Biosorption of Divalent ion onto Treated Prosopis juliflora Bark from Aqueous Solutions - Isothermal and Statistical Analysis
N. Muthulakshmi Andal*, S. Charulatha and N. S Gayathri
1Department of Chemistry, PSGR Krishnammal College for Women, Coimbatore-4, India
Corresponding Author Email: andaldr@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/320238
Article Received on :
Article Accepted on :
Article Published : 13 Apr 2016
The present work emphasizes the utilization of Prosopis juliflora bark, an agro waste material for the adsorption of Cu(II). The raw Prosopis juliflora bark (PJB) is treated using 0.1N hydrochloric acid to enhance the sorption efficiency. The characterization studies of TPJB using Scanning Electron Microscopy (SEM), Energy Dispersive X-ray Analysis (EDAX), Brunauer-Emmet-Teller(BET) and Barrett-Joyner-Halenda (BJH) analyses carried out. The batch mode experimental set up is verified to assess the sorption capacity of the chosen material for the operating factors viz., particle sizes/ doses of the sorbent material upon a range of initial concentrations of Cu(II) at different temperatures, agitation time and pH of the Cu(II)- TPJB system. The amount of Cu(II) ion adsorbed on to TPJB surface is found to be 43.11 mg/g (97.4%) under optimized conditions, its efficiency 3 fold times more than the Ce values reported by other researchers. The sorption characteristic of TPJB is quantitatively estimated through column experiments based on the Ce value by batch mode. The removal is observed as 98%. Langmuir, Freundlich and Tempkin isothermal curves at various initial concentrations are plotted for Cu(II)-TPJB system wherein the straight line fit is best suited for the Freundlich isotherm model. The results show that the response of TPJB in trapping Cu(II) ions are influenced by various parameters being statistically verified using SPSS software, indicative of good correlation.
KEYWORDS:adsorption; characterization; copper ions; isotherms; operating factors
Download this article as:| Copy the following to cite this article: Andal N. M, Charulatha S, Gayathri N. S. Biosorption of Divalent ion on to Treated Prosopis juliflora Bark from Aqueous Solutions - Isothermal and Statistical Analysis. Orient J Chem 2016;32(2) |
| Copy the following to cite this URL: Andal N. M, Charulatha S, Gayathri N. S. Biosorption of Divalent ion on to Treated Prosopis juliflora Bark from Aqueous Solutions - Isothermal and Statistical Analysis. Orient J Chem 2016;32(2). Available from: http://www.orientjchem.org/?p=15315 |
Introduction
Rapid industrialization with the utilization of the heavy metals in industries viz., paints, pigments, batteries, ceramic glazes, electroplating and textiles (1), over the past few decades has led to serious environment problems because of their increased disposal(2). Heavy metals like copper, nickel, zinc, cadmium, lead, chromium and mercury have been extensively employed in industries like paints, pigments, textile and electroplating industries, leading to higher concentrations in effluent discharges. Copper is a widely used metal in industries like mining, metallurgical, paints, pigments, electroplating and electronics (3) The permissible limits recommended by WHO for Copper discharge in waste water and drinking water are 0.05 and 0.005 mg/L, only [4]. Beyond the permissible limit, it is declared carcinogenic and leads to ailments like gastrointestinal problems apart from headaches, fatigue, depression, skin rashes, learning disorders, the accumulation being in kidneys, brain, skin, pancreas and heart (5).
The present work deals with the employment of Prosopis juliflora bark in adsorption of Cu(II) which has not been reported for any heavy metal removal so far in literature. Prosopis juliflora is a fast growing small tree in India. It is mainly cultivated and consumed for timber and furniture industries in the developing nations[8].
