Synthesis of Some Pyrazolone Derivatives and Evaluation of its Antibacterial and Cytotoxic Activity
Rishiram Prajuli , Janmajoy Banerjee*, and Hemant Khanal
Department of pharmacy Sunsari Technical College Dharan (NEPAL) Corresponding Author Email: jj.banerjee983@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/310430
Article Received on :
Article Accepted on :
Article Published : 24 Nov 2015
A series of novel pyrazolone derivative were synthesized by two different schemes (scheme-1 by the reaction of phenyl hydrazine and ethyl acetoacetate with substituted benzaldehydes PYR-1 to PYR-4) and (by the reaction of synthesized chalcone with phenyl hydrazine PYR-5) and characterised with its physical parameters (M.P, colour, %yield, solubility etc.). The entire synthesized compound was tested for their antimicrobial activity against Gram-positive and Gram-negative strains of bacteria and brimeshrimp bioassay was conducted for evaluation of cytotoxic activity The Investigation of antimicrobial screening data revealed that most of the tested compounds showed moderate to good antimicrobial activity. And cytotoxicity activity of compounds was also found to be satisfactory.
KEYWORDS:Pyrazolone; Synthesis; Antibacterial; Cytotoxicity Activity
Download this article as:| Copy the following to cite this article: Prajuli R, Banerjee J, Khanal H. Synthesis of Some Pyrazolone Derivatives and Evaluation of its Antibacterial and Cytotoxic Activity. Orient J Chem 2015;31(4). |
| Copy the following to cite this URL: Prajuli R, Banerjee J, Khanal H. Synthesis of Some Pyrazolone Derivatives and Evaluation of its Antibacterial and Cytotoxic Activity. Orient J Chem 2015;31(4). Available from: http://www.orientjchem.org/?p=12699 |
Introduction
Pyrazolone derivatives are the subject of many research studies due varieties of potential biological activities such as antimicrobial antiviral, antitumor, antihistaminic, antidepressant, anti-inflammatory anticancer, Antioxidant anticonvulsantand antidiabetic activities., cytotoxic activities [1-11].Pyrazolone derivatives is a important moiety of numerous pharmaceuticals, agrochemicals, dyes and pigments, chelating and extracting agent[12].Pyrazolone can be considered as intermediate compound for synthesis of various cyclic compounds of high biological activity. Day to day growing application on their synthesis and bioactivity, chemists and biologists in recent years has directed considerable attention on the research of pyrazolone derivatives. Based on various literature surves it was found that methyl and phenyl substituted pyrazolone derivatives exert significant pharmacological properties,as per as synthesis is concerned pyrazolens are synthesised by various ways, one common method is to synthesize from chalcones. [13] The synthesis of chalcones from substituted benzaldehyde and substituted acetophenone precursors proceeded according to the Claisen-Schmidt condensation. as chalcones are reported to be very biologically active[14]. In the present work 3-methyl N-phenyl derivatives of pyrazolones/pyrazole were synthesized by two schemes, scheme one with reaction of chalcone with phenyl hydrazine and scheme two includes reaction of phenyl hydrazine and ethylacetoacetate with substituted benzaldehyde and those synthesized compounds were subjected to cytotoxic and antibacterial activities to compare their potencies.
Experimental
Materials and Methods
All the chemicals and solvents were obtained from E-Merck, and SD fine chemicals L.T.D India (AR,LR grade)Melting points were determined by open capillary tube in paraffin, melting point bath and therefore the values reported are uncorrected .The purity of the compounds was checked by TLC, was run on silica gel.
General Procedure for synthesis of 3-Methyl-pyrazole-5-one [15]
Preparation of all methyl and phenyl pyrazolones derivatives were performed according to (G.M et al 2010) with slight modification.32.5 gm. of ethylacetoacetate was taken in 250 ml conical flask and stirred magnetically during slow drop wise addition of a solution of hydrazine hydrate (12.5 gm. hydrazine hydrate in 20 ml ethanol).The temperature of the reaction mixture was regulated at 60ºC as temperature rises during reaction. A crystalline deposit was separated after continuous stirring for 1 hour. The mixture was cooled in an ice bath as the product crystallized, and the solid obtained was filtered in a buchner funnel followed by washing with cold alcohol. The white colourless crystal was obtained which was dried, recrystallized form ethanol and used for further step.
