Extraction and Antioxidative Activity of Essential Oil From Star Anise (Illicium verum)
Y. C. Wong*, P. P. Lee and W.A. Wan Nurdiyana
Faculty of Agro Based Industry, Universiti Malaysia Kelantan, Jeli Campus, Locked Bag 100, 17600 Jeli, Kelantan, Malaysia.
DOI : http://dx.doi.org/10.13005/ojc/300329
Article Received on :
Article Accepted on :
Article Published : 27 Aug 2014
Star anise (Illiciumverum) essential oil was extracted using solvent extraction method. The extraction yields and antioxidant activities of essential oils at different extraction times (1, 3, 5, 7 and 9 days) and temperatures (30, 40, 50, 60, 70 °C) were studied. The results showed that the highest yield of essential oil was 8.56 % by extracting star anise at 60 ⁰C for 7 days.The antioxidant activities of the extracted star anise essential oils were investigated using 2, 2-diphenyl-1-picrylhydrazyl (DPPH) assay on Thin Layer Chromatography (TLC) plates and DPPH radical scavenging method. The results showed that at least two different bands with antioxidant activity with different polarity were appeared on the TLC plates after spraying with DPPH and incubated for 30 minutes. The highest antioxidant activity of star anise essential oil was obtained when the sample was extracted at 60 ⁰C for 1 day (EC50 value = 0.089±0.05 mg/ml). HPLC analysis showed that the concentration (%) of trans-Anethole present in the essential oils extracted at varied extraction times and temperatures was ranged from 77.29 % to 91.87 %.Gas Chromatography-Mass Spectrometry (GC-MS) analysis was also done on a sample of star anise essential oil and a distinctive peak at retention time 13.84 minutes with peak area 100% was found to be Estragole compound. Anethole compound was also found to be present at two peaks.
KEYWORDS:solvent extraction; star anise essential oil; extraction yield; antioxidant activity; trans-Anethole
Download this article as:| Copy the following to cite this article: Wong Y. C., Lee P. P, Nurdiyana W. A W. Extraction and Antioxidative Activity of Essential Oil From Star Anise (Illiciumverum). Orient J Chem 2014;30(3). |
| Copy the following to cite this URL: Wong Y. C., Lee P. P, Nurdiyana W. A W. Extraction and Antioxidative Activity of Essential Oil From Star Anise (Illiciumverum). Orient J Chem 2014;30(3). Available from: http://www.orientjchem.org/?p=4376 |
Introduction
Long term and extensive use of synthetic antioxidant such as butylatedhydroxytoluene (BHT) and butylatedhydroxyanisole (BHA) have been proven to cause carcinogenic effects in living organisms [1][2][3]. Due to the potential side effects of synthetic antioxidants, essential oil which derived from organic products can be served as an alternative source for the further improvement of synthetic antioxidant. Star anise (Illiciumverum) is a small star-shaped fruit of an evergreen of the Illiciaceae family[4]. This fruit is classified as a spice and it looks like a symmetrical eight-pointed star [5]. Essential oil of star anise has a sweetish, anise flavour and a highly aromatic odour. Due to its high level of phenolic volatile oils, star anise essential oil could be used as a potential alternative antioxidant for synthetic antioxidant.
The objectives of this study were to extract essential oil from star anise using solvent extraction method, to determine the yields and antioxidant activities of essential oils at different extraction times and temperatures, to determine the quantity of trans-Anethole present in star anise essential oil using high performance liquid chromatography (HPLC), as well as tovalidate the presence of Anethole in star anise using Gas Chromatography–Mass Spectrometry (GC-MS).
Materials and Method
Collection of star anise
Star anise dried fruits were purchased from Tong Chun Tang traditional medicine store, Batang Kali, Selangor. The star anise fruits originated from Guangxi province, China. Star anise fruits were stored in plastic bags covered with newspapers to protect them from direct light. Finally, they were kept at room temperature.
Soaking of star anise
50g of dried star anise fruits were weighed using analytical balance (Model: ATX 224, Shimadzu, Japan). The whole star anise fruits were divided into separated carpels and were blended using blender (Model: MX-800S, Panasonic) for 20 seconds. The ground star anise was dissolved in 250 ml of absolute ethanol (ratio of crushed star anise to the ethanol was 1:5, w/v) for a given time (1, 3, 5, 7 and 9 days) at different temperatures (30, 40, 50, 60 and 70 °C).