Material and Methods
The collected Prosopis juliflora barks were washed well with distilled water, dried, pulverized using electrical mixer and categorized into different mesh sizes using scientific test molecular sieves, further treated with 0.1 N HCl to enhance the sorption capacity, thereafter being referred as Treated Prosopis juliflora Bark (TPJB). TPJB with different mesh sizes (0.18mm, 0.21mm, 0.30mm, 0.42mm and 0.71mm) were subjected to image microscopic analysis in order to determine the particle sizes of TPJB using Binocular Microscope (OLYMPUS make, Model- CX21I). The physio chemical characteristic studies employing various methods were performed and the values were registered. Morphology and elemental constitutions of the adsorbent material were identified using Scanning Electron Microscope (SEM) and Energy Dispersive X-ray Analysis (EDAX)(JEOL JFM- 6390).
Batch Mode Adsorption Studies
The agitation of Cu(II) ions with TPJB in a mechanical shaker (KEMI) was experimentally verified to define the role of variable parameters viz., particle sizes, (0.71 mm, 0.42 mm, 0.30 mm, 0.21 mm and 0.18 mm) and dosages, (200 mg, 300 mg, 400 mg and 500 mg ) of TPJB, initial concentrations of the aqueous Cr(VI) ion solutions, (20-100 ppm: 20 ppm intervals) preset time intervals between the sorbate and sorbent species, (30- 120 min: 30 mins intervals) pH of the medium, ( 3, 5, 7, 9 and 11) and temperature of the system (293K- 333 K: 10 K intervals) to assess maximum sorption capacity of the chosen material. The initial and residual concentrations of Cu(II) in solutions were analyzed using an Shimadzu (AA 6200) Atomic Absorption Spectrophotometer.
Statistical Analysis
The relationship between adsorbed Cu(II) ions and variable parameters was correlated using Pearson Moment Coefficient Method. The extend of statistical fit was verified using SPSS 20 software, the output variable being ANOVA, Pearson Correlation and descriptive analysis with a significance based on 95% confident level.
Results and Discussion
Physio- Chemical Characterization
The registered physiochemical characteristic values of TPJB listed in table 1 are discussed as follows: The decreased bulk density value (0.63g/L) of TPJB may be the cause for enhanced adsorption of Cu(II) ions, implying the availability of large number of pores. The standardized bulk density and particle density values < 1.2 and < 2.2 respectively support the fine nature of TPJB. Moisture content value (≈ 5%) indicates favorable adsorption, since the extent of sorption activity shows reduction upto 25%. The porosity of TPJB is 56.55, it is the measure of micro pore content of the material. BET and BJH methods were used for the evaluation of the particle and mesopore size distribution. Pore sizes are classified in accordance with the classification adopted by the International Union of Pure and Applied Chemistry (IUPAC manual., 1982)i.e) [micropores diameter(d<20A°),mesopores (20A°< d< 500A°) and macropores (d > 500A°]. Because of the larger sizes of liquid molecules, the adsorbents for liquid phase adsorbates should have predominantly mesopores in the structure (9).The micro pores are responsible for the large surface area (3.28m2/g) of TPJB, which is created by the activation process and the mean pore diameter is found to be 6.56nm. PJB is found to possess mesopores predominantly, as their pore diameter lie in the range of 20A° < d < 500A°. The lesser ash content shows the presence of low quantity of inorganic matter and high quantity of the carbon content. (10). The amount C, H and S contents present in TPJB are given in table 1. The physio-chemical characteristic of other adsorbents are given in table 1a.
