base (3d)
In a GPC bottle, glacial acetic acid (1 mL); 5,6,11,12-tetrahydrodibenzo [b, f] [1,5] diazocine (1 mmol); p-toluene sulfonic acid (2-3 g) and C6F5CHO (1.5 mmol) were introduced. The mixture was allowed to stir for a week at room temperature. The reaction mixture was extracted with chloroform (10 mL) and washed with a saturated solution of NaHCO3 (20 mL x 3). The organic phase was dried with MgSO4, filtered and concentrated. The orange-brown oil obtained crystallized rapidly. These crystals were washed with cold isopropanol and filtered.
Yield: 0.12 g, 32%, 1 H NMR (400 MHz, CDCl3) δ 7.25-6.78 (m, 8H), 5.70 (s, 1H), 4.92 (d, 2JHH = 16.6 Hz, 1H), 4.42 (d, 2JHH = 16.6 Hz, 1H), 4.19 (q, 2H).
Synthesis of chloroform adduct of Tröger’s base unsubstituted or bearing chloro group (4c-4d).
In a GPC bottle, chloral (2 mmol); glacial acetic (1 mL) acid and 5,6,11,12-tetrahydrodibenzo [b, f] [1,5] diazocine (0.5 mmol) or 2,8-Dichloro-5,6,11,12-tetrahydrodibenzo [b, f] [1,5] diazocine (0.5 mmol) were introduced. After 16h of stirring at room temperature, the precipitate was formed and filtered through a Pasteur pipette containing a cotton wool and celite. After washing with cold methanol (-18 ° C) the resulting solid was extracted with dichloromethane. The latter was evaporated after and the solid has been analyzed by NMR.
Chloro derivative (4c): white powder, yield: 0.02 g, 34.31%, 1H NMR (400 MHz, CDCl3) δ 7.30-6.78 (m, 6H), 4.86-4.68 (m, 3H), 4.32-4.25 (d , 2JHH = 28 Hz, 1H), 4.14-4.07 (d, 2JHH = 28 Hz, 1H).
Hydrogenated derivative (4d): yellow powder, yield: 0.1 g, 58.82%, 1H NMR (400 MHz, CDCl3) δ 7.30-6.80 (m, 8H), 4.93-4.72 (m, 3H), 4.42-4.35 (d , 2JHH = 28 Hz, 1H), 4.23-4.16 (d, 2JHH = 28 Hz, 1H).
Synthesis of C6F5 adduct of 1,3-dimesitylimidazolin-2-ylidene (6a)
In a GPC bottle, N,N’- dimesityl-ethanediamine (2 mmol) and C6F5CHO (3 mmol) were introduced. The mixture was crushed with a glass rod and a few drops of glacial acetic acid were added while stirring. After adding glacial acetic acid (1 mL) a precipitate was formed. Another glacial acetic acid (1 mL) was added, and the precipitate obtained after filtration was washed with cold isopropanol (-20 ° C) and then dried. The product was obtained as a white powder.
Yield: 0.34g, 35%. 1H NMR (250 MHz, CDCl3) δ 6.79 (s, 4H), 6.37 (s, 1H), 3.89 (m, 2H), 3.51 (m, 2H), 2.51 (s, 9H), 2.20 (s, 9H) . 13C NMR (250 MHz, CDCl3) δ 139.07, 135.46, 130.12, 71.56, 51.04, 20.71.
Synthesis of chloroform adduct of 1,3-dimesitylimidazolin-2-ylidene (6b)
In a GPC bottle, N, N’-ethylene-dimesityl (2 mmol), glacial acetic acid (2 mL) and chloral (6 mmol) were introduced. After 17 h of stirring at room temperature, there was formation of a precipitate. The latter has been filtered on a Buchner funnel and washed with isopropanol. The substrate has been washed with a saturated solution of NaHCO3 (10 mL x 3) and extracted with chloroform. The combined organic layers were dried over MgSO4, filtrated, and concentrated under reduced pressure. The product was obtained as a white powder on which 1H NMR and TGA analyses have been performed.
