Environmental Effect / Impact Assessment of Industrial Effulent on Ground Water
Dr. Parmod Kumar
CCS University Meerut ,U.P. ( INDIA )
DOI : http://dx.doi.org/10.13005/ojc/290360
Article Received on :
Article Accepted on :
Article Published : 28 Oct 2013
In the present study the aim of investigation is physical and chemical parameters of ground water and soil. By selected Physical and chemical parameters it is found that (1).Biological oxygen demand (BOD) and chemical oxygen demand (COD) are directly proportional to each other where dissolved oxygen (DO) is indirectly proportional to BOD and COD. (2). Total dissolved solids, alkalinity and hardness are significantly higher in pre monsoon and winter season as compared to monsoon season.(3). High values of different parameters of ground water sources indicate the influence of industrial wastes on ground water.
KEYWORDS:pH;conductivity;DO.;BOD;COD;hardness;calcium hardness;magnesium;chlorides;total dissolved solids (T .D.S);alkalinity;sulfates;nitrates;sodium;potassium
Download this article as:| Copy the following to cite this article: Kumar P. Environmental Effect / Impact Assessment of Industrial Effulent on Ground Water. Orient J Chem 2013;29(3). doi : http://dx.doi.org/10.13005/ojc/290360 |
| Copy the following to cite this URL: Kumar P. Environmental Effect / Impact Assessment of Industrial Effulent on Ground Water. Orient J Chem 2013;29(3). Available from: http://www.orientjchem.org/?p=382 |
INTRODUCTION
Water, a natural resource, is one of the most common and unusual substance. The effect of water on almost everything in our environment is far more consequential than might be imagined. Water is called “the universal solvent” because of its extraordinary ability to dissolve more substances in greater quantity than any other liquid. The salinity of world’s oceans is a direct result of water’s ability to dissolve rock materials as water flows over land to the sea.
If the amount of waste increase beyond the limit of homeostasis of this system, then the problem of biological significance arises. Under such conditions, water becomes unfit for use. The contribution of chemical industry is evident range of marketable products like cosmetics, plastics, drugs, synthetic, fibres, paints, cleaning agents etc. Most of these chemicals ultimately find their way into fresh water resources via sewage. Some of them find their way into food chain, crippling the natural biotic organisms to extinction. Still others causes general pollution of water, which then becomes unfit for human use. Unrestrained release of heavy metals via discharge of industrial effluents, sewage and agro-chemicals into the water resources has not only rendered unusable but also has adversely affected the Gground water & aquatic life.
The speciation of effluent is likely to charge when these metals enter the aquatic environment. The dissolved cadmium in fresh water is predominantly present as either the free Cd+2 ion, Cd CO3 or CdS with minimal complexation with organic ligands. In contrast, copper is invariably complexed in polluted and unpolluted fresh water
Cadmium is a common pollutant in harbours surrounded with industrial installations. The effects of acute cadmium poisoning in humans are very serious. . Zinc is used primarily n galvanized metals and metal alloys. In addition, various inorganic Zn salts have numerous commercial uses.Zn oxide is used in rubber industry and ZnS is used in rayon manufacture, agriculture, Zn plating bombs, antiseptic, astringents etc. Zinc is an essential element with daily allowances ranging from 5 mg for infants and 15 mg for adults (NRC 1989).
In humans acutely toxic oral doses of Zinc cause several harmful effects. In the present study different concentrations of Fe, Zn and Cu were found in ground water samples from affected area. The possible reason may be the seepage of effluents and leach ate from waste water of Buria nala and from sludge dumped on site and in low lying area in the vicinity of the industry .The industry has been discharging waste since its establishment. Therefore, in due course of prolonged exposure to the disposed effluents, the prominent ground water resources have become polluted to a significant level.
HISTORICAL RESUME
Manivasakam (1976) reported physicochemical examination of water, sewage and industrial effluents, Kumar and Mishra (1990) studied the effect of sewage and paper mill effluents on ground water.
Water pollution caused by industrial effluents loaded with heavy metals is being considered as a serious problem and is attracting heavy metals in ground water of Manideep were conducted by Verma et al (1994). Metals such as Mn, Pb and Cu were found to be not within detectable limits in untreated water samples while iron values were found to show variation from station to station. Vangosh and Rosenthal (1994) found that the deterioration of ground water in Israel is caused by numerous natural processes such as encroachment of sea water, by subsurface dissolution of soluble salts etc. The ground water quality of Kirakund, an industrial town in Sambalpur district of Orissa has been studied by Badapanda et al (1996). Kulkarni and Dharwadkar (1997) studied the physicochemical and bacteriological parameters of bore well water samples of industrial area and a residential area near to it in Aurangabad city .The percentage of bacteriological contamination was higher in industrial area as compared to the residential area.
