Bioethanol Production from Sugarcane Bagasse using Fermentation Process
Y. C. Wong1* and V. Sanggari1
1Faculty of Agro Based Industry, University Malaysia Kelantan, Jeli Campus, Locked Bag 100, 17600 Jeli, Kelantan, Malaysia.
DOI : http://dx.doi.org/10.13005/ojc/300214
Article Received on :
Article Accepted on :
Article Published : 05 Jun 2014
The aim of this study is to produce bioethanol from sugarcane bagasse using fermentation process and to determine the effect of pH and temperature on bioethanol yield. Enzymes such as alpha- amylase and glucoamylase were used to breakdown the cellulose in sugarcane bagasse. Saccharomyces cerevisiea, (yeast) also was used in the experiment for fermentation. Five samples were prepared at different pH was varied to determine the effects of pH on ethanol yield at 370 C and another five samples were prepared to determine the effect of temperature on ethanol yield, the pH was kept constant at 4.5. The ethanol concentrations were determined by running the samples in High Performance Liquid Chromatography (HPLC). The results showed that at highest ethanol concentration was obtained pH 4.5 and temperature 350C. This indicated that pH 4.5 and 350C was the optimum parameter for the yeast to produce ethanol.
KEYWORDS:Bioethanol Production; Sugarcane Bagasse;fermentation process
Download this article as:| Copy the following to cite this article: Wong Y. C, Sanggari V. Bioethanol Production from Sugarcane Bagasse using Fermentation Process. Orient J Chem 2014;30(2). |
| Copy the following to cite this URL: Wong Y. C, Sanggari V. Bioethanol Production from Sugarcane Bagasse using Fermentation Process. Orient J Chem 2014;30(2). Available from: http://www.orientjchem.org/?p=3650 |
Introduction
Bioethanol is simply ethanol is a renewable energy source which is made by fermenting the sugar and starch components of plant. It is produced from the agricultural product such as corn, sugarcane, potatoes, rice, beetroot and recently using grapes, banana, dates and other wastes.This is due to the decreasing amount of fossil fuels, alternative energy sources need to be renewable, sustainable, efficient, cost effective, convenient and safe [2].
The demand for oil is expected to increase to 57% from 2002 to 2030. The average price of gasoline in 2005 was $2.56 per gallon, which was $0.67 higher than the average price of gasoline in the previous year. Yet in June 2008, the average price of gasoline in the US reached $4.10 per gallon [6]. Rise in energy demand in worldwide and the progressive demising of oil reserves motivate the search for alternative energy resources, especially for those derived from renewable materials such as biomass [18].
As a result, they come up ethanol production as substitutes to fossil fuel. The lower cost to produce bioethanol is come from biomass waste because the raw materials are available in abundance.
The main objective of this thesis is to successfully produce bioethanol from sugarcane bagasse. Sugarcane bagasse is an agricultural waste which used to produce bioethanol using fermentation process. Next is to determine the yield of bioethanol produced from sugarcane bagasse. The bioethanol will be determined using HPLC. The highest peck shows the ethanol content. Last objective is to study the effects of temperature and pH of fermentation process in bioethanol production.
Material And Method
Preparation Of Sugarcane Bagasse
The sugarcane bagasse was obtained from area around Tanah Merah and Bazzar Ramadan in Ayer Lanas, Kelantan. The sugarcane bagasse was thoroughly washed with tap water and cut into smaller pieces. Then sugarcane bagasse was dried in oven at 600C for 3 days. It was treated at 600C because if the temperature was higher, it will affect the enzymes in the sugarcane bagasse. Once dried, the sugarcane bagasse was grinded using the grinding machine. The sugarcane bagasse sample was sealed in the seal bag or poly bag and stored in room conditions.
Buffer preparation
Buffer was used to dilute the enzyme alpha-amylase and glucoamylase. Enzyme reaction will be more effective if dilute with buffer compared to distilled water. There were two types of buffers prepared which were phosphate buffer for α-enzymes and acetic acid with sodium acetate buffer for glocoamylase. The prepared buffer was covered with aluminum foil and kept at room temperature for further use.