Yield: 0.3g, 36%. 1H NMR (250 MHz, CDCl3) δ 6.85 (d, 4H), 5.57 (s, 1H), 3.91 (m, 2H), 3.29 (m, 2H), 2.47 (s, 6H), 2.45 (s, 6H), 2.25 (s, 6H).
Synthesis of the thiourea from Tröger’s base (7)
In a round bottom flask cooled at 0 ° C, 2,8-Dimethyl-5,6,11,12-tetrahydrodibenzo [b, f] [1,5] diazocine (5 mmol) and Na2CO3 (0.795 g, 3.75 mmol)were introduced. In another flask also cooled at 0 ° C, was added THF (5 mL) and thiophosgene (6 mmol). The solution of the second flask was added to the solution of the first flask and stirred overnight at room temperature. The THF was evaporated and the resulting solid was dissolved in dichloromethane (50 mL). The organic phase was washed with water (50 mL x 3), dried over MgSO4, filtered and evaporated. The crude product was purified by column chromatography on silica gel with EtOAc:Petroleum Ether (8:2). The yellow solid obtained after concentration was analyzed by 1H and 13C NMR.
Yield: 0.65g, 46.4% 1H NMR (250 MHz, CDCl3) δ 7.19-6.81 (m, 6H), 6.11-6.07 (d, 3JHH = 16 Hz, 2H), 4.45-4.43 (d, 2JHH = 8 Hz, 2H), 2.18 (s, 6H), 13C NMR (250 MHz, CDCl3) δ 174.73, 140.05, 139.2, 131.7, 130.93, 130.7, 127.05, 59.99, 21.19. )
Results and discussion
Our work started by the synthesis of starting materials. Firstly, Tröger’s base and its analogues (1a-d) were synthesized according to the method described in the litterature 13, 14, 15, 11. Thereafter, we proceeded on the removal of methylene bridge by using the method of Mahon 16. This lead to the corresponding diazocines (2a-d) that were used in the synthesis of perhalogenated and thiourea adducts.
Synthesis of perhalogenated adducts of the Tröger’s base and its analogues
The diazocines (2a-d) were treated with 1.5 equiv. of C6F5CHO, in glacial acetic acid. After a stirring at room temperature for various time according to the diazocine, this formally lead to the insertion of a carbene into the C-H bond of C6F5CHO (schemes 1). Interestingly, the chloro derivative 2c was very reactive and lead to the corresponding carbene 3c just after 5 minutes with a yield of 43%. The carbenes 3a and 3b from methyl and methoxy derivatives were isolated easily with good yields of 76% and 78% respectively. However, the unsubstituted derivative 2d was less reactive in the conditions of the reaction and p-toluene sulfonic acid (10 equiv.) was added (see experimental procedure). In these last conditions, the corresponding carbene 3d was isolated after one week with a low yield 33%; comparing to other derivatives. Several examples from the literature show that N-heterocyclic carbenes of imidazolin-2-ylidene type can be synthesized by heating such compounds 17, 18. In such reactions, there is a loss of C6F5 group and the formation of a free carbene.
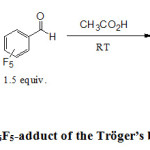 |
Scheme 1. Synthesis of C6F5-adduct of the Tröger’s base and its analogues Click here to View figure |
With these interesting carbenes in hands, we were secondly interested by the synthesis of other carbenes by using Cl3CHO (chloral) instead of C6F5CHO (scheme 2).
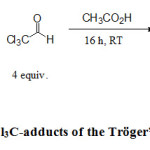 |
Scheme 2. Synthesis of Cl3C-adducts of the Tröger’s base and its analogues Click here to View figure |
The results of these syntheses were pretty random. In fact, after dissolving and mixing all the reagents in the glacial acetic acid, we were expecting that the products precipitate as it was the case for the preceding synthesis ; but this was not the case. Surprisingly, no reaction was observed for compounds 4a and 4b after an overnight stirring at room temperature. Impure products were isolated containing chloral for methyl derivative 4a. For chlorine derivative 4c and unsubstituted derivative 4d there was no appearance of a precipitate and the reaction mixture was partitioned between chloroform and an aqueous solution of NaHCO3. Evaporation of the organic layers led to oils that crystallized after one night.
