Curing Study of Epoxy/clay Nanocomposite by a New Aromatic Diamine Functional Curing Agent
Seyed Mahdi Saadati1 , Mohammad Hossein Mosslemin1 and Hossein Behmadi2
1Departmentof Chemistry, Yazd Branch, Islamic Azad University, Yazd, Iran 2Departmentof Chemistry, Mashhad Branch, Islamic Azad University, Mashhad, Iran
DOI : http://dx.doi.org/10.13005/ojc/290333
Article Received on :
Article Accepted on :
Article Published : 28 Oct 2013
The curing behavior of an epoxy/clay nanocomposite system composed of diglycidyl ether of bisphenol A (DGEBA) epoxy resin with a new aromatic diamine curing agent and organically modified clay was investigated. Epoxy/clay nanocomposite samples were prepared by mixing DGEBA, different percentages of nanoclay (1, 3, 5, 7, and 9%) and 4-(4-(2,6-diphenylpyridin-4-yl)phenoxy)benzene-1,3-diamine (DAP). The optimum ratio of the epoxy/clay nanocomposite system (3%) was investigated by total enthalpy of the curing reaction from differential scanning calorimetry (DSC) thermograms. The kinetic parameters such as activation energy, pre-exponential factor, and rate constant were obtained from DSC data.
KEYWORDS:Nanocomposite;Aromatic Diamine;Epoxy resin;DSC
Download this article as:| Copy the following to cite this article: Saadati S. M, Mosslemin M. H, Behmadi H. Curing Study of Epoxy/clay Nanocomposite by a New Aromatic Diamine Functional Curing Agent. Orient J Chem 2013;29(3). doi : http://dx.doi.org/10.13005/ojc/290333 |
| Copy the following to cite this URL: Saadati S. M, Mosslemin M. H, Behmadi H. Curing Study of Epoxy/clay Nanocomposite by a New Aromatic Diamine Functional Curing Agent. Orient J Chem 2013;29(3). Available from: http://www.orientjchem.org/?p=327 |
INTRODUCTION
Nanoclays have been used as fillers in polymer systems because of the improved mechanical properties1–4, thermal stability4–7, gas barrier properties8, and flame retardant properties of the resulting polymer composites7,9. Fiber-reinforced composites are a type of engineering material that has exhibited high strength–weight and modulus–weight ratios, even compared with some metallic materials. Many industries use unidirectional laminated composites, consisting of a stack of glass fibers oriented in the same direction, in applications that exploit their high longitudinal strength when the same properties are not required in the transverse direction.
A method to improve the physical properties of a polymer is the addition of reinforcing particles or fibers to the polymer to form composite materials. Owing to the development of high-strength and light-weight fibers, such as carbon fiber and silicon carbide fiber, the fiber-reinforced composites have been widely used. The recent development of polymer nanocomposites containing nanometer scale fillers, such as layered silicates or carbon nanotubes, provides a new promising opportunity in composite materials10–12.
Layered materials are well suited for the design of hybrid composites, because their lamellar elements have high in-plane strength and a high aspect ratio13. The smectite clays (e.g. montmorillonite) and related layered silicates are the materials of choice for polymer nanocomposite design, because they cannot only be obtained easily from nature at low cost but can also be modified chemically to make themselves compatible with organic polymers. Each silicate layer has a lateral dimension of 200–2000 nm and a thickness of about 1 nm. The stacking of silicate layers forms tactoids that are typically 100–1000 nm thick. The cationexchange property of smectite clays is an important aspect of their use in nanocomposite formation14.
The overall properties of a polymer/clay nanocomposite material are determined not only by the parent components but also by the composite phase morphology and interfacial properties. Nanocomposites usually exhibit improved performances compared to conventional microcomposites, owing to their unique phase morphology and improved interfacial properties13–18. Major differences between conventional microcomposites and nanocomposites result from the fact that the latter has much larger surface (or interface) area per unit volume. Since many important chemical and physical interactions are governed by surfaces, a nanostructured composite can have substantially different properties from a microstructured composite of the same composition19, 20.