MATERIAL AND METHODS
WATER QUALITY ANALYSIS:
International standards given by WHO for drinking water in 1971 and further guidelines for drinking water in 1983 together with European commission directive on quality of water to be used for human consumption provide criteria to assess drinking water quality.
On the basis of physical, chemical and biological impurities present in ground water, quality parameters are broadly classified into two categories given below:
a. Physical parameters
b. Chemical parameters
Physical parameters are based on physical impurities of water affecting physical characteristics as observed through sense of sight, touch, taste and odour. Suspended solids, turbidity, taste and odour, colour and temperature fall into this category .These parameters have no direct relationship with health but health but may have indirect consequences. Chemical parameters are based on various chemical impurities in water. Chemical impurities in water are directly related to health. The parameters include total dissolved solids, alkalinity, hardness, dissolved inorganic matter which are of greater concern in water quality management. Water is an excellent medium to support thousands of biological species.
Ground water pollution and Physico-chemical parameters:
Quality of ground water is declining due to heavy industrialization and human activities. The water pollution is mainly due to indiscriminate disposal of industrial wastes both on land and surface water channels. Jais et al (1993) assessed the physicochemical and bacteriological quality of ground water and surface water of Vijaypur(Bhopal) and around Vijaypur district Guna (M.P.) respectively. Many other workers made useful studies on ground water pollution and have shown that it is polluted to varying degrees at different places.
Mehta and Trivedi (1993) studied ground water contamination in many regions of Bhavnagar recharge areas affected by agricultural, residential and industrial activities.
RESULTS
Physicochemical characteristics of water in Buria nalla:
Site 1 – Buria nalla:
The physicochemical characteristics of Buria nallah (Site I) water of different seasons showed water was alkaline. The pH fluctuated narrowly between 8.14 and 8.98. Highest pH and dissolved oxygen were recorded in the monsoon season respectively. BOD and COD were lowest in the monsoon season. Total dissolved solids were in excessive amounts and not much variable in different seasons. Water was quite alkaline through the year; alkalinity was lowest in monsoon .Hardness and calcium hardness were lowest in the monsoon season. Electrical conductance showed low value in monsoon season. Water was saline; salinity was highest during the pre-monsoon season while in monsoon and post-monsoon season it was comparatively lower. Sodium concentration was higher than potassium and not much variable. Magnesium level was higher in pre-monsoon. In the level of sulfate and nitrate fluctuations was narrow (Table 1a-1c).
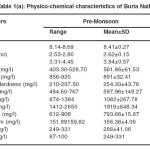 |
Table 1(a): Physico-chemical characteristics of Buria Nalla Click here to View table |
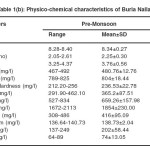 |
Table 1(b): Physico-chemical characteristics of Buria Nalla Click here to View table |
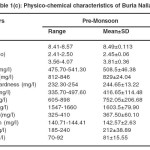 |
Table 1(c): Physico-chemical characteristics of Buria Nalla Click here to View table |
Site 2 – Buria nalla:
Buria nallah water collected during different months of the year reveal that there was not much variation in pH. It was alkaline ranging between 8.14 and 8.91. Alkalinity ranged between 477.56 and 625.75. Narrow variation was noticed in the hardness. Highest values of calcium hardness were noticed in pre monsoon season. Electrical conductance with narrow fluctuations ranged between 2.17 and 3.16 Chloride content was lowest during the monsoon season.. Magnesiumlevel varied in a narrow range. Sulfate and nitrate contents fluctuated in a narrow range and were also above the I.S.I. standards (2a-2b)
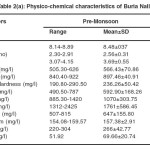 |
Table 2(a): Physico-chemical characteristics of Buria Nalla Click here to View table |
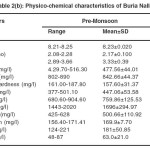 |
Table 2(b): Physico-chemical characteristics of Buria Nalla Click here to View table |
Physicochemical characteristics of soil sample
Site 1- Buria Nalla:
pH of soil sample fluctuated between 9.01and 9.11 the mean pH did not alter much in different seasons. Electrical conductance and alkalinity level varied in a narrow range. Alkalinity was lowest in the monsoon. Moisture content was approximately same in all seasons except in monsoon month, where it was highest.. Sodium with less variation was lowest in post-monsoon months. Sulfate concentration was near about same in all seasons. (table 4.15). (Buria Nalla, Industrial Area)
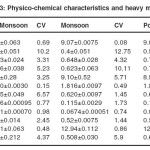 |
Table 3: Physico-chemical characteristics and heavy metals Click here to View table |
Site 2 –Residence:
Soil samples collected from site 3 during different months of the year reveal that there was not much variation in pH. It was alkaline ranging between 8.24and 8.56. Similarly much variation was not observed in electrical conductance and alkalinity in different seasons. Alkalinity ranged between 0.415and 0.477. Maximum was in pre-monsoon. Chloride levels were minimum during the monsoon period. Moisture content was highest in monsoon and lowest in pre-monsoon. Sodium concentration was higher than potassium with minimum variation. Potassium showed highest concentration in winter season. Sulfate and phosphate were lowest in monsoon and post-monsoon (Table 4.17)
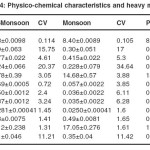 |
Table 4: Physico-chemical characteristics and heavy metals Click here to View table |
Heavy metals in water and soil: Analysis of Buria nala in different seasons revealed that heavy metals detected were iron, copper and zinc. Iron was recorded more in comparison to other metals. It ranges between 0.80-1.98. Maximum concentration of iron was recorded in pre-monsoon.. Zinc concentration was more in comparison to copper. Both copper and zinc were found highest (0.149and 0.17) in the pre monsoon season.In ground water samples iron concentration was found more in comparison to zinc and copper. Zinc values ranges between (0.12-0.49) and was maximum during the pre monsoon season in all samples.