Table 1: Physicochemical Characteristics-TPJB
|
Factors |
Methods/Instruments |
Values |
| pH (1 % solution) | pH meter | 5.62 |
| Conductivity (Mv) | Conductivity Meter | 43.23 |
| Bulk density (g/L) | Specific gravity bottles | 0.63 |
| Moisture Content(%) | Xylene | 5.97 |
| Specific gravity | Pycnometer | 1.45 |
| Porosity | BET | 56.55 |
| Ash content (%) | Muffle Furnace | 3.93 |
| Surface area (m2/g) | BET | 3.28 |
| Mean Pore diameter (nm) | BET | 6.56 |
| Carbon (%) | CHNS Analyzer | 44.95 |
| Hydrogen (%) | CHNS Analyzer | 4.69 |
| Nitrogen (%) | CHNS Analyzer | 0.67 |
Table 1a: Physio chemical properties of some reported adsorbents
|
Factors |
Values |
||
|
Rice Husk |
Mesquite tree |
Aeromonas hydrophila |
|
| pH of 1 % solution | 6.3-6.5 | 7.4 | 3.5 |
| Bulk density (g/L) | 0.31 | 0.82 | 0.119 |
| Moisture (%) | 8.25 | 3 | 6.8 |
| Porosity | 0.38 | 54.95 | 0.629 |
| Ash content (%) | 18.39 | 3.9 | 4.28 |
| Carbon (%) | 30.56 | 40.65 | 25.79 |
Surface Morphology
The surface morphological pictures of the unloaded and metal-loaded TPJB recorded using SEM are depicted in figures 1a & 1b respectively. Fig 1a shows a high porous nature and a coarse surface texture with pores of different shapes and sizes, but fig 1b registers the effect of Cu(II) binding onto the adsorbent surface as cluster arrangement over the adsorption period. This is obvious from the distinct surface pores visible in 1a, but unseen in 1b indicative of adherence of the metal ion on TPJB.
 |
Figure 1a: SEM-TPJBFigure 1b: SEM- TPJB-Cu(II) Click here to View figure |
 |
Figure 2a: EDAX- TPJBFigure 2b: EDAX-TPJB- Cu(II) Click here to View figure |
EDAX Analysis
The EDAX spectra were recorded to analyze the elemental constitution qualitatively the % of O, C, Ca and Cl being 47.13, 52.2, 0.58 and 0.09 respectively in TPJB. These have been reported as the principle elements of any adsorbent (11). The new peak at the energy range of 8-9 KeV in figure 2b, confirms the Cu(II) adsorption onto TPJB.
Effect of Particle size
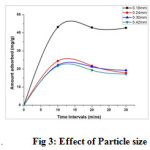 |
Figure 3: Effect of Particle size Click here to View figure |
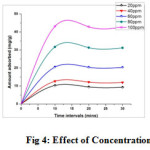 |
Figure 4: Effect of Concentration Click here to View figure |
The influence of particle sizes of the modified material revealed that the uptake of ions increased with a decline in its size. The uptake to Cu(II) ions by 0.18 mm particle size of TPJB (fig 3) is observed as a smooth steep curve indicating greater amount adsorbed (44 mg/g) against other particle sizes (0.18mm,0.24mm, 0.30mm and 0.42mm), the amount adsorbed range being 44 to 23.3 mg/g. This can be attributed to the fact smaller the particle size, greater is the surface area per unit weight of the adsorbent (12). Based on the statistical analysis mentioned in experimental part, the p- value calculated for the influence of particle sizes are as follows. The model F-value is the ratio of mean square for the individual term to the mean square for the residual. The Prob > F value is the probability of F-statistics value and is used to test the null hypothesis. The parameters having an F-statistics probability value less than 0.05 are said to be significant. The P-value of 0.0497 at 95% confident level (P<0.05) shows statistically that there is significant correlation in adsorption pattern. The negative correlation value r= -7503 were found between the particle sizes and the corresponding the values of amount adsorbed. The negative signs represented that as the particle sizes increased, the amount adsorbed values decreased.
Effect of Initial concentration and Agitation time
The initial metal ion concentration provides an important driving force to overcome all mass transfer resistance of Cu(II) between the aqueous and solid phase. The effect of time course profiles (10-30mins:10mins intervals) for the adsorption of ions under study at varied concentrations (20 to 100 mg/L) is shown in figure 4. The initial uptake rate was very rapid and thereafter adsorption was gradual and an equilibrium was reached. About 98 % maximum removal of Cu(II) ions was registered at 10 min contact time, after which an equilibrium was attained. The metal uptake curve is single, smooth and continuous leading to saturation, suggesting the possible monolayer coverage of metal ions onto the surface of adsorbent (13). The P-value of 0.0045 at 95% confident level (P<0.05) shows statistically that there is very significant correlation in adsorption pattern. The positive correlation coefficient r=0.9759 were found between the initial concentrations and the corresponding the values of amount adsorbed. The positive signs demonstrated that the patterns of changes in corresponding values were relatively similar.