Two types of polymer/clay nanocomposite are possible. Intercalated nanocomposites are formed when some organic molecules are inserted between the silicate layers. Exfoliated nanocomposites are formed when the silicate layers are individually dispersed in the polymer matrix. Exfoliated nanocomposites show greater phase homogeneity than do intercalated nanocomposites. This structural distinction is the primary reason why the exfoliated structure is especially effective in improving the reinforcement and other performances of polymer/clay nanocomposite materials14. The ability of smectite clays to improve the mechanical properties of an engineering plastic (Nylon 6) was first demon-strated by Toyota researchers21–23. The key to this extraordinary performance of Nylon 6/clay hybrids was the complete dispersal exfoliation of the silicate layers in the polymer matrix.
The broad interest in epoxy resins originates from the extremely wide variety of chemical reactions that can be used for the curing and different properties that results24,25. The chemistry is unique among the thermosetting resins. Furthermore, knowledge of the chemistry involved permits the user to cure the resins over a wide range of temperatures and to control the degree of crosslinking26. The latter plays an important role in the physical properties. Depending on the chemical structure of curing agent and the curing conditions, it is possible to obtain toughness, chemical resistance, mechanical properties ranging from extreme flexibility to high strength and hardness, high adhesive strength, good heat resistance and high electrical insulation. Uncured, the resins have a variety of physical forms, ranging from low viscosity liquids to tack free solids that, along with the curing agents, afford the fabricator a wide range of processing conditions. As a result of versatility these products have found use in protective coatings, adhesives for most substrates, body solders and caulking compounds, flooring tooling compounds for molds, stamping dies and patterns, foams, potting and encapsulation compounds, low-pressure molding resins, and glassreinforced plastics27–30.
Curing kinetics of epoxy resins have been studied with different techniques, such as Fourier transform infrared spectroscopy (FTIR)31, near infrared spectroscopy32, and differential scanning calorimetry (DSC)33–35. DSC is the most widely used experimental technique to obtain the degree and reaction rate of cure of thermosetting resins and to determine the ∆H of curing reaction.
In this paper, the study of curing kinetics of diglycidyl ether of bisphenol A (DGEBA)/Clay Nanocomposite System, with new curing agent 4-(4-(2,6-diphenylpyridin-4-yl)phenoxy)benzene-1,3-diamine (DAP) has been reported along with thermal behavior of cured epoxy.
MATERIALS AND METHODS
Material
The epoxy compound used in this study was diglycidyl ether of a bisphenol A-based epoxy (DGEBA) provided from Shell Chemical Co., Epon 828, with an epoxy equivalent weight (Epox.Eq.w) of 185. The organophilic smectite clay (Cloisite 30B, a chemically modified montmorillonite) was supplied by Southern Clay Products, USA; and all other chemicals were purchased either from Merck or Fluka. 4-(4-(2,6-diphenylpyridin-4-yl)phenoxy)benzene-1,3-diamine (DAP) as an curing agent was synthesized according to the literature as shown in Fig. 136.
Apparatuses
FTIR spectra were recorded with potassium bromide plates on a Bruker Tensor 27 over the range of 400–4,000 cm-1. DSC was recorded on a Metteler Toledo, DSC822e under nitrogen atmosphere (20 cm3/min). Thermogravimetric analysis (TGA) was performed with TGA 50 from Shimadzu analyzer at a heating rate of 10°C/min under air atmosphere and in the temperature range of 30–1,000 ºC.
Sample Preparation and Optimum Formulation
A certain amount (1, 3, 5, 7 and 9 percent of epoxy resin) of the nanoclay was mixed with the epoxy resin andultrasonic dispersion treatment in an ultrasonic bathat ambient temperature was then utilized for 3 h to producehomogenous blends. Then, the binary mixture of the epoxy resin and the nanoclay was mixed again with the curing agent (DAP) by stoichiometry (58 phr), and stirred until the systems became homogeneous. The mixtures were poured into Teflon moulds, which were highly polished. The mixtures for DSC use and other applications were stored in a refrigerator at -20°C.