Extraction of essential oil from star anise
This method was adapted from the work of Thuat and Ngoc (2010) with some modifications [6]. After the extraction time, the solution and solid part were separated by filtration using Whatman filter paper. The brown coloured filtrate was concentrated to volume of 100 ml using rotary evaporator (Model: Hei-vap Precision MLG3B, Germany) at 40 °C. The 100 ml concentrate was mixed with 100 ml of petroleum ether and 100 ml of dH2O. The mixture was shook vigorously. After some time of settling, the petroleum ether solution (the upper layer) was dried over anhydrous Na2SO4. The petroleum ether solvent was removed using rotary evaporator to obtain essential oil. All the essential oils were stored in bijou glass bottles in dark condition at 4 °C before analysis.
Qualitative test using DPPH on TLC
All the star anise essential oils extracted at varied extraction times and temperatures showed antioxidant activities (Table 2). The degree of antioxidant activity of all samples was determined qualitatively from observation of the yellow colour intensity. Based on the yellow intensity of the bands on the chromatograms, the essential oil from star anise extracted at 60 ⁰C and 70 ⁰C for 1 day, 3 days, 5 days, 7 days and 9 days apparently showed stronger antioxidant activity than those extracted at 30 ⁰C, 40 ⁰C and 50 ⁰C.
Besides, it was observed that at least two different bands with antioxidant activity with different polarity were appeared on the TLC plate after spraying with DPPH and incubated for 30 minutes. (Figure 1-5). Samples extracted at 30 ⁰C for five days, 40 ⁰C for three days, 50 ⁰C and 60 ⁰C for nine days had two active bands while the rest of the samples showed three active bands with antioxidant activity (Table 3). The Rfvalues of the active bands in all samples were in the range of 0.41 to 0.65.
 |
Figure1: Chromatogram of essential oil extracted at 30 ⁰C for 1 day, 3 days, 5 days, 7 days and 9 days (from left to right). Click here to View Figure |
 |
Figure2: Chromatogram of essential oil extracted at 40 ⁰C for 1 day, 3 days, 5 days, 7 days and 9 days (from left to right). Click here to View Figure |
 |
Figure3: Chromatogram of essential oil extracted at 50 ⁰C for 1 day, 3 days, 5 days, 7 days and 9 days (from left to right). Click here to View Figure |
 |
Figure4: Chromatogram of essential oil extracted at 60 ⁰C for 1 day, 3 days, 5 days, 7 days and 9 days (from left to right). Click here to View Figure |
 |
Figure5: Chromatogram of essential oil extracted at 70 ⁰C for 1 day, 3 days, 5 days, 7 days and 9 days (from left to right). Click here to View Figure |
Quantitative test using DPPH scavenging assay
The antioxidant activity of star anise essential oil was measured in term of radical scavenging ability, using the stable free DPPH radical. This method was adapted from the work of Brand-Williams, Cuvelier and Berset (1995) with some modifications [8]. Stock solution (1mg/ml) was twofold diluted to different concentrations (0.0625, 0.125, 0.25, 0.5 and 1 mg/ml) with methanol. An aliquot of each dilution, about 1 ml was mixed with 1 ml of 0.004 % methanolic DPPH (0.004 g of DPPH in 100 ml of methanol). The mixture was vortexed and incubated in the dark at room temperature for 30 minutes. The absorbance was measured at 517 nm against a blank (1 ml of methanol with 1 ml of methanolic DPPH without the test oil) using spectrophotometer (Model: Genesys 20 4001/4). The radical scavenging activity or antioxidant activity (%) of each concentration of oil was calculated using the following formula
Antioxidant activity (%)= [ (ODblank – ODassay) / (ODblank) ] x 100 %……………………..(Equation 1)
The test was performed in triplicate. The antioxidant activities of the essential oils were expressed as EC50, defined as the oil concentration in mg/ml required to scavenge 50 % of the DPPH free radical. BHT was used as positive control.