Effect of pH
The pH dependence of Cu2+ biosorption under optimized conditions onto TPJB is shown in Fig. 5. The inverted parabolic curve represents that maximum qe had occurred at pH 9, thereafter a decline was observed. The reduction can be attributed to the fact that the precipitation of ions occur during ion exchange experiments by NaOH. Also, the lower qe values at corresponding pH values indicate the competition between the protons (H+) and metal ions (Cu2+) for the exchange sites (14). From the observation pH 9 is declared as effective pH for maximum trapping of Cu(II) by TPJB. The P-value of 0.0355 at 95% confident level (P<0.05) shows statistically that there is significant correlation in adsorption pattern. The positive correlation coefficient r=5356 were found between the pH and the corresponding the values of amount adsorbed.
Effect of Temperature
Temperature has a pronounced effect on the adsorption capacity. Fig 6 reveals that the removal of Cu(II) registered a sudden increase (38% to 97.4%) from 293K to 303K after which the curve reached a plateau nature, with increase in temperature up to 313K (15,16,17). The higher percentage removal may be attributed to the fact that increased temperature rises the mobility of Cu(II) ions facilitating enhanced adsorption. The P-value of 0.465 at 95% confident level (P<0.05) shows statistically that there is significant correlation in adsorption pattern. The positive correlation coefficient r=7178 were found between the temperature and the corresponding the values of amount adsorbed.
 |
Fig 5: Effect of pH Click here to View figure |
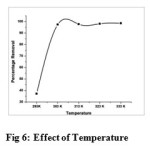 |
Figure 6: Effect of Temperature Click here to View figure |
Two way ANOVA results of various parameters are given in table 2. The linear regression plots of various parameters are given below
Table 2: Descriptive analyses results for the different parameters
| Dependent Variables | Sum Square | Degree of Freedom | Mean Square | F | Standard Deviation | Standard Error |
| Particle Size | 314.4 | 4 | 68.71 | 2.576 | 10.237 | 5.119 |
| Dosage | 752.53 | 4 | 11.93 | 60 | 13.716 | 6.134 |
| pH | 743.72 | 4 | 247.91 | 1.205 | 16.144 | 7.22 |
| Temperature | 1430.4 | 4 | 476.81 | 3.188 | 27.16 | 12.15 |
 |
figure 6b Click here to View figure |
Langmuir Isotherm
The Langmuir sorption isotherm describes the removal of adsorbate that had occurred on a homogeneous surface by monolayer sorption, without interaction between adsorbed molecules (18)
The Langmuir constants qm and b were determined from the linear plot (fig 7) of Ce/qe versus Ce, with a slope of 1/qm and intercept of 1/bqm (table 3). Higher the value of b, higher is the affinity of adsorbent for the metal to be sorbed (19)
The essential characteristics of Langmuir isotherm can be expressed in terms of a dimensionless parameter called the separation factor or equilibrium parameter RL, which is defined by the following relationship (20)

where, Ci is the initial metal ion concentration (mg/L). The parameter RL indicates the shape of the isotherm and the nature of the sorption process (21)
The correlation coefficient values (R2) for the metal is approximately 0.9851 (Table 4). The RL values for Cu(II)-TPJB lying between 0.01 and 0.74 (table 3) indicate favourable adsorption.
Freundlich Isotherm
The Freundlich equation is commonly used for mathematical description of adsorption in aqueous system. Kf and 1/n are Freundlich constants related to adsorption capacity and adsorption intensity, respectively. Both Kf and1/n are empirical constants being indicative of sorption capacity and adsorption intensity, respectively (22)
Freundlich constants (adsorption capacity and adsorption intensity) were calculated from the linear plot. Both Kf and1/n are empirical constants being indicative of sorption capacity and adsorption intensity, respectively (23). From the slope and intercept of plots log qe vs log Ce (fig 8), values of Kf and R2 were determined (table 4). The R2 values (0.9985) from the plot suggests maximum linearity and the better fit in of Cu(II)-TPJB on Freundlich adsorption isotherm.