Liquefaction of sugarcane
10g of sugarcane was weighed. The weighed sample was placed into conical flask and 200ml of distilled water was added to the sample. 0.5 ml of NaOH was prepared to be added at this step to adjust the pH of the slurry to 4.5. Next 0.2 microliters of enzyme alpha- amylase was added to the mixture. The alpha-amylase was diluted with phosphate buffer before added to the slurry. The mixture was heated until 500C. Alpha- amylases will breakdown the cellulose into smaller size called dextrin.
Saccharifaction of sugarcane bagasse
The mixture was cooled down to 400C. Then, 0.2 microliters of secondary enzyme, glucoamylase was added to the mixture. Glucoamylase was diluted with acetic acid with sodium acetate buffer before added to the slurry.The mixture was maintained at 500C as the glucoamylase hydrolyzed the dextrin to fermentable glucose. The mixture was cooled down to 320C and 10 ml of Saccharomyces cerevisiea (baker yeast) was added to the sample before transferred to conical flask.
Fermentation of sugarcane
In fermentation process, Saccharomyces cerevisiea (baker yeast) was used to ferment the simple sugar to ethanol and carbon dioxide. To determine the effects of pH on ethanol yield, the temperature was kept constant at 370C while the pH was varied from 3, 3.5, 4, 4.5 and 5. To determine the effect of temperature on ethanol yield, the pH was kept constant at 4.5. The fermentation process continued for 48 hours.
Distillation of ethanol
After 48 hours, the sample was filtered using Whatman Filter Paper to separate the ethanol from the residue. The bioethanol was distilled using rotary evaporator. The sample was heated at 800C to get the bioethanol.
Determine bioethanol yield
Bioethanol produced was analyzed by high performance liquid chromatography(HPLC). 20 µL of the sample was injected into HPLC system to determine the bioethanol yield. Fatty acid composition in virgin coconut oil was analyzed using HPLC method. The HPLC analysis parameters were determined using the following conditions: column, C18 RP (53 x 7mm); injector temperature was 30oC, 20 µL of the sample was injected into the HPLC system. The mobile phase was phosphoric acid and the flow rate was 1.5mL/min; and detection was set at a wavelength of 210 nm.
Statistical Analyses
The data obtained was subjected to One Way ANOVA using statistical package for social science (SPSS) computer program to find out the significance at p < 0.01.
Results and Discussion
Calibration curve
In this study calibration cure was drawn to determine total ethanol concentration in water from sugarcane samples. A linear graph of standardization of ethanol was drawn using 95% ethanol as a standard. The standard was prepared at different concentrations to such as 25%, 50%, 75% and 100%. The amount of ethanol added to each vial showed in the Table 1. The calibration equation of ethanol standard was determined to be y = 367.94x- 2853 (R2 = 0.9515) where y is the peak area of ethanol and x is the concentration of ethanol. The Ethanol standard curve is shown in the Figure 1.
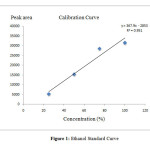 |
Figure 1: Ethanol Standard CurveClick here to View Figure |
Table 1: Standard calculation
| Concentration (%) | Volume of ethanol Standard (ml) | Volume of mobile phase (ml) | Peak Area |
| 25 | 0.5 | 1.5 | 5229 |
| 50 | 1.0 | 1.0 | 15330 |
| 75 | 1.5 | 0.5 | 28520 |
| 100 | 2.0 | 0 | 31494 |
Effects of pH of fermentation process on ethanol concentration in water
The study was carried out to determine the significant influences of pH on fermentation due to its effect on yeast growth and fermentation rate. The sample was fermented at different pH values from 3, 3.5, 4, 4.5 and 5 while the temperature was kept constant at 370C to obtain maximum yield of bioethanol. The total bioethanol content in each sample was determined and recorded in the Table 2 and Figure 2.
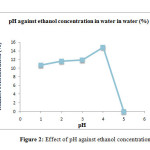 |
Figure 2: Effect of pH against ethanol concentration in water Click here to View Figure |
Table 2: The Effects of pH on Ethanol Concentration (%) in water
|
pH |
Ethanol concentration in water (%) |
|
3.0 |
10.7 |
|
3.5 |
11.6 |
|
4.0 |
11.9 |
|
4.5 |
14.8 |
|
5.0 |
0.0 |
Based on the results obtained, pH 4.5 showed the highest ethanol content in water which is 14.8 %, followed by pH 4.0 which is 11.9 %, then pH 3.5 at 11.6 % and pH 3.0 at 10.7 %. The lowest ethanol concentration in water with water was achieved at pH 5.0. Figure 4.1 show that the ethanol concentration in water gradually increases along with the increases in pH and reaches a maximum percentage of ethanol production when pH is equals to 4.5 and later it start to declining.