HPLC analysis
HPLC analysis was performed using Prominence, Shimadzu, Japan. The separation column was Hypersil C18 column (250 mm x 4.6 mm, 5 μm). Essential oil was dissolved in acetonitrile (1 mg/ml). The sample was filtered using syringe with 0.45 µm filters prior to being filled into the vial. 5 ul of sample was injected into HPLC system and the injector temperature was 30 ⁰C. A gradient elution system using mobile phase of 15 %-65 % of acetonitrile with a flow rate of 1 ml/min for 45 minutes was used to identify the compounds present in the star anise essential oil.The detection wavelength was 275 nm.GC-MS analysis of the star anise sample was also done using GCMS 5977A from Agilent Technologies.
Results and Discussion
The extraction yields of essential oils
Maximum yield of star anise essential oil (8.56 %) could be obtained by soaking star anise at 60 ⁰C for 7 days (Table 1). In contrast, extraction of essential oil at temperature of 30 ⁰C for 1 day gave minimum yield of essential oil (4.62 %). It was about 46.03 % less of extraction yield compared to the sample extracted at 60 ⁰C for 7 days.
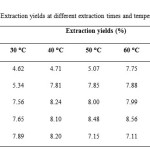 |
Table1: Extraction yields at different extraction times and temperatures. Click here to View table |
There was contradictory report from previous researchers. From the previous study done by Ngoc (2006), dried star anise fruits have an essential oil content of 8-10 % [9]. The extraction yield of star anise essential oils in this study was 4.62-8.56 %, which was lower than that of extracted in previous study done by Ngoc. Different extraction yield of essential oil content might be due to several factors such as different geographical locations of the botanical materials, varying in cultivation condition, climate as well as post-harvest factors [10].
Besides, it was observed that the yield of essential oil increased slightly with increasing extraction time and temperature until a certain point. This result was in accordance with Dent and co-workers’ (2012) study, reflected that yield increased with increasing extraction temperature and extraction time [11]. At temperature of 30 ⁰C, the extraction yield increased gradually with days. This result was in accordance with Kumar’s previous study (2010), stated that the longer the extraction time, the longer the contact time of the plant material with the solvent and hence more yields of complete oil [12]. For temperature of 40 ⁰C and 70 ⁰C, the extraction yields decreased when star anise soaked for more than 5 days. Same behavior was observed at temperature of 50 ⁰C and 60 ⁰C, the yields decreased with an increase in extraction time at 9 days. This may be due to the loss of solvent by evaporation when extraction time and temperature increased simultaneously during extraction.
For star anise soaked for 1, 3 and 7 days, the yield of essential oil increased from 30 ⁰C to 60 ⁰C. The extraction yield decreased with a further increase in temperature at 70 ⁰C due to loss of solvent and volatile oil at high temperature. The increase of extraction yield with increasing extraction time and temperature may be due to increased solubility and diffusion coefficient [11]. In this present study, the extraction temperature of 60 ⁰C and the extraction time of 7 days were the best condition to increase the extraction efficiency of star anise essential oil. It is therefore suggested that an extraction temperature of no higher than 60 ⁰C is employed.
Qualitative test using DPPH on TLC
All the star anise essential oils extracted at varied extraction times and temperatures showed antioxidant activities (Table 2). The degree of antioxidant activity of all samples was determined qualitatively from observation of the yellow colour intensity. Based on the yellow intensity of the bands on the chromatograms, the essential oil from star anise extracted at 60 ⁰C and 70 ⁰C for 1 day, 3 days, 5 days, 7 days and 9 days apparently showed stronger antioxidant activity than those extracted at 30 ⁰C, 40 ⁰C and 50 ⁰C.
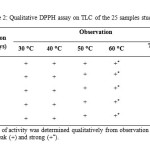 |
Table2: Qualitative DPPH assay on TLC of the 25 samples studied. Click here to View table |
Besides, it was observed that at least two different bands with antioxidant activity with different polarity were appeared on the TLC plate after spraying with DPPH and incubated for 30 minutes. (Figure 1-5). Samples extracted at 30 ⁰C for five days, 40 ⁰C for three days, 50 ⁰C and 60 ⁰C for nine days had two active bands while the rest of the samples showed three active bands with antioxidant activity (Table 3). The Rfvalues of the active bands in all samples were in the range of 0.41 to 0.65.