DSC Measurement
In order to investigatethe curing behavior of the epoxy/clay nanocompositesystem, about 5 mg of the ternary mixture before curing was placed in an aluminum liquid sample pan, and the sample pan was placed in the DSC sample cell at ambient temperature and an empty pan was also placed in the DSC reference cell, and it was heated at constant heating rates of 5, 10, 15, and 20°C/min from room temperature to 400°C under nitrogen purge gas.
TGA Studies
Thermal stability of the cured sample was measured in a temperature range of 50–1,000°C at the heating rate of 10°C/min under air atmosphere. About 3 mg of the cured nanocomposite sample was cut into small discs for TGA tests.
RESULTS AND DISCUSSION
The curing agent (DAP) was prepared according to literature as shown in Fig. 136. To determine the optimum ratio of the reacting components, the effect of nanoclay content on the total enthalpy of the curing reaction was investigated and the optimal amount of nanoclay was 3 mol%. The results are shown in table 1. Accordingly, the level of nanoclay was set to be 3% as the result of maximum reaction heat for this composition.
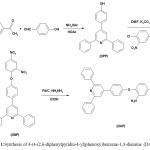 |
Fig. 1:Synthesis of 4-(4-(2,6-diphenylpyridin-4-yl)phenoxy)benzene-1,3-diamine (DAP). Click here to View figure |
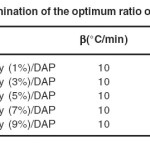 |
Table 1: Determination of the optimum ratio of the nanoclay. Click here to View table |
Kinetic Study
Many equations were developed to investigate the cure kinetics of the epoxy system, including the nth order reaction model, the autocatalytic reaction model, and the diffusion control model37. The recorded heat flow data were processed to obtain the fractional conversion, α, and the rate of reaction under the basic assumption so that the heat flow (dH/dt) is proportional to the reaction rate, dα/dt. For an exothermic reaction, Eq. 1 applie.
![]()
where dα/dt is the rate of the kinetic process, dH/dt is the measured heat flow, and ΔHTot is the enthalpy of the curing reaction. The rate of the kinetic process in the kinetic analysis can be described as
![]()
where K(T) is a temperature-dependent reaction rate constant; f(α) is a function of the fractional conversion38, α, and dα/dt is the rate of conversion at a given time and temperature.The fractional conversion was calculated by Eq. 3:
![]()
where the subscript t indicates the value of conversion at time t. K(T) is assumed to have an Arrhenius temperature dependence.
![]()
where A is the pre-exponential factor, Ea is the activation energy, and R is the gas constant. Therefore, the Ea value can be determined by an isoconversional method under the assumption of f(α) being the same at different heating rates (β) when the value of α is the same that is, in Eq. 5, f(α) does not depend on the heating rate at a fixed value of the variable α. From the logarithmic form of Eq. 2, we can have
![]()
Figure 2 shows dynamic DSC curves for DGEBA/nanoclay (3%)/DAP at different heating rates. The exothermic peak decreased from 175.2°C at heating rate of 20°C/ min to 138.7°C at heating rate of 5°C/min. Slower heating rates give more time to the sample to cure and the exothermic peak shifts toward lower temperatures. All DSC scanning data obtained for DGEBA/nanoclay (3%)/DAP at different heating rates (5, 10, 15, and 20°C/min) are summarized in Table 2.
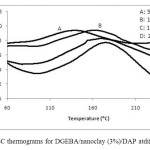 |
Fig. 2: Dynamic DSC thermograms for DGEBA/nanoclay (3%)/DAP atdifferent heating rates. Click here to View figure |
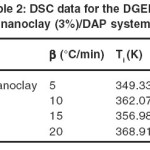 |
Table 2: DSC data for the DGEBA/nanoclay (3%)/DAP system. Click here to View table |
The exothermic transition was characterized concerning the following parameters: Tp (the temperature of the peak position of the exotherm), Ti (the onset temperature of the exotherm peak) and ΔH (the heat of curing that calculated from area under the exothermic transition). The kinetic parameters of curing reaction were obtained by the data summarized in Table 3 and using Kissinger, Ozawa, and Isoconversional methods.