The maximum ethanol concentration in water at pH 4.5 reflects enzyme function in an environment [1] while the lower ethanol concentration in water at pH reflects lesser yeast activity. The maximum ethanol productivity was observed at pH of 4.2 to 4.5 [3][12]. Furthermore, the increase in ethanol concentration in water is more efficient with the increase in pH from 4.0-4.5 and also found that the optimum pH range for S.cerevisiae to be pH 4.5 [22].
In general, yeast is an acidophilic organism and as such, grows better under acidic condition. The optimum pH range for yeast growth can vary from pH 4.0 to 6.0, depending on the temperature, the presence of oxygen and strain of yeast. Optimum pH values are required for the activity of plasma membrane bound proteins, including enzymes and transport proteins [10]. During growth, it is important for the yeast to maintain a constant intercellular pH.
There are many enzymes functioning during within yeast cell during growth and its metabolism. Each enzyme works best at its optimal pH, which is acidic because of the acidophilic nature of the yeast itself. When the extracellular enzymes pH changes from the optimal level, the yeast cell required using energy to either pump in or pump out the hydrogen ions in order to maintain the optimum intercellular pH [11].
If the extracellular pH changes too much from the optimum pH range, it may too difficult for the cell to maintain constant intracellular pH and the enzyme may not function normally. Furthermore, if the enzymes are deactivated, the yeast cell will not be able to grow and make ethanol efficiently [10]. This is the most likely explanation for the observed reduction in ethanol production when the initial medium pH was at 3.0. There were also low carbon dioxide productions at pH3 because the low pH encourages the production of acid instead of alcohol [5].
However this study shows lowest ethanol concentration in water at pH 5. This may be due to the disability of the yeast strain to tolerate at pH 5. Different yeast strain has different pH range to activate and produce ethanol. There are other possibilities; the yeast that was used to conduct the experiment may be old. Old yeast will not carry out fermentation process efficiently compared to new yeast. According to Misonoet al. (1990) [9], there is increased rate of ethanol production at pH 5. This statement is not applicable for this study since at pH 5 there is no ethanol production.
Effects of temperature of fermentation process on ethanol concentration in water
Temperature is one of the major factors that determine the ethanol production. Table 3 and Figure 3 showed the ethanol concentration in water (%) that obtained at different temperature. Based on the result obtain, no ethanol concentration in water was observed at 25 and 300C.
Table 3: Effects of temperature on ethanol concentration in water (%)
|
Temperature (0C) |
Ethanol concentration in water with water (%) |
|
25 |
0.0 |
|
30 |
0.0 |
|
35 |
13.7 |
|
40] |
12.3 |
|
45 |
11.1 |
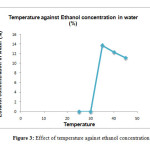 |
Figure 3: Effect of temperature against ethanol concentration in water Click here to View Figure |
However, as the temperature increases beyond 300C it showed increase in production of ethanol. At 350C ethanol concentration in water were maximum and turned out to be 13.7% followed by 400C where 12.3% ethanol was obtained. Fermentation process required a suitable temperature for the yeast to react [16]. Temperature that is too high kills yeast, and low temperature slows down yeast activity. Thus, to keep a specific range of temperature were required.
However the ethanol concentration in water was decreased at 450C. This indicates that 350C were the optimum temperature for ethanol production. This finding is in agreeable with last studies about temperature on ethanol concentration in water [14][15][17]. This studies result also denied the study of Yah et al., (2010) [23], who found optimum temperature of ethanol production to be 250C.
From the result we can conclude that higher the temperature, lower the ethanol concentration. The rate of enzyme catalyzed reaction increases with temperature up to a certain temperature and then the enzymes begins to denature. Higher temperature inhibits the growth of the cells and fermentation significantly decreases. In this study, ethanol concentration in water declined considerably at 400C, which showed the inhibition effects on the cell growth at higher temperature.
This statement supported by study from [8], [13]. Based on the high temperature might denature the ribosome and enzymes. Furthermore, higher temperature would alter the structure of the membrane and decreases its functionality [7]. Above the optimum temperature, the enzyme reaction drops precipitously as the enzyme denatures [19].