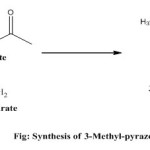 |
Figure 1 Click here to View figure |
General Procedure for synthesis of 3-Methyl-1H-Pyrazolone derivative
About 3.92 gm. of synthesized Pyrazolone (PYR-1)was taken in a 100 ml R.B flask and then 100 ml of freshly prepared 20% sodium hydroxide ethanolic solution was poured into it. The mixture was stirred for 30 minutes with magnetic stirrer followed by addition of 5.6228 gm. of 4-chlorobenzaldehyde to the reaction mixture and keep under stirring for (8-10) hours,completion of reaction was monitored by T.L.C later the reaction mixture was transferred into the crushed ice and neutralized with dil HCl to precipitate the product and kept in freeze overnight. It was then filtered, dried and purified by recrystallization from ethanol.
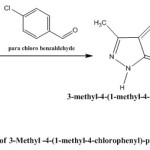 |
Figure 2 Click here to View figure |
General Procedure for synthesis of 3-Methyl-1H-Pyrazolone derivative
About 3.92 gm. of synthesized Pyrazolone (PYR-1)was taken in a 100 ml R.B flask and then 100 ml of freshly prepared 20% sodium hydroxide ethanolic solution was poured into it. The mixture was stirred for 30 minutes with magnetic stirrer followed by addition of 5.6228 gm. of 4-chlorobenzaldehyde to the reaction mixture and keep under stirring for (8-10) hours,completion of reaction was monitored by T.L.C later the reaction mixture was transferred into the crushed ice and neutralized with dil HCl to precipitate the product and kept in freeze overnight. It was then filtered, dried and purified by recrystallization from ethanol.
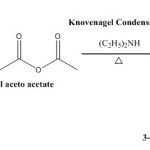 |
Figure 3 Click here to View figure |
General Procedure for synthesis of 3-Methyl-1-Phenyl-Pyrazolone Derivative
About 3.5 gm. of synthesized Pyrazolone (PYR-3) was taken in a 100 ml R.B flask and then 60 ml of freshly prepared 20% sodium hydroxide ethanolic solution transferred to it and the mixture was stirred in magnetic stirrer for 30 min.2.98 gm. of 4-dimethylaminobenzaldehyde was added to the reaction mixture and keep under further stirring for (8-10) hours, completion of the reaction was monitored by T.L.C. later reaction mixture was transfer into crushed ice and neutralized with dil.HCl to precipitate the product and kept in freeze overnight. It was then filtered, dried and purified by recrystallization.
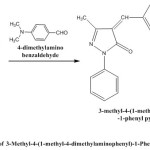 |
Figure 4 Click here to View figure |
General Procedure for Synthesis of Chalcones:
A mixture of 6 ml acetophenone and 7.45 gm. of 4-dimethylaminobenzaldehyde mixed in 30% ethanolic NaOH was poured into conical flask and the mixture was stirred in presence of 50 ml of petroleum ether under room temperature for 4 hour. The resulting solution was allowed to stand overnight in refrigerator and poured into ice-cold water and then it was neutralized with HCl. The solid obtained was filtered at the pump dried and recrystallized from ethanol.
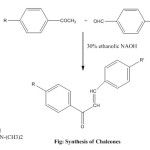 |
Figure 5 Click here to View figure |
General Procedure for Synthesis of Pyrazolone from chalcones:
Synthesis o chalcone was performed according to (Shaik Abdul Rahaman et al 2013) with slight modification. A mixture of synthesized chalcones 5.02 gm. and 4.32 gm. phenyl hydrazine was refluxed in in 80 ml glacial acetic acid for 8 hours. The mixture was cooled and poured into crushed ice as the solid precipitated and the solid mass was filtered in the buchner funnel, and made free of acid by washing with cold water, dried and recrystallized from ethanol.
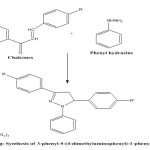 |
Figure 6 Click here to View figure |
Antibacterial Activity
Method
Cup plate Agar Diffusion Method
In this technique, Petridishes of Agar are prepared by pouring melted Agar media previously inoculated with selected microorganism. After the solidification of Agar cups are made with the help of borer and cups are filled with solution of suitable concentration of sample and standard respectively and are inoculated at 37º c for 24 hours. Petri dishes, cork borer, conical flask, glass syringe, test tubes, were sterilised by autoclaving at 121ºC temperature and 15 Lb/inch2 pressure for 15 minutes, four strains were selected Staphylococcusaureus, Staphylococcus faecalis inoculation of these bacterial strains were made in Nutrient Broth by transferring a loop full of organisms from a laboratory made mother culture, and incubated at 37º C for 18-24 hours. Each test compounds of 8 mg was dissolved in 10 ml of sterile water for injection to give a stock solution of 800µg/ml. Then 5 ml of it was taken and diluted with another 5 ml of sterile water to produce 400µg/ml and serially 200µg/ml, 100µg/ml, 50µg/ml, 25µg/ml was produced. 0.1 ml of this solution was used for antibacterial testing. The antimicrobial agents diffuses through the agar around its cups and produces a characteristics zone of inhibition of microorganism sensitive to the sample, the diameter of which can be measured and MIC is to be calculated.
