Synthesis of Coumarin Derivatives Using Glutamic Acid Under Solvent-Free Conditions
Setareh Sabetpoor*, Farhad Hatamjafari
Department of Chemistry, Faculty of Science, Islamic Azad University-Tonekabon Branch, Tonekabon, Iran
DOI : http://dx.doi.org/10.13005/ojc/300265
Article Received on :
Article Accepted on :
Article Published : 12 Apr 2014
An efficient,simple and one-pot protocol for synthesis of coumarin derivatives on the basis condensation reaction of phenols with ethyl acetoacetate employing glutamic acid as a novel catalyst is described.
KEYWORDS:One-pot; Glutamic acid; Coumarins; Solvent-Free Conditions
Download this article as:| Copy the following to cite this article: Sabetpoor S, Hatamjafari F. Synthesis of Coumarin Derivatives Using Glutamic Acid Under Solvent-Free Conditions. Orient J Chem 2014;30(2). |
| Copy the following to cite this URL: Sabetpoor S, Hatamjafari F. Synthesis of Coumarin Derivatives Using Glutamic Acid Under Solvent-Free Conditions. Orient J Chem 2014;30(2). Available from: http://www.orientjchem.org/?p=2934 |
INTRODUCTION
Multicomponent reactions are efficient methods in thesynthesis of heterocycles. In Multicomponent reactions synthesis to produce massive amounts of waste,according to the isolation of the complex, toxic and hazardous solvent at each step andwill discovered, economically and environmentallyfriendly. Multicomponent reactions advantages, synthesized of heavy compound molecules by the reaction of small molecule1.
Coumarins derivatives are heterocyclic units in the field of natural and synthetic organic chemistry due to their wide range of biological and therapeutic properties such asanti-inflammatory, anti-tumor, antioxidant,anti-viral and anti-bacterial activities.Recently, coumarin appropriate analogue functionalized gland known as antibiotics agents, and receptor antagonists have emerged. In addition, several flavonoid containing the coumarin core unit have been isolated from natural sources also show interesting biological properties. However, Thesereactions often require harsh conditions and was happened long reaction time and low efficiency. Kinds of phenols used to replace for synthesiscoumarins2-9. Although numerous methods are capable of affecting these synthesis has been previously reported. Glutamic acidhas been used previously as a catalyst for synthesis of organic compound10,Previously, we have synthesized a number of heterocyclic compounds11-23. Herein we report glutamic acida new catalyst for the synthesis of Coumarins at one pot reaction, environmentally friendly easy separation with high yields (Scheme 1).
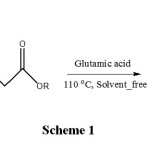 |
Scheme 1 Click here to View Scheme |
All chemicals were obtained from Merck or Flukawithout further purification. Silica gel SILG/UV 254 plates were used for TLC. IR spectra were measured on a Shimadzu IR-470 Spectrophotometer. 1H NMR spectra were determined on Bruker400 DRX AVANCE instrument at 400400 MHz, respectively.
Typical procedure adopted for the synthesis of 7-hydroxy-4-methyl-2H-chromen-2-one (S1)
A mixture of resorsinol (1 mmol), ethy lacetoacetate (1 mmol), and glutamic acid (20 mol%) was stirred at 110°C for a 15 min. The progress of the reaction was monitored by using TLC. After completion of the reaction, the solid catalyst (glutamic acid) was washed with water, and finally purified by recrystallization in ethanol/water.
Data of compounds (S1-S5)
7-hydroxy-4-methyl-2H-chromen-2-one (S1)
White powder, Yield: (93%), mp: 187-1890C
IR (νmax/cm-1)(KBr): 3440(OH), 1683(C=O), 1604(C=C)cm-1
1H NMR (400MHz; DMSO-d6) δ = 2.34 (3H, s, CH3), 6.10 (1H, s, CH), 6.68(1H, d, J=2.0 Hz, Hc), 6.79(1H, d, J=2.0 Hz, Hb), 7.76(1H, d, J=8.8 Hz, Ha),8.85(1H, brs, OH).
7,8-dihydroxy-4-methyl-2H-chromen-2-one (S2)
White powder, Yield: (90%), mp: 241-243 0C
IR (νmax/cm-1)(KBr): 3444(OH), 1677(C=O), 1600(C=C)cm-1
1H NMR(DMSO) δ = 2.34(3H, s, CH3), 6.11(1H, s, CH), 6.83-7.09(1H, m, Harom), 6.82(1H, d, J=8.0 Hz, Ha), 7.09(1H, d, J=8.0 Hz, Hb), 9.30(1H, brs, OH), 10.0(1H, brs, OH).