Enzymes are sensitive to temperature changes. At temperature above 400C the rate of respiration slows down and drops. This was because all the enzymes are made up of the protein chains of amino acid. It exists in the form of a helix structure with hydrogen bonds holding them together. When heat was applied to the enzyme, energy was given off. The active enzyme cell deforms
and the hydrogen bonds break, denature the yeast enzyme. This process called as denaturizing. The optimum temperature in which yeast enzyme work best is around 350C, below this temperature the rate of reaction was slow and above 450C the yeast enzyme would denature.
At low temperature the cells showed no ethanol concentration. This may be due to enzymes low tolerance to produce ethanol at lower temperature [4][21]. Furthermore, at low temperature the enzyme deactivated and reaction slow down or stop altogether [20]. At lower temperature, the molecules move slower than at higher temperature. These explain that the enzyme may not have enough energy to cause chemical reaction. Overall we can conclude that temperature 350C was the optimum temperature for ethanol production.
Statistical analysis for effects of pH and temperature on fermentation process
One way analysis of variance (ANOVA) was conducted to evaluate the relationship between effects of pH on ethanol concentration. The Independent values were pH while the dependent values were ethanol concentration. AVOVA used determine whether there was a significance difference in ethanol concentration. Table 4 shows the ANOVA table for the effect of pH on ethanol concentration. The one way ANOVA results indicate that there was a significant difference in pH at (P<0.01) level for three conditions [F (4, 10) = 84.051, p = 0.000].
Table 4: Summary for one way ANOVA table for effect of pH and ethanol concentration
|
ANOVA |
|||||
| Ethanol concentration | |||||
| Sum of Squares | df | Mean Square | F | Sig. | |
| Between Groups | 447.149 | 4 | 111.787 | 84.051 | .000 |
| Within Groups | 13.300 | 10 | 1.330 | ||
| Total | 460.449 | 14 | |||
One way analysis of variance (ANOVA) was conducted to evaluate the relationship between effects of temperature on ethanol concentration. The independent values were temperature while the dependent values were ethanol concentration. AVOVA used determine whether there was a significance difference in ethanol concentration. Table 5 shows the one way ANOVA table for the effect of temperature on ethanol concentration. The one way ANOVA results indicate that there was a significant difference in temperature at (P<0.01) level for three conditions [F (4, 10) = 14.384, p = 0.000].
Conclusion
This study shows that pH 4.5 showed the highest ethanol content in water which is 14.8 %, followed by pH 4.0 which is 11.9 %, then pH 3.5 at 11.6 % and pH 3.0 at 10.7 %. The lowest ethanol concentration in water with water was achieved at pH 5.0. The study also shows that at 350C ethanol concentration in water were maximum and turned out to be 13.7% followed by 400C where 12.3% ethanol. However there is no ethanol production at temperature 25 and 300C.Statistical analysis one way ANOVA results indicate that there was a significant difference in pH at (P<0.01) level for three conditions [F (4, 10) = 84.051, p = 0.000]. The one way ANOVA results for temperature indicate that there was a significant difference in temperature at (P<0.01) level for three conditions [F (4, 10) = 14.384, p = 0.000].Further study should conduct on more parameter that effect the fermentation process on ethanol production. There are other parameter such as amount of subtract, time and glucose concentration which affects ethanol production during fermentation. This will gives the overall view how ethanol production affected. In conclusion, pH 4.5 and 350C is the optimum condition for ethanol production.
Acknowledgement
The authors express their sincere thanks to University Malaysia Kelantan (UMK) for the financial support and providing the necessary facilities for the successful completion of this research work.
Reference
- Berg, J. (2007). Biochemistry, sixth ed. Freemantle and Company, London.
- Chum, H. L.; Overend, R. P. (2001). Biomass and Renewable Fuels. Fuel Processing Technology. Vol. 71; pg. 187-195;
- De Vasconcelos,,J. N., Lopes, C. E., & de França, F. P. (1998). Yeast immobilization on cane stalks for fermentation. International Sugar Journal,100, pg. 73-75.
- Gao, C., Fleet, G.H., (1988). The effects of temperature and pH on the ethanol tolerance of the wine yeasts: Saccharomyces cerevisiae, Candida stellata and Kloeckeraapiculata. Journal of Applied Bacteriology 65, pg. 405– 410.