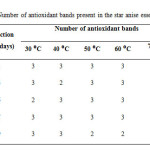 |
Table3: Number of antioxidant bands present in the star anise essential oils. Click here to View table |
Quantitative test using DPPH scavenging assay
The radical scavenging activity or antioxidant activity (%) of each concentration of oil and BHT were calculated using the formula (Equation 1) and the results were shown in Table 4. Generally, all the samples extracted at 30 ⁰C for 1, 3, 5, 7 and 9 days showed low antioxidant activity, which was below 60 %. Figure 6 showed that the antioxidant activity (%) of essential oils extracted at 30 ⁰C increased steadily with increasing concentration from 6.25 x 10-3 mg/ml to 1.00 mg/ml. The antioxidant activity of each sample was then expressed as EC50, which is the concentration of sample required to decrease the initial DPPH free radical by 50 %.
 |
Figure6: DPPH radical scavenging activity of star anise essential oils extracted at 30 ⁰C for 1, 3, 5, 7 and 9 days. Click here to View Figure |
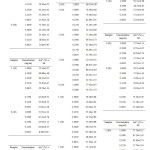 |
Table4: Antioxidant activity (%) of each concentration of star anise essential oils. Click here to View table |
It was observed that star anise essential oil extracted at 30 ⁰C for 1 day had the greatest EC50 value of 0.973 mg/ml (Table 5). This means that the essential oil extracted at 30 ⁰C for 1 day had lowest antioxidant activity as it required 0.973 mg/ml to scavenge 50 % of the free DPPH radicals. The EC50 values for samples extracted at 30 ⁰C for 3 days (0.967 mg/ml), 5 days (0.928 mg/ml), 7 days (0.893 mg/ml) and 9 days (0.622 mg/ml) were declined gradually. This indicated that the degree of antioxidant activity increased with increasing extraction time at 30 ⁰C. This might be due to longer contact time of star anise with solvent and hence increased the rate of extraction of potential active compounds with antioxidant activity [12]. The ascending order for the antioxidant activity was 1 (30 ⁰C) ˂ 3 (30 ⁰C) ˂ 5 (30 ⁰C) ˂ 7 (30 ⁰C) ˂ 9 (30 ⁰C).
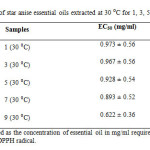 |
Table5: EC50 of star anise essential oils extracted at 30 ⁰C for 1, 3, 5, 7 and 9 days. Click here to View table |
The antioxidant activity (%) of all the star anise essential oil extracted at 40 ⁰C was above 60 % (Table 4 and Figure 7). The EC50 value for essential oil extracted at 40 ⁰C for 1 day was 0.585 mg/ml (Table 6). The EC50 value dropped to 0.391 mg/ml when the essential oil extracted at 40 ⁰C for 3 days. After that, there was an increase in EC50 values until day 9. The increase of EC50 values corresponded to a weaker antioxidant.The order of antioxidant capacity for samples extracted at 40 ⁰C was 1 (40 ⁰C) < 9 (40 ⁰C) < 7 (40 ⁰C) < 5 (40 ⁰C) < 3 (40 ⁰C).
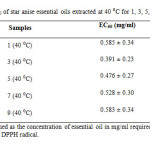 |
Table6: EC50 of star anise essential oils extracted at 40 ⁰C for 1, 3, 5, 7 and 9 days. Click here to View table |
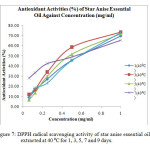 |
Figure7: DPPH radical scavenging activity of star anise essential oils extracted at 40 ⁰C for 1, 3, 5, 7 and 9 days. Click here to View Figure |
In general, the antioxidant activity (%) of star anise essential oils extracted at temperature of 50 ⁰C was higher than 70 % (Table 4 and Figure 8). According to Table 7, essential oil extracted at 50 ⁰C for 1 day showed EC50 value of 0.098 ± 0.06 mg/ml. The concentration of essential oil needed to scavenge 50 % of DPPH radical increased from day 1 to day 7. However, there was a marked decrease of concentration of essential oil to scavenge 50 % of the radicals. The antioxidant power of sample extracted at 50 ⁰C for 9 days (0.099 ± 0.06 mg/ml) was as good as the sample extracted for 1 day. In other words, essential oil extracted at 50 ⁰ C for 1 day was the strongest antioxidant, followed by those essentail oil extracted at 9 days, 3 days, 5 days and 7 days.