Thermolysis tests of perhalogenated adducts of Tröger’s base
It is possible to obtain various complexes of rhodium or ruthenium bearing NHC ligands by a conventional heating a suitable metal precursor with C6F5 or chloroform adducts of SIMes ligand17. We tried to apply this method to the C6F5 adducts of the Tröger’s base. A first test was performed using as substrate the complex [RuCl2 (PCy3) 2 (2-phenylindenylidene)]. The Grubb’s metathesis catalyst of first generation was refluxed in toluene for 24 h in the presence of 3a (2 equiv.) (Scheme 3). The reaction was monitored by a 31P-NMR and a gradual appearance of a free PCy3 was observed in the reaction mixture. These observations seemed auspicious, the expected reaction involving the substitution of a phosphine ligand by analogous carbene. However, after purification of the crude by column chromatograph on silica gel, 1H-NMR and 13C-NMR analyses performed on the white solid obtained revealed that we did not isolate the expected product. But it was a mixture of uncharacterized products. It is therefore likely that the peak from the free phosphine observed in 31P NMR came from the decomposition of the starting complex, which did not withstand the applied temperature.
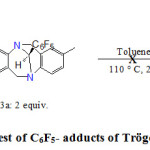 |
Scheme 3. Thermolysis test of C6F5– adducts of Tröger’s base with Ru complex |
In parallel, another attempt was carried out by choosing the dimer [RhCl (1,5-cyclooctadiene)] 2 as a binding site of carbene ligand (Scheme 4) in the same conditions as preceding. In fact, the rhodium complex is an air-stable ruthenium alkylidene species which should better withstand the thermolysis adopted to generate a carbene from the adduct 3a. The subsequent replacement of cyclooctadiene with carbonyl ligand, followed by the determination of vibration frequencies n CO with infrared spectroscopy, should also measure the electron-donor power of the carbene derived from the Tröger’s base. However, the NMR analysis of the crude of the reaction proved that the starting materials were isolated unchanged; hence no reaction.
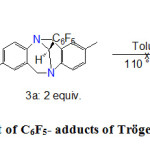 |
Scheme 4. Thermolysis test of C6F5- adducts of Tröger’s base with Rh complex Click here to View figure |
A third attempt to synthesize the free carbene of the Tröger’s base or its dimer from the adduct 3a was carried out in a microwave reactor. No metal complex was used in this experiment. The adduct 3awas simply dissolved in dry degassed toluene and the solution was heated in the microwave for half an hour at 200 ° C under pressure. To verify the presence of the carbene, few drops of CS2 were added to the reaction mixture. If a carbene was present in solution, the formation of a colored zwitterion should be observed, but it did not happen. There was no free carbene in solution. A 1H-NMR spectrum recorded on the crude corresponded to the starting materials.
Synthesis of perhalogenated SIMes adducts and thermogravimetric analyzes
In aim to compare the stabilities of the perhalogenated adducts of the Tröger’s base with those of classic N-heterocyclic carbenes, we have synthesized C6F5 and chloroform adducts of SIMes ligand. The synthesis method used is similar to that followed with the Tröger’s base and its analogues, i.e., the secondary diamine ligand 5 was stirred at room temperature for 17 h with 3 equiv. of C6F5CHO or chloral in the presence of acetic acid (Scheme 5).
 |
Scheme 5. Synthesis of perhalogenated SIMes adducts Click here to View figure |
Compounds 6a and 6b have been isolated as white powders with moderate yields; 49%, 36% respectively. .
A thermogravimetric analysis of perhalogenated adducts of the Tröger’s base, 3a, 3d and reference compound 6b were performed. These measures consisted of heating products in a microbalance and record the mass loss as a function of temperature. From a certain threshold, adducts decompose to form free carbene and expel chloroform or the C6F5CHO. The results of these analyzes showed that the adducts of the Tröger’s base remain stable at least up to 200 ° C and the exact nature of the adduct (chloroform or C6F5) has a very little influence on the decomposition temperature. For example, compounds 3a and 3d both degrade at about 220 ° C (Figure 1). In contrast, compound 6b releases chloroform at a very lower temperature, around 145 ° C, which explains the difference in reactivity between the two families of carbene precursors.
