Influence of Electronic Factors on “Solvent-Free and Catalyst-Free Biginelli Reaction
Tanay Pramanik1*, Tariq Ahmad Wani2 and Ambrish Singh1
1Department of Chemistry, Faculty of Technology and Sciences, School of Civil Engineering, Lovely Professional University, P.O.Box 144411, India. 2Department of Chemistry, Faculty of Technology and Sciences, School of Science, Lovely Professional University, P.O.Box 144411, India.
DOI : http://dx.doi.org/10.13005/ojc/290354
Article Received on :
Article Accepted on :
Article Published : 28 Oct 2013
A series of Biginelli condensation were carried out employing urea, ethyl acetoacetate with a series of different derivatives of benzaldehyde. A green synthetic procedure was followed for performing the Biginelli reactions. The reaction conditions were found to be different for electron rich and electron deficient benzaldehydes. So the electronic effect has shown a significant impact on the reaction condition of the Biginelli condensation.
KEYWORDS:Benzaldehyde;Ethyl Acetoacetate;Urea;Electron donating and Electron withdrawing group;Biginelli reaction
Download this article as:| Copy the following to cite this article: Pramanik T, Wani T. A, Singh A. Influence of Electronic Factors on “Solvent-Free and Catalyst-Free Biginelli Reaction. Orient J Chem 2013;29(3). doi : http://dx.doi.org/10.13005/ojc/290354 |
| Copy the following to cite this URL: Pramanik T, Wani T. A, Singh A. Influence of Electronic Factors on “Solvent-Free and Catalyst-Free Biginelli Reaction. Orient J Chem 2013;29(3). Available from: http://www.orientjchem.org/?p=370 |
Introduction:-
The multicomponent condensation reaction has attracted a huge interest from the researchers in the field of organic chemistry for constructing complicated heterocyclic scaffolds1-3. The Biginelli reaction is one of the most popular multicomponent condensation that is used to do the coupling among urea, ethyl acetoacetate and aromatic aldehydes. In 1893 Biginelli reported the first synthesis of dihydropyrimidines of type 4 by a simple one-pot condensation reaction of ethyl acetoacetate (1), benzaldehyde (2), and urea (3)3b.
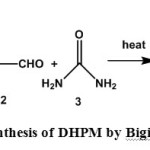 |
Scheme 1: Synthesis of DHPM by Biginelli reaction. Click here to View figure |
Dihydropyrimidine (DHPM) which is an N-contained heterocycle, attracted much attention due to its activities as anticancer, anti-inflammatory, antibacterial, antifungal, anthelmintic, and antitopoisomerase4 agent. DHPM is also known to be used as amidohydrolase, dihydropyrimidinase, and dehydrogenase5-7. Hence, the old acid catalyst Biginelli reaction has been used extensively varying all three reactant components, allowing access to a large number of DHPM derivatives3c. The conventional method of carrying out Biginelli reaction is to use a solvent (usually alcohol) and a catalyst. . So, investigation on this reaction was mainly focused on the exploration of catalyst and solvents4-6. A large varieties of catalyst like Fe(NO3)3. 9H2O7, TiCl48, InCl39has already been employed for Biginelli reaction.
The growing concern for the environment demands the development of eco-friendly green synthesis of Biginelli reaction. So the solvent-free and catalyst-free green chemistry of Biginelli reaction has been reported by the researchers10.
The mechanism of this Biginelli reaction as it’s reported by Sweet et al11 has made us think that the electronic effect of the aromatic aldehyde moiety may influence the nature, such as yield, reaction time and reaction temperature of this reaction.
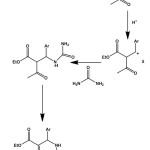 |
Scheme 2: Mechanism of Biginelli reaction. Click here to View figure |
Although Biginelli reaction has already been well studied by the researchers and it’s well documented in literatures since it was discovered by Pietro Biginelli in the year 1891, but our extensive literature search has revealed that the need of studying the effect of electronic factors on the solvent-free and catalyst-free Biginelli reaction still remains. So based on the research gap stated above, we herein report the green synthesis of a series of Biginelli condensation, carried out by employing urea, ethyl acetoacetate with a series of different electron rich and electron deficient benzaldehydes. Our work will also answer the research question that is “How the electronic factor can affect the nature of this reaction?”
Results and Discussion.
Synthesis:
In the 150ml round bottom flask the desired aldehyde (10-2moles) , ethylacetoacetate(10-2moles) and urea (10-2moles) were heated on the oil bath at desired temperature until a solid yellow colour product were obtained. After the formation of the product the compound was washed and filtered. All the compounds were characterized by IR and NMR. The melting points, IR spectra and NMR spectra of the compounds were matched well with literature reported data for the corresponding compounds.