Tempkin Isotherm
Tempkin isotherm equation contains a factor that explicitly takes into account adsorbent-adsorbate interactions. It assumes that the heat of adsorption of all the molecules in the layer decreases linearly with coverage due to adsorbate-adsorbate repulsions and the adsorption is a uniform distribution of maximum binding energy (24).
where, BT= RT/bT, T is the absolute temperature in Kelvin and R is the universal gas constant, 8.314 J/mol K. The constant bT is related to the heat of sorption. The constant AT is the equilibrium binding constant corresponding to the maximum binding energy.
Tempkin isotherm was applied to the adsorption data under investigation, as per equation (6). Tempkin constants AT and bT corresponding to the equilibrium binding constant and heat of adsorption are obtained from the linear plot of ln Ce versus qe (fig. 9).
The constants and the correlation coefficient values (table 4) imply that Tempkin (R2= 0.9661) and Langmuir (R2= 0.9851) isotherms are obeyed by the system less effectively when compared to Freundlich model (R2= 0.9985).
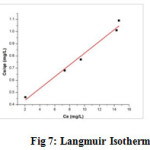 |
Figure 7: Langmuir Isotherm Click here to View figure |
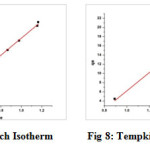 |
Figure 8a: Freundlich Isotherm Figure 8b: Tempkin Isotherm Click here to View figure |
Table 3: Equilibrium parameter RL for Cu(II) system
|
Conc. of metal ion (mg/L) |
Cu(II)- TPJB |
|
20 |
0.26 |
|
40 |
0.15 |
|
60 |
0.1 |
|
80 |
0.08 |
|
100 |
0.07 |
Table 4: Isotherm constants
|
Isotherm |
Cu(II) -TPJB |
|
|
parameters |
||
|
Langmuir isotherm |
||
|
qm (mg/g) |
20.73 |
|
|
b (L/g) |
0.1424 |
|
|
R2 |
0.9851 |
|
|
Freundlich isotherm |
||
|
KF (mg/g) |
4.0945 |
|
|
n |
0.9815 |
|
|
R2 |
0.9985 |
|
|
Tempkin isotherm |
||
|
AT (L/g) |
4.9762 |
|
|
bT |
1.0493 |
|
|
R2 |
0.9661 |
|
 |
Figure 9 Click here to View figure |
Column Studies
From the optimized batch equilibration results, Column studies were performed to quantify the removal of Cu(II)-TPJB system. Fixed–bed columns, made of cylindrical glass tube (2.5 cm inner diameter and 30 cm height) were packed with the TPJB between two supporting layers of glass wool, spread with the glass beads at the bottom layer of glass wool. TPJB were loaded from the top of the column and allowed to settle by gravity force. 100ppm of Cu(II) solution was added from the top and the flow rate was adjusted by collection of 5 ml of the sorbate at 10 minutes time interval. The maximum removal of TPJB was registered as 98%.
Conclusion
The selected agricultural waste (Prosopis juliflora) was modified using HCl for the removal of Cu(II) ions. The physio chemical characteristic studies viz., pH, conductivity, moisture content, bulk density, specific gravity, porosity, ash content and elements (C,H,N,S) were determined. The surface morphological changes, elemental constitutions and surface area of TPJB were determined using SEM, EDAX and BET respectively for unloaded and loaded material. The changes in porosity structure (SEM) and the appearance of Cu(II) peaks in the EDAX images support the adsorption positively. The Batch equilibration conditions for maximum percentage removal (98%) of Cu(II)- TPJB system was optimized at: 0.18mm particle size, 200 mg dosage, 10 minutes agitation time, 100 mg/L initial concentration of Cu(II) ions, pH 9 of the solution medium at 300C. Isothermal analysis of the data emphasized that the adsorption pattern exhibited a better fit for Freundlich isothermal plot than other all isothermal studies (Langmuir and Tempkin). Experiments were performed to assess the efficiency of TPJB through continuous column running for quantitative estimation of Cu(II) removal from the bulk of the solution. The statistical output data for viz., particle sizes, initial Cu(II) concentrations, dosages, pH of the solutions and temperatures had a significant effect on Cu(II) adsorption. A good correlation between the input and output variables is observed by applying SPSS 20 software.