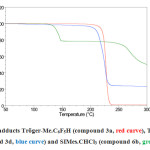 |
Figure 1. TGA adducts Tröger-Me.C6F5H (compound 3a, red curve), Tröger-H.CHCl3 (compound 3d, blue curve) and SIMes.CHCl3 (compound 6b, green curve). |
Synthesis of thiourea of the Tröger’s base and its analogues
Thermolysis of perhalogenated adducts of the Tröger’s base and its analogues was clearly much more difficult to accomplish than expected. Therefore, we have explored an alternative synthetic route that should lead to the achievement of the desired carbene. This new pathway is based on the use of thiourea as carbene precursors. These compounds can be obtained in one step from compounds 3a-d previously synthesized. They were treated overnight in THF at room temperature with 1.2 equiv. of thiophosgene in the presence of Na2CO3 (75%) to neutralize the hydrochloric acid formed (Scheme 6).
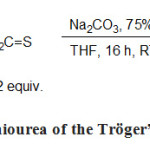 |
Scheme 6. Synthesis of thiourea of the Tröger’s base and its analogue Click here to View figure |
After purification by column chromatography on silica gel, the product 7 was isolated as a pure yellow solid with moderate yield of 46%. It was analyzed by NMR. The presence of a characteristic peak of C=S between 170 and 180 ppm in 13C NMR spectroscopy revealed that the expected product was formed.
Conclusion
The synthesis of carbenes precursors derived from Tröger’s base is feasible. Optimization of experimental conditions will be further investigated to improve yields and purity of isolated products. With comparative thermogravimetric analysis, we have shown that adducts pentafluorobenzene and chloroform of the Tröger’s base are much stable than those derived from a classic NHC. Refluxing in toluene in the presence of a complex of Ru or Rh, and the irradiation at 200°C in a microwave reactor haven’t allowed the desired thermolysis. The study of an alternative way to achieve the carbene from Tröger’s base involving thioureas as an intermediate key should be further investigated. A further study should also be undertaken to investigate the effectiveness of these new catalysts especially in cross-coupling metathesis reactions.
References
- Schuster M., Blechert S., Angew. Chem. Int. Ed., 36, 2036 (1997).
- Astruc D., Actualité chimique, 273, 3 (2004).
- Chauvin Y., Angew. Chem. Int. Ed., 45, 3740 (2006).
- Grubbs R. H., Angew. Chem. Int. Ed., 45, 3760 (2006).
- Herrmann W. A., Angew. Chem. Int. Ed., 41, 1290 (2002).
- Samojlowicz C., Bienik M., Grela K., Chem. Rev., 109, 3708 (2009).
- Schrock R. R., Chem. Rev., 109, 3211 (2009).
- Vougioukalakis G. C., Grubbs R. H., Chem. Rev., 110, 1746 (2010).
- Arduengo III A. J., Harlow R. L., Kline M., J. Am. Chem. Soc., 113, 361 (1991).
- Hahn F. E., Jahnke M. C., Angew. Chem. Int. Ed., 47, 3122 (2008).
- Sergeyev S., Helv. Chim. Acta, 92, 415 (2009).
- Schrock R. R., Angew. Chem. Int. Ed., 45, 3748 (2006).
- Didier D., Sergeyev S., Chimie nouvelle, 96, 92 (2007).
- Didier D., Tylleman B., Lambert N., VandeVelde C. M. L., Blockhuys F., Collas A., Sergeyev S., Tetrahedron, 64, 6252 (2008).
- Irving S., Vincent L., Ly P., Craig S. W., J. Org. Chem., 53, 98 (1988).
- Mahon A. B., Craig D. C., Try A. C., Arkivoc, xii, 148 (2008).
- Blum A. P., Ritter T., Grubbs R. H., Organometallics, 26, 2122 (2007).
- Nyce G. W., Csihony S., Waymouth R. M., Hedrick J. L., Chem. Eur. J., 10, 4073 (2004).

This work is licensed under a Creative Commons Attribution 4.0 International License.