5,7-dihydroxy-4-methyl-2H-chromen-2-one (S3)
White powder, yield (87%), mp: 281-2820C
IR (νmax/cm-1)(KBr): 3498, 3440(2OH), 1674(C=O), 1602(C=C)
1H NMR(DMSO) δ=2.47(3H ,s, CH3), 5.82(1H, s, CH), 6.16(1H, d, J=2.4 Hz, Ha), 6.26(1H, d, J=2.4 Hz, Hb), 10.37(2H, brs, 2OH).
7-hydroxy-4,8-dimethyl-2H-chromen-2-one (S4)
White powder, yield (89%), mp: 261-263 0C IR (νmax/cm-1)(KBr): 3494(OH), 1670(C=O), 1606(C=C) 1HNMR(DMSO): δ =2.27, 2.54(6H, s, 2CH3), 6.03(1H, s, CH), 6.56(1H, d, J=0.8 Hz, Ha), 6.60(1H, d, J=0.8 Hz, Hb).
4-methyl-2H-chromen-2-one (S5)
White powder, Yield: (83%), mp: 80-820C
IR (νmax/cm-1)(KBr): 1683(C=O), 1604(C=C)cm-1
1H NMR (400MHz; DMSO-d6) δ = 2.34 (3H, s, CH3), 6.10 (1H, s, CH), 6.68(1H, d, J=2.0 Hz, Hc), 6.79(1H, d, J=2.0 Hz, Hb), 7.76(1H, d, J=8.8 Hz, Ha)
RESULTS AND DISCUSSION
Herein, we report glutamic acid ascatalyst which could provide high yield, an efficient, environmentally friendly, easy separation and simple route for the synthesis of coumarins.
ACKNOWLEDGEMENTS
We gratefully acknowledge the financial support from the Research Council of TonekabonBranch Islamic Azad University.
References
- Balme G., Bossharth E andMonteiro N.,Eur. J. Org.Chem., 21: 4101 (2003).
- KostovaI., Cur. Med. Chem., 5: 29 (2005).
- Al-AmieryA. A., KadhumA andMohamadA., Molecules, 17: 5713 (2012).
- ShikishimaY., TakaishiY., Honda G andet al., Chem. Pharm. Bull.,49: 877 ( 2001).
- ManolovI., Maichle-MoessmerC andDanchevN., Eur. J. Med. Chem., 41: 882 (2006).
- KontogiorgisC. A andHadjipavlou-LitinaD. J.,J. Med. Chem., 48: 6400 (2005).
- FylaktakidouK. C., Hadjipavlou-LitinaD. J., LitinasK. E andNicolaidesD. N., Cur. Pharm. Design., 10: 3813 (2004).
- BabaM., JinY., Mizuno A and et al., Bio. Pharm. Bull., 25: 244 (2002).
- Thornes D., DalyL., Lynch G and et al., Eur. J. Surgl. Oncol.,15: 431 (1989).
- Hatamjafari F and Abbasi E., Orient. J. Chem., 29:731 (2013).
- Azizian J., Hatamjafari F., Karimi A. R. and Shaabanzadeh M., Synthesis, 5: 765 (2006).
- Azizian J., Shaabanzadeh M., Hatamjafari F. and Mohammadizadeh M.R., Arkivoc, (xi): 47 (2006).
- Hatamjafari F., Synthetic Communications, 36: 3563 (2006).
- Azizian J., Hatamjafari F. and Karimi A. R., Journal of Heterocyclic Chemistry,43:1349 (2006).
- Hatamjafari F and Montazeri N., Turkish Journal of Chemistry,33: 797 (2009).
- Bidram A., Hatamjafari F and Doryeh A., Orient. J. Chem., 29: 123 (2013).
- Hatamjafari F., Orient. J. Chem., 28: 141 (2012).
- Hatamjafari F., Orient. J. Chem., 29: 93(2013).
- Hatamjafari F and Alijanichakoli F., Orient. J.Chem.,29: 145(2013).
- Hatamjafari F and Hosseinian A., Orient. J.Chem.,29: 109(2013).
- Hatamjafari F and Keyhani A., Orient. J.Chem.,29: 783 (2013).
- Hatamjafari F and Khojastehkouhi H., Orient. J. Chem., 30(1) (2014).
- Hatamjafari F and GermaniNezhad F., Orient. J. Chem., 30(1) (2014).

This work is licensed under a Creative Commons Attribution 4.0 International License.