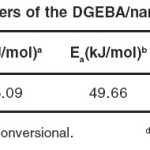 |
Table 3: Kinetic parameters of the DGEBA/nanoclay (3%)/DAP system.: Click here to View table |
Kissinger derived the following equation[39, 40] for the time when the temperature varies with time at a constant heating rate:
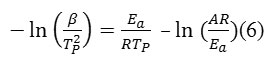
Where β is the heating rate, Tp the temperature at which dα/dt is maximum, and R the gas constant. This method gives a relatively accurate activation energy (Ea) and preexponential factor (A) by calculating the relationship between and 1/Tp. According to the Kissinger equation (Eq. 6), the linear plots of vs. 1/Tp is shown in Fig. 3. The Ea and A values were calculated from the slope and from the y-intersect of the linear plots, respectively, and are listed in Table 3. The values of A and Ea were introduced to the following Arrhenius equation (Eq. 7) to get rate constant at a selected temperature as listed in Table 3.

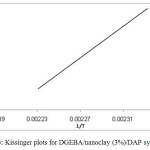 |
Fig. 3: Kissinger plots for DGEBA/nanoclay (3%)/DAP systems. Click here to View figure |
Ozawa–Flynn–Wall method based on Doyle’s approximation39, 41 is an alternative method for calculating Ea and is expressed as follows:

Using Ozawa method, (Eq. 8), plots of ln(β) vs. (1/Tp) gave straight lines, as shown in Fig. 4, with slope of 1.052Ea/R. The calculated Ea values are listed in Table 3.
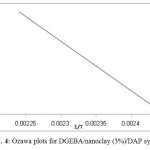 |
Fig. 4: Ozawa plots for DGEBA/nanoclay (3%)/DAP systems. Click here to View figure |
The Ea values obtained in this study by Kissinger and Ozawa methods (Table 3) are almost in the same range of the Ea values reported by other workers42,43 for epoxy–diamine systems. DSC analysis has shown23 that etherification occurs at elevated temperatures once all the primary amines are exhausted. High activation energies ([150 kJ/mol by DSC) are characteristics of this process. Therefore, the primary amine addition is considered as a single step taking place during the cure reaction and controls the overall heat release at lower conversions. In the above case, the obtained Ea value is supposed to be constant throughout the entire period of the amine conversion. This idea does not correspond to the autocatalytic mechanism of the process. The Ea value does not remain constant with the progress of cure reaction when compared with the very beginning of the process where the nonautocatalyzed reaction occurs. Therefore, the low values of Ea listed in Table 3, most likely represent some average values between the Ea values of nonautocatalyzed and autocatalyzed reactions.
The Kissinger equation gives only one Ea from Tp data. A more complete determination of Ea at any selected conversion can be calculated by the isoconversional equation44,45 which is:
![]()
where E, β, and R are the same terms as in the Kissinger equation and T is the temperature for a selected conversion at each heating rate. Activation energies can be obtained from the slope of the straight line of lnβ vs. (1/Tp). Isoconversional kinetic analysis offers a viable alternative in this situation. The basic idea behind this type of analysis is that the reaction rate at a constant conversion depends only on the temperature. To perform Isoconversional analysis, the original DSC data (Fig. 2) were transformed into the conversion form (αi) vs. temperature (Ti) for each ith heating rate. Conversions were determined as fractional areas of a DSC peak. The resulting dependence of αi vs. Ti for each heating rate is shown in Fig. 5 for DGEBA/nanoclay (3%)/DAP system. Plotting ln(β) vs. 1/T, using Eq. 9, produced straight lines as shown in Fig. 6. The Ea value for each conversion was obtained from the slope of the straight line.