Brine Shrimplethality Bioassay[17]
50mg of all synthesized compounds to be tested was dissolved in 5ml of distilled water (stock solution). Artificial Seawater was prepared in laboratory by referred procedure. Hatching of brine shrimp was done in the beaker with 300ml of seawater by sparkling about 50mg of brine shrimp eggs. This was then illuminated with table lamp of 100 watt for twenty-four hours to achieve the temperature of about 35 °C to hatch nauplii. After 24 hours, the nauplii were collected by dropper. From the stock solution 1000μl, 500μl, 250μl and 125μl were transferred to total of twelve different test tubes, three test tubes for each doses level after evaporating the solvent. Similarly in the other four test tubes, the process was repeated by taking same of the distilled water as control group. The nauplii were counted macroscopically in the stem of the dropper against the lighted background and ten matured and highly motile shrimp larvae were then transferred to each test tube and the volume was made up to 5ml on each test tube by adding the seawater. Similarly ten matured brine shrimp larvae were transferred in each test tube of control group and the volume was made 5 ml in each. After 24 hours, the test tubes were observed and the number of survived nauplii in each test tube was counted using magnifying glass. From the data obtained, the LC50 value was calculated.
Physical Parameters
Table 1: Physical Parameters of synthesized Compounds
|
S.N |
Product Code |
Molecular Formula |
Physical state |
% yield |
Solubility |
|
|
Soluble |
Insoluble |
|||||
|
PYR-1 |
C4H6ON2 |
Colourless white crystalline powder |
75.88 |
Ethanol Water |
Chloroform Benzene |
|
|
PYR-2 |
C11H9N2OCl |
Dark yellow solid |
59.50 |
Water
|
Chloroform |
|
|
PYR-3 |
C10H10N2O |
Light yellow solid |
89.0 |
Ethanol Water Chloroform |
Benzene |
|
|
PYR-4 |
C19H20N3O |
Brick red solid |
70.9 |
Water
|
Benzene
|
|
|
PYR-5 |
C23H22N3 |
48.23 |
Water |
Benzene |
||
Table 2: Melting Point of Synthesized Compounds
|
S.N |
Product Code |
IUPAC Name |
Melting Point(ºC) |
|
PYR-1 |
3-Methyl-pyrazole-5-one |
110 |
|
|
PYR-2 |
3-Methyl-4-(1-methyl-4-chlorophenyl)-pyrazol-5-one. |
118 |
|
|
PYR-3 |
3-Methyl-1-Phenyl-Pyrazolone |
105 |
|
|
PYR-4 |
3-Methyl-4-(1-methyl-4-dimethylaminophenyl)-1-Phenyl-Pyrazole-5-one |
112 |
|
|
PYR-5 |
3-phenyl-5-(4-dimethylaminophenyl)-1-phenyl-pyarazolone |
110 |
Table 3: Rf value of Synthesized Compounds
|
S.N |
Product Code |
Solvent system |
Proportion of Solvent |
Rf Value |
|
PYR-1 |
Benzene:Chloroform:Glacial Acetic Acid |
2:4:1 |
0.76 |
|
|
PYR-2 |
Benzene:Chloroform:Glacial Acetic Acid |
2:4:1 |
0.71 |
|
|
PYR-3 |
Benzene:Chloroform:Glacial Acetic Acid |
2:4:1 |
0.67 |
|
|
PYR-4 |
Benzene:Chloroform:Glacial Acetic Acid |
2:4:1 |
0.65 |
|
|
PYR-5 |
Benzene: Methanol |
9:1 |
0.70 |
Table 4: Antibacterial Activity of synthesized Compounds
Compound PYR-2
|
SL.NO |
Organism |
25 µg/ml | 50 µg/ml | 100 µg/ml | 200 µg/ml | 400 µg/ml | 800 µg/ml |
Standard (100µg/ml) |
| Staphylococcus faecalis | – | 08 | 10 | 11 | 13 | 14 | 21 | |
| Staphylococcus aureus | – | 11 | 16 | 17 | 18 | 18 | 20 | |