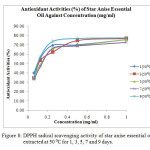 |
Figure8: DPPH radical scavenging activity of star anise essential oils extracted at 50 ⁰C for 1, 3, 5, 7 and 9 days. Click here to View Figure |
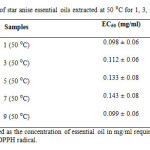 |
Table7: EC50 of star anise essential oils extracted at 50 ⁰C for 1, 3, 5, 7 and 9 days. Click here to View table |
All the essential oils extracted at 60 ⁰C showed antioxidant activity greater than 70 % (Table 4 and Figure 9). The results in Table 8 showed that essential oils extracted at 60 ⁰C for 1 day was the strongest antioxidant. This was because only 0.089 ± 0.05 mg/ml of essential oil was needed for the scavenging activity of free radicals. The antioxidant power was decreasing in the order 1(60 ⁰C) > 5(60 ⁰C) > 7(60 ⁰C) > 3 (60 ⁰C) > 9 (60 ⁰C).
 |
Figure9: DPPH radical scavenging activity of star anise essential oils extracted at 60 ⁰C for 1, 3, 5, 7 and 9 days. Click here to View Figure |
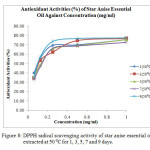 |
Table8: EC50 of star anise essential oils extracted at 60 ⁰C for 1, 3, 5, 7 and 9 days. Click here to View table |
Compared with essential oil extracted at 60 ⁰C for 1 day (EC50 value= 0.089 ± 0.05), essential oil extracted at 60 ⁰C for 9 days (EC50 value= 0.583 ± 0.34) showed a significant difference in term of concentration in mg/ml necessary to scavenge 50 % of the DPPH radicals. The antioxidant activities of star anise essential oils extracted at 60 ⁰C were undulated with increasing extraction time. This might be due to the possible synergistics effects of different compounds on the total oil antioxidant activity [13].
All the star anise essential oils extracted at 70 ⁰C for varied extraction time showed antioxidant activity (%) above 70 % (Table 4 and Figure 10). Since lower EC50 value indicated higher antioxidant activity, essential oil extracted at 70 ⁰C for 3 days exhibited stronger antioxidant activity than essential oil extracted for 1 day. The EC50 value for essential oil extracted for 3 days and 1 day were 0.100 ± 0.06 mg/ml and 0.135 ± 0.08 mg/ml respectively as shown in Table 9. Essential oils extracted more than 3 days, which were 5 days, 7 days and 9 days showed decreasing of antioxidant power as the EC50 value increased markedly from 0.100 ± 0.06 mg/ml to 0.310 ± 0.18 mg/ml. The antioxidant activity was in order 3 (70 ⁰C) > 5 (70 ⁰C) > 7 (70 ⁰C) > 1 (70 ⁰C) > 9 (70 ⁰C).
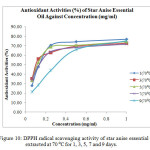 |
Figure10: DPPH radical scavenging activity of star anise essential oils extracted at 70 ⁰C for 1,3, 5, 7 and 9 days. Click here to View Figure |
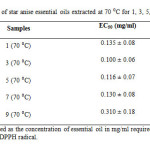 |
Table9: EC50 of star anise essential oils extracted at 70 ⁰C for 1, 3, 5, 7 and 9 days. Click here to View table |
There was an increase in antioxidant activities of star anise essential oils from 30 ⁰C to 50 ⁰C. This might due to ambient or physiological temperatures could reduce thermal degradation of volatile essential oil [14]. The antioxidant activities of star anise essential oils decreased slightly at 60 ⁰C. This was in agreement with Dent and research group (2012), stated that degradation of phenolic compounds may occured at temperature of 60 ⁰C [11]. At temperature 70 ⁰C, the antioxidant activities of star anise essential oils increased moderately. Typically, antioxidant activity decreased with increasing temperature, but it does not have universal validity [15].