References
- Amudaa, O.S.; Giwa, A.A.; Bello, I.A. Biochem. Eng. J. 2007, 36, 174–181.
CrossRef - Mohammad, W; Amer, Fawwaz I. Khalili; Akl M. Awwad. J. Env. Chem.& Ecotoxi. 2010, 2(1), 001-008.
- Karthika, C; Sekar, M. Res. J. Chem. Sci. 2013, 3(3), 65-69.
- WHO, Guidelines for Drinking- Water Quality (1984).
- Sureshkumar; Halnor Maqdoom Farooqui; Milind Ubale, Inter.J. Appl. Inno. Engin.& Manag. 2013, 2(3), 125-135.
- Nazar Abdualaziz Elnasri Hamza; Almoeiz Yousif Hammad; Mohammed Abuzeid Eltayeb, Sci J. of Analy. Che., 2013, 1(2), 12-20.
- HakanDemiral; IlknurDemiral; FatmaTumsek; BelginKarabacakoglu, Chem. Eng. J. 2008, 144, 188-196.
- Demirbas, E; Kobya, M; Konukman, A.E.S. J. Hazard. Mater. 2008, 154, 787-794.
CrossRef - Efficient adsorption of Cr (VI) from aqueous solution on low cost adsorbent developed from Limonia acidissima (Wood apple) shell. Ads. Sci.& Tech., 2010, 28 (6), 547-560.
CrossRef - El-Ashtoukhya, E.-S.Z; Amina, N.K; Abdelwahab, O. Desal. 2008, 223, 162–173
CrossRef - Adie, D.B; Okuofu, C.A; Osakwe, C; Inter. J. of Appl. Sci.& Tech., 2012, 2(7), 165-172
- Michael Horsfall Jnr; Jöse L. Vicente, Bull. Chem. Soc. Ethiop., 2007, 21(3), 349-362
- Sivakumar, K.K; Dheenadayalan, M.S; Eur. Chem. Bull. 2012, 1(5), 161-167
- Makeswari, M; Santhi, T. Arab. J. Chem. 2014
- Montanher, S.F.; Oliveira, E.A.; Rollemberg,M.C. J. Hazard. Mater., 2005, 117, 207-211.
CrossRef - Karthikeyan, T.; Rajagopal, S.; Lima Rose Miranda. J. Hazard. Mater.,2005, 124, 192-199.
CrossRef - Bethedsa, MD; National toxicology information Program, National Library of Medicine. 1993
- Minaxi, B; Lohani; Amarika Singh; Rupainwar, D.C; Dhar D.N. J. Hazard. Mater., 2008, 159, 626-629.
- Gupta, V.K; Rastogi, A. J. Hazard. Mater., 2008, 152, 407-414.
CrossRef - Kumar, M; Tamilarasan, R; J. of Che. Tech., 2013, 15(2), 29- 39.
- Zabihi, M; Ahmadpour, A; HaghighiAsl, A. J. Hazard. Mater., 2009, 167, 230-236
CrossRef - Hanim Awab; Thanalechumi Paramalinggam, J. of Fund. Sci., 2012, 7(2), 113-119.
- ZouhairElouear, J; Bouzidand, N; Boujelben. Worldwide workshop for young Environmentalists, 2010
- Stella Triyantafylou; Eirini Christoudolou; Paraskevi Neou-Singouna. Clay& Clay Min. 1999, 47, – ]D567-572.

This work is licensed under a Creative Commons Attribution 4.0 International License.