- Jennings, D.H. (1995). The physiology of Fungal Nutrition. Cambridge University Press,Cambridge, pg. 392-394.
- Kumar, P., Barrett, D. M., Delwiche, M. J., &Stroeve, P. (2009). Methods for pre-treatment of lignocellulosic biomass for efficient hydrolysis and biofuel production.Industrial and Engineering Chemistry Research, 48, pg. 3713–3729.
- Lucero, P., Pen˜alver, E., Moreno, E., Lagunas, R., (2000). Internal rehalose protects endocytosis from inhibition by ethanol in Saccharomyces cerevisiae. Applied and Environmental Microbiology, 66, pg.4456– 4461.
- McMeckin, T. A., Olley J, Ratkwsky, D. A., Ross, T. (2002). Predictive microbiology: towards the interface and beyond. International Journal Food Microbiology,73, pg. 395-407
- Misono, H.M., Yamaguchi Y. (1990). The alcoholic fermentation of molasses.Journal of Fermentation Technology 8, pg. 210-218.
- Narendranath, N. V., and Power, R. (2005). Relationship between pH and medium dissolved solids in terms of growth and metabolism of Lactobacilli and Saccharomyces cerevisiaeduring ethanol production.Applied andEnvironmental Microbiology.71, pg. 2239-2243.
- Narendranath, N. V., Thomas, K. C., and Ingledew, W. M. (2001). Acetic acid and lactic acid inhibition of growth of Saccharomyces cerevisiaeby different mechanism.Journal of the American Society of Brewing Chemists. 59, pg. 187-194.
- Nigam, J. N. (1999). Continuous ethanol production from pineapple cannery waste.Journal.of Biotechnology, 72, 197-202.
- Phisalaphong, M., Srirattana, N., Tanthapanichakoon, W. (2006). Mathematical modeling to investigate temperature effect on kinetic parameters of ethanol fermentation.Biochemistry Engineering Journal;28, pg.36-43.
- Pramanik, K. (2003). Parametric Studies on Batch Alcohol Fermentation Using Saccharomyces Yeast Extracted from Toddy. J. Chin. Institution Chemistry Engineers. 34: pg. 487-492
- Redzepovic, S., Orlic, S., Sikora, S., Majdak, A., Pretorius, I. S. (2002). Identification and characterization of Saccharomyces cerevisiaeand Saccharomyces paradoxusstrains isolated from Croatian vineyards, Whileyinterscience journals, pg. 350
- Rivera, M., Cardona, C.A. (2006). Analysis of simultaneous reaction extraction processes at productive level. Process generalities, simultaneous phase and chemical equilibria.Ingenierı´a y Competitividad 6, pg. 17–25.
- Roehr, M. (2001). The Biotechnology of Ethanol: Classical and Future Applications. Chichester: Wiley-VCH. Pg. 232.
- Saxena, R.C., Adhikari, D.K. and Goyal, H.B. (2009). Biomass-based energy fuel through biochemical routes: a review. Renewable and Sustainable Energy Reviews, 13, pg. 167–178.
- Southerland, W. M. (1990). Foundations of Medicine: Biochemistry, Churchill Livingstone. inc., New York
- Togarepi E, MapiyeC.Muchanyereyi N. Dzomba P, (2012). “Optimization of Fermentation Parameters for Ethanol Production from Ziziphusmauritiana Fruit Pulp Using Saccharomyces cerevisiae (NA33)”.International Journal of Biochemistry Research & Review, 2(2): pg. 60-69.
- Torija, M.J., Roze`s, N., Poblet, M., Guillamo´n, J.M., Mas, A., (2003). Yeast population dynamics in spontaneous fermentations: comparison between two different wine areas during three consecutive years. Antonie van Leeuwenhoek 79, pg. 345– 352.
- Yadav, B.S., Sheoran A., Rani U. and Singh D. (1997). “High ethanol productivity in an immobilized cell reactor”, Indian Journal Microbiology, 37 pg. 65-67.
- Yah, C.S., S.E. Iyuke, E.I. Unuabonah, O. Pillay, C. Vishanta and S.M. Tessa, (2010). Temperature optimization for bioethanol production from corn cobs using mixed yeast strains. Journal Biology.Science., 10, pg. 103-108.

This work is licensed under a Creative Commons Attribution 4.0 International License.