In this quatitative antioxidant test using DPPH method, a well known synthetic antioxidant, namely butylated hydroxytoluene (BHT) was used as positive control. The concentration of BHT for a 50 % scavenging activity was about 0.018 mg/ml [16]. In this study, 0.016 ± 0.01 mg/ml of BHT was required in scavenging action towards DPPH free radicals (Table 10).By comparing the antioxidant activity of star anise essential oil with BHT, it was revealed that antioxidant activity of essential oil from star anise was comparable with the synthetic antioxidant BHT, particularly essential oils that were extracted at temperature 50 ⁰C and 60 ⁰C.
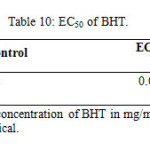 |
Table10: EC50 of BHT. Click here to View table |
HPLC and GC-MS analysis
The HPLC chromatogram of standard trans-Anethole with retention time of 35.487 minutes was shown in Figure 11. It was observed that the highest peaks of all samples have a retention time near to the retention time of standard trans-Anethole, which was around ±35 minutes. Similarities of the highest peak and retention time between samples and standard trans-Anethole in the HPLC chromatogram confirmed that trans-Anethole was present in star anise essential oil. In order to identify the concentration (%) of trans-Anethole present in all the essential oils, a standard curve of trans-Anethole was plotted (Figure 12). The retention times, peak areas, area percentage and concentration (%) of trans-Anethole for all samples were shown in Table 11.
 |
Figure11: HPLC chromatogram for standard trans-Anethole. Click here to View Figure |
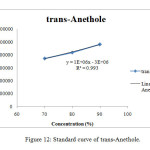 |
Figure12: Standard curve of trans-Anethole. Click here to View Figure |
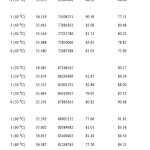 |
Table11: Retention time, peak area, area percentage and concentration (%) of trans-Anethole. Click here to View table |
All the star anise essential oils had high area percentage of trans-Anethole, which was ranged from 77.29 % to 91.87 %. High percentage area indicated trans-Anethole was the most abundant compounds in all the star anise essential oils. Similar results were reported previously by Chempakam andBalaji (2008), stated that the main component of star anise essential oil is trans-Aanethole, which accounts for 80-90 % [4]. From GC-MS analysis, Estragole was found to have the most significant peak with peak area 100% at retention time 13.840 minutes as shown in Figure 13 and 14 and Table 12.Estragole was found at the tenth peak.Anethole was also found at two peaks which were peak 11 and 12 as shown in Figure 15 and Figure 16. Thus, the data validates the presence of Anethole in the star anise sample.
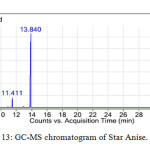 |
Figure13: GC-MS chromatogram of Star Anise. Click here to View Figure |
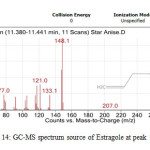 |
Figure14: GC-MS spectrum source of Estragole at peak 10. Click here to View Figure |
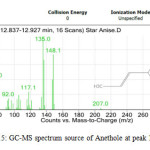 |
Figure15: GC-MS spectrum source of Anethole at peak 11. Click here to View Figure |
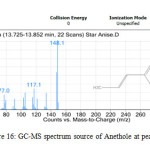 |
Figure16: GC-MS spectrum source of Anethole at peak 12. Click here to View Figure |
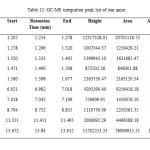 |
Table12: GC-MS integration peak list of star anise. Click here to View table |
In addition, it was observed that the concentration (%) of trans-Anethole in the star anise essential oils varied considerably as function of temperature and extraction time. Generally, star anise essential oils extracted at 40 ⁰C, 50 ⁰C, 60 ⁰C and 70 ⁰C contained relatively high concentration (%) of trans-Anethole compared to essential oils extracted at 30 ⁰C. As temperature increases, higher mass fraction of trans-Anethole was extracted. It was in agreement with Chen and co-workers’ finding (2007), which concluded that higher temperature will reduce the solvent viscosity and facilitate the diffusion of molecules, resulting to an increase of extraction efficiency [17].