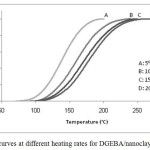 |
Fig. 5: Conversion curves at different heating rates for DGEBA/nanoclay (3%)/DAP system. Click here to View figure |
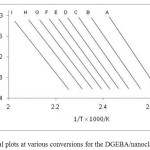 |
Fig. 6: Isoconversional plots at various conversions for the DGEBA/nanoclay (3%)/DAP system. Click here to View figure |
TGA Studies
The thermal stabilities of the cured resin were evaluated by TGA in air atmosphere at the heating rate of 10°C/min. The obtained results are summarized in Table 4 and the representative TGA curves are shown in Fig. 7. The temperatures for 10% weight loss (T10) that are important criterion for evaluating thermal stability were about 368.9°C and 373.1°C for DGEBA/DAP and DGEBA/nanoclay (3%)/DAP respectively. Therefore, nanocomposite system (DGEBA/nanoclay (3%)/DAP) is comparatively more thermally stable than DGEBA/DAP system.
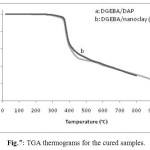 |
Fig.7: TGA thermograms for the cured samples. Click here to View figure |
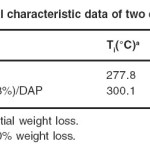 |
Table 4: Thermal characteristic data of two cured systems. Click here to View table |
CONCLUSION
In this study, epoxy composite samples were prepared by mixing of the epoxy resin, nanoclay (1, 3, 5, 7, and 9%) and stoichiometric amount of the 4-(4-(2,6-diphenylpyridin-4-yl)phenoxy)benzene-1,3-diamine (DAP) with vigorous stirring until the systems became homogeneous. The optimum ratio of the epoxy/clay nanocomposite system (3%) was investigated by total enthalpy of the curing reaction from differential scanning calorimetry (DSC) thermograms. Curing kinetics of DGEBA/nanoclay (3%)/DAP system by non-isothermal DSC with three methods, Ozawa, Kissinger, and Isoconversional, was studied. The kinetic parameters, Ea, A, and K were determined.
REFERENCES
- Chen, C.and Curliss, D., J. Appl. Polym. Sci.90:2276-2287(2003).
- Kinloch, A. J.and Taylor, A. C., J. Mater. Sci. Lett.22:1439-1441 (2003).
- Haque, A., Shamsuzzoha, M., Hussain, F.and Dean, D., J. Compos. Mater.37:1821-1837 (2003).
- Zhang, Y., Lee, J. H., Jang, J.and Nah, C. W., Composites: Part B35:133-138 (2004).
- Nah, C., Han, S.H., Lee, J.H., Lee, M. H., Lim, S. D. and Rhee, J. M., Composites: Part B25:125-131 (2004).
- Hussain, M., Varley, R., Mathys, J.Z., Cheng, Y. B.andSimon, G. P., J. Appl. Polym. Sci.91:1233-1253 (2004).
- Becker, O., Varley, R. J.and Simon, G.P., Eur. Polym. J.40:187-195 (2004).
- Yano, K., Usuki, A.and Okada, A.,J. Polym. Sci. A: Polym. Chem.35:2289-2294 (1997).
- Hussain, M.and Simon, G.P., J. Mater. Sci. Lett.23:1471-1475 (2003).
- Andre, L.and Lichtenhan, J. D., J. Appl. Polym. Sci.73:1993(1999).
- Mark, J. E.and Bianconi, P. A., “Hybrid Organic–InorganicComposites” (ACS Symposium Series 585), American Chemical Society, Washington, DC (1995).
- Miller, B.,Plast. Formulat. Compound3:30(1997).
- Pinnavaia, T. J.,Science220:365-371 (1983).
- Wang, Z.J.and Pinnavaia, T. J.”Epoxy-Clay Nano-composites“, Wiley, New York, 127(2000).