Effect of Electronic factor on the reaction:
The catalyst-free and solvent-free Biginelli reactions employing ethyl acetoacetate, urea with a series of aromatic aldehydes were performed using standard literature reported procedure10. The variation of yield, reaction time and reaction temperatures with the variation of aromatic aldehydes are summarized in table-1.
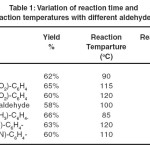 |
Table 1: Variation of reaction time and reaction temperatures with different aldehydes. Click here to View table |
From the data reported in table-1, it’s clearly evident that the electronic effect of the aromatic aldehyde has significant impact on the nature of the reaction. The percent yields of the products were almost comparable, so the percent yield did not change much with the change in aldehyde group. But it was interesting to note that by incorporating a nitro group into the phenyl ring of the aromatic benzaldehyde, the product was formed only after heating the reaction mixture at a higher temperature and for a longer time period compared to simple benzaldehyde. It’s not only the nitro group but also the chloro/ cyano group that has the similar effect on the reaction. Both the reaction time and the reaction temperature were higher for chloro/ cyano benzaldehyde compared to simple benzaldehyde. On the other hand, the product was formed by heating the reaction mixture at a comparatively lower temperature for a shorter period of time, if the phenyl ring of the aromatic aldehyde contains a electron donating group, such as methyl. In case of anisaldehyde, it’s the –I effect of the oxygen atom not the +M effect of the methoxy group which is predominant. As a result, the aromatic ring in this anisaldehyde becomes electron deficient; hence the reaction demands a longer time and higher temperature of heating to form the product compared to simple benzaldehyde.
So the product was formed at much lower temperature and at a shorter period of time for electron rich aromatic aldehyde. On the other hand, the solid product was formed only after heating the reaction mixture at a higher temperature for longer period of time if an electron deficient aromatic aldehyde was employed as a reactant. The reason can easily be understood by looking at the reaction mechanism. It can be seen from the mechanism stated above that a carbocationic intermediate ( compound 5 ) was formed during the course of the reaction. As we know that a carbocation is stabilized by electron donating group and destabilized by an electron withdrawing group, formation of the cationic intermediate (5) is facilitated by an electron donating group or with an electron rich aromatic aldehyde. On the other hand, the formation of this cationic intermediate (5) becomes difficult for an electron deficient aromatic aldehyde.
Thus an electron rich aromatic aldehyde increases the overall rate of the reaction by favoring the formation of the cationic intermediate (compound 5) and an electron deficient aromatic aldehyde decreases the overall rate of the reaction by disfavoring the formation of the cationic intermediate (compound 5).
Conclusion
In this work, a green synthetic procedure was followed for carrying out Biginelli reaction. A series of Bigielli reaction were performed using urea, ethyl acetoacetate with various electron rich and electron deficient aromatic aldehydes. It was observed that although the percent yields of the corresponding Biginelli products did not vary much by varying the aromatic aldehyde, the reaction time and reaction temperature varies significantly by varying the aromatic aldehyde.
Employing an electron rich aromatic aldehyde, the product formation was observed by heating the reaction mixture for a shorter period of time as well as at a lower temperature. Whereas, the product was formed only by heating the reaction mixture at a relatively higher temperature and for a longer period of time, if an electron deficient aromatic aldehyde was used as reactant.
So our work gives an insight about the effect of electronic factor on the green synthesis of Biginelli product.
References
- Wang R and Zai-Qun L., J. Org. Chem,, 77: 3952−3958 (2012)
- Yu J., Shi F., Gong L., Acc. Chem. Res., 44: 1156−1171 (2011)
- a) Dondoni A., Massi A. Acc. Chem. Res., 39: 451−463 (2006). b) Biginelli P. Gazz. Chim. Ital., 23: 360-416 (1893) c) Kappe C. O. Tetrahedron., 49: 6937-6963 (1993).
- Kappe C. O. Acc. Chem. Res., 33: 879−888 (2000).
- Kappe C. O. Eur. J. Med. Chem., 35: 1043−1052 (2000).
- Dondoni A., Massi, A. Mol. Diversity., 6: 261−270 (2003)
- Phukan M., Kalita M. K., Borah, R. Green Chem. Lett. Rev., 3: 329−334 (2010).
- Nagawade R R., Kotharkar S. A., Shinde D. B., Mendeleev Commun., 15: 150−151 (2005).
- Ranu B. C., Hajra A., Jana U. J. Org. Chem., 65: 6270− 6272 (2000).
- Ranu B. C., Hajra A., Dey S S., Org. Process Res. Dev., 6: 817−818 (2002).
- Sweet F., Fissekis J D., J. Am. Chem. Soc., 95: 7841-8749 (1973).

This work is licensed under a Creative Commons Attribution 4.0 International License.