Besides, it was found that the concentration (%) of trans-Anethole corresponded to the antioxidant activities of star anise essential oils. This statement was in agreement with Padmashree and research group (2007), reported that the antioxidant activity is due to high percentage of trans-Anethole, which is more than 80 % [18]. In the present study, star anise essential oil extracted at 60 ⁰C for 1 day has the highest concentration of trans-Anethole (91.61 %) and antioxidant activity (EC50=0.089 mg/ml) while star anise essential oil extracted at 30 ⁰C for 1 day has the lowest concentration of trans-Anethole (45.03 %) and antioxidant activity (EC50=0.973 mg/ml). Therefore, it might be possible to infer that trans-Anethole found in the samples contributed to the antioxidant activities of the essential oils.
Conclusion
Essential oil from star anise was successfully extracted using solvent extraction method. Different extraction times (1 day, 3 days, 5 days, 7 days and 9 days) and temperatures (30 ⁰C, 40 ⁰C, 50 ⁰C, 60 ⁰C and 70 ⁰C)did affect the yield of essential oil. The best condition to obtain the highest yield (8.56 %) of essential oil was at temperature of 60 ⁰C with an extraction time of 7 days. A rapid screening using DPPH assay on TLC showed thatat least two different yellow spots with antioxidant activity with different polarity were appeared on all TLC plates at solvent system 95:5, v/v toluene/ ethyl acetate. In addition, the highest antioxidant activity of star anise essential oil was observed when the sample was extracted at 60 ⁰C for 1 day (EC50 value = 0.089±0.05 mg/ml).From the HPLC analysis results, samples with higher trans-Anethole concentration have higher antioxidant activities. Star anise essential oil extracted at 60 ⁰C for 1 day showed the highest concentration (%) of trans-Anethole (91.61 %) while essential oil extracted at 30 ⁰C for 1 day showed the lowest concentration (%) of trans-Anethole (45.03 %). The antioxidant activity of the high level of trans-Anethole-containing star anise essential oil was comparable with the synthetic antioxidant BHT.
Acknowledgement
The authors express their sincere appreciation to Faculty of Agro Based Industry, Universiti Malaysia Kelantan, Jeli Campus for the financial support which made this study possible.
References
- Ito, N.; Hirose, M.; Fukushima, S.; Tsuda, H.; Tatematsu, M. Food and Chem. Tox.,1986,24(10-11), 1071.
- Pokorny, J. Trends in Food Science Technology,1991, 9, 223-227.
- Nanditha, B.; Prabhasankar, P. Critical Reviews in Food Science and Nutrition,2009, 49, 1-27.
- Chempakam, B.; Balaji, S. Star Anise. United Kingdom: CAB International, 2008, 319-330.
- Ong, H.C.Utusan Publication & Distributors Sdn. Bhd., 2008, 94.
- Thuat, B.Q.; Ngoc, B.T.B. VNU Journal of Science and Technology,2010,26, 110-113.
- Saleh, M.A.; Clark, S.; Woodard, B.; Deolu-Sobogun, S.A. Ethnicity and Disease,2010, 20, 78-82.
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Journal of Food Science Technology,1995, 28, 25-30.
- Ngoc, B.T.B. Hanoi University of Polytechnique, Vietnam ,2006.
- Abu-Darwish, M.Z.; Abu, D.B.; Mufeed, M.; Abdel, R.A.; Al-Tawaha, S.; Al-dalain. Journal of Food, Agriculture and Environment,2009,7(3&4), 920-924.
- Dent, M.; Dragović-Uzelac, V.; Penić, M.; Brnčić, M.; Bosiljkov, T.; Levaj, B. Food Technology & Biotechnology,2012,51(1), 84.
- Kumar, K.S. National Institute of Technology Rourkela, India, 2010.
- Abdalla, A.E.; Roozen, J.P.Food Chemistry,1999,64, 323-329.
- Laguerre, M.; Lecomte, J.; Villeneuve, P.Progress in Lipid Research,2007,46, 244-282.
- Pokorny, J.Journal of Food Science,1986, 4, 299-307.
- Kulisic, T.; Radonic, A.; Katalinic, V.; Milos, M. Food Chemistry,2003,85, 633-640.
- Chen, Y.; Xie, M.Y.; Gong, X.F.Journal of Food Engineering,2007, 81, 162-170.
- Padamshree, A.; Roopa, N.; Semval, A.D.; Sharma, G.K.; Agathian, V.; Bawa, A.S. Food Chemistry, 2007, 104(1), 59-66.

This work is licensed under a Creative Commons Attribution 4.0 International License.