- Messersmith, P. B.and Stupp, S.I., J. Mater. Res.7:2599-2611 (1992).
- Okada, A.and Usuki, A.,Mater. Sci. Eng. C3:109-115 (1995).
- Giannelis, E. P.,Adv. Mater.8:29-35 (1996).
- Novak, B. M.,Adv. Mater.5:422-433 (1993).
- Jyi-Jiin, L.and Daniel, I. M., Compos. Sci. Technol.63:1607-1616 (2003).
- Komarneni, S., J. Mater. Chem.2:1219-1230 (1992).
- Usuki, A., Kawasumi, M., Kojima, Y., Okada, A., Kurauchi, T.and Kamingatio, O., J. Mater. Res.8:1174-1178 (1993).
- Usuki, A., Kojima, Y., Kawasumi, M., Okada, A., Fuku-shima, Y., Kurauchi, T.and Kamingatio, O., J. Mater. Res.8:1179-1184 (1993).
- Kojima, Y., Usuki, A., Kawasumi, M., Okada, A., Fuku-shima, Y., Kurauchi, T.and Kamingatio, O., J. Mater. Res.8:1185-1189 (1993).
- Potter, W. G., “Epoxide Resins“, Springer-Verlag, New York(1970).
- Lee, H.and Neville, K. “Handbook of Epoxy Resin“, McGraw-Hill Book Co.,New York(1976).
- Sedlacek, B.and Kahovec, J., (Eds.), “Crosslinked Epoxies“, de Gruyter, Berlin (1987).
- Balcerzak, E. A., Janeczek, H., Kaczmarczyk, B., Bednarski, H., Sek, D.and Miniewicz, A., Polymer45:2483-2493 (2004).
- May, C. A. (Ed.), “Epoxy Resins-Chemistry and Technology“, second ed., Marcel Dekker, New York(1988).
- Opaliki, M., Kenny, J.and Nicolais, N., J. Appl. Polym. Sci.61:1025-1037 (1996).
- He, X., Conner, A. H.and Koutsky, J. A., J. Polym. Sci.: Part A: Polym. Chem.30:533-542 (1992).
- Omrani, A., Simon, L.C., Rostami, A. A.and Ghaemy, M., Int. J. Chem. Kinet.40:663-669 (2008).
- Mijovic, J.and Andjelic, S., Macromolecules28:2787-2796 (1995).
- Ghaemy, M., Barghamadi, M.and Behmadi, H., Iran. Polym. J.15:375-383 (2006).
- Ghaemy, M., Amini-Nasab, S. M.and Barghamadi, M., Polym. Comp.29:165-172 (2008).
- Ghaemy, M.and Barghamadi, M., J. Appl. Polym. Sci.112:1311-1318 (2009).
- Ghaemi, M., Alizadeh, R.and Hashemi Nasr, F., J. Appl. Polym. Sci.118:3407-3415 (2010).
- Barghamadi, M.,Polym. Compos.31:1465-1473 (2010).
- Ampudia, J., Larrauri, E., Gil, E. M., Rodriguez, M.and Leon, L. M., J. Appl. Polym. Sci.71:1239-1245 (1999).
- Doyle, C. D., Nature207:290-291 (1965).
- Kissinger, H. E., Anal. Chem.29:1702(1957).
- Ozawa, T., J. Therm. Anal. Calorim.2:301-324 (1970).
- Vyazovkin, S.andSbirrazzuoli, N., Macromolecules29:1867-1873 (1996).
- Girard-Rejdet, E., Riccardi, C. C., Sautereau, H.and Pasault, J. P., Macromolecules28:7599-7607 (1995).
- Salla, J. M., Ramis, X., Morancho,J. M.and Cadenato, A., Thermochim. Acta.388:355-370 (2002).
- Zhou, T. L., Gu, M. Y., Jin,Y. P.and Wang, J. X., J. Polym. Sci. A Polym. Chem.44:371-379 (2006).

This work is licensed under a Creative Commons Attribution 4.0 International License.