Iron was recorded between (0.12-0.87) with minimum values in monsoon in all samples. Iron was found less in ground water. Zinc and copper values were also recorded high in comparison to Buria nala water.
CONCLUSION
World today is facing environmental catastrophe and the problem is how to protect the global environment which is being threatened by the growing population, rapid strides in industrialization, high density of automobiles, excessive use of agro -chemicals, process of urbanization and extensive uses of natural resources. Addition of effluents o the aquatic ecosystem is a world wide problem and is more acute in industrialized countries where million of tones of pollutants are discharged into ground water. In turn it effects human life .
REFERENCES
- Abbasi, S.A., Arya, D.S., Hameed A.S. and Abbasi N. (1996). Water quality of a typical river of Kerala Punnurpuzha. Poll. Res., 15(2): 163-166.
- Agrawal, A., Shrivastava, R,M, and Verma, N.K. (1995). Chemical study of the effect of industrial effluents on surface and ground water surrounding Mandideep, District Raisen, M.P. (India). Oriental J.Chem., 11 :270-271.
- Ajmal, M. and Munshi, D. (1991). Scanning electron microscopic evaluation of changes on the morphology of blood cells of an Indian major carp, Catla catla following exposure to copper. J. Environ. BioI., 13(4): 297-301.
- Ali, M. and Tiwari, T.N. (1988). Preliminary survey of some toxic metals in ground water of Rourkela, Indian J. Env. Prot., 8: 338-341.
- Ambrose, T. and Vincent, S. (1994). Chromium induced anaemia in freshwater carp, Catla (Ham.). J.Natcon, 6:67-71.
- Amiard, I.C., Amiard- Triquet, C., Berthel, B. and Metayer, C. (1987). Comparative study of the patterns of bioaccumulation of essential (Cn, Zn) and coastal organisms. J.Exp. Mar. Biol.Ecol., 106:73-89.
- APHA (1985). Standard methods for the examination of water and waste water. American Public Health Association. Washington, D.C.
- Aranzo, R.M., Arribas, R.M., Luveena, F. and Pases, R. (1989). Relation between Aeromonas and fecal coliforms in fresh water. I Appl. Bact., 67:213-217.
- Badapanda, R.K., Pati, S.S. and Sahu, S.K. (1996). Ground water quality of an industrial town in Orissa, India. I. Ecotoxicol,. Environ. Monit. 6(3-4), 195-198.
- Bahaduria, S. (1997). Characterization of agricultural soils irrigated with petroleum refinery effluent: Physico-chemical analysis. Iouornal of Environment & Pollution, 4:295-302.
- Baruah, B.K. and Das, M. (1998). Study on the effect of paper mill emuent on physicochemical characteristics of soil. I. Natcon, 10: 101-106.
- Bowonder, B. and Chandamnas, M. (1973). Water pollution and Environment. Ind. Chem. Man, 10:9-19.
- Dash, S.C., Behera, D.K. and Rout, S.P. (1992). Water pollution from major industries in Rourkela area. Indian J. Env Prot., 12:890-899.
- Dhanapal, S., Sivakumar, A.A., Logankumar, K. and Aruchami, M. (1990).
- Fetter C.W. (1988). Applied hydrogeology, CBS Publishers, New Delhi, India. 592.

This work is licensed under a Creative Commons Attribution 4.0 International License.