| E coli | – | 15 | 17 | 19 | 20 | 21 | 23 |
Compound: PYR4
|
SL.NO |
Organism | 25 µg/ml | 50 µg/ml | 100 µg/ml | 200 µg/ml | 400 µg/ml | 800 µg/ml | Standard(100 µg/ml) |
| Streptococcus faecalis | 10 | 15 | 17 | 19 | 20 | 21 | 22 | |
| Staphylococcus aureus | – | 08 | 11 | 15 | 17 | 19 | 20 | |
| E coli | – | 10 | 12 | 18 | 18 | 20 | 21 |
Compound PYR-5
| SL.NO |
Organism |
25 µg/ml | 50 µg/ml | 100 µg/ml | 200 µg/ml | 400 µg/ml | 800 µg/ml | Standard(100 µg/ml) |
| Staphylococcus faecalis | – | 09 | 10 | 12 | 18 | 20 | 17 | |
| Staphylococcus aureus | – | – | 11 | 15 | 19 | 20 | 21 | |
| E coli | – | – | 10 | 12 | 14 | 16 | 21 |
Table 5:Results of Brine Shrimp bioassay
|
S.N |
Compound |
Concn(µg/ml) |
No of Alive brine shrimp |
No of dead brine shrimp |
Mean of dead brine shrimp |
||||
|
1. 2. |
PYR-2 |
1000 |
0 |
0 |
1 |
10 |
10 |
9 |
9.66 |
|
500 |
2 |
3 |
1 |
8 |
7 |
9 |
8.0 |
||
|
250 |
9 |
7 |
8 |
1 |
3 |
2 |
2.0 |
||
|
125 |
9 |
9 |
10 |
1 |
1 |
0 |
0.67 |
||
|
PYR-4 |
1000 |
2 |
1 |
2 |
8 |
9 |
8 |
8.33 |
|
|
500 |
4 |
3 |
3 |
6 |
7 |
7 |
6.67 |
||
|
250 |
8 |
6 |
7 |
2 |
4 |
3 |
3 |
||
|
125 |
8 |
10 |
9 |
2 |
0 |
1 |
1 |
||
Table 6: % Lethality at log Concentration
|
S.N |
Concn (µg/ml) |
Log Concn |
% Lethality |
|
|
PYR-2 |
PYR-4 |
|||
|
1000 |
3 |
96.6 |
83.3 |
|
|
500 |
2.69 |
80.0 |
66.7 |
|
|
250 |
2.397 |
20.0 |
30.0 |
|
|
125 |
2.096 |
6.70 |
10.0 |
|
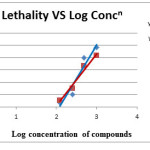 |
Figure 7 Click here to View figure |
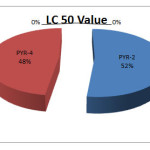 |
Figure 8 |
Discussion
Among all the derivatives of pyrazolone (PYR-1 to PYR-5) antibacterial activities of PYR1,PYR3 against different strains were determined previously so here PYR2,PYR4 and PYR5 was selected. PYR-2, PYR-4 are active against S.aureus and E.coli whereas PYR-5 is more active against S.aureus. Brine shrimp bioassay revealed that Compound PYR-2 showed LC50 value of 375.83 mcg/ml and PYR-4 showed LC50 value of 345.15 mcg/ml. Substitution on position-4 of pyrazole or pyrazolone ring are responsible for execution of the different IC50 values. [19]The LC50 values, thus, indicate the cytotoxic property of the compound, since these values are less than 1000mcg/ml. The IC50 of an agent is the doses, which will kill, or inactivate 50% of the test animal. IC50 is inversely proportional to the toxicity of a compound, i.e. lower is the IC50, and higher is the activity. [19]. PYR-4 is more active in comparison to PRY2,probably due to presence of phenyl group in position 1in PYR-4. Detail studies of toxicity are necessary. Detailed SAR studies are required for complete conformation of the work.
Acknowledgements
The authors want to acknowledge Sunsari Technical College, Dharan, Nepal for supporting to carry out the research work.
References
- Mohamed Aboul-Enein N, AidaElAzzouny A, Mohamed AttiaI, Yousreya Maklad A, Kamilia Amin M, Mohamed Abdel-Rehim , and Mohammed El-Behairy F, Design and synthesis of novel stiripentol analogues as potential anticonvulsants, Eur. J. Med. Chem., 2012.,47(1): 360-369
- Manish Agrawal, Pankaj Kumar Sonar, Shailendra Saraf K, Synthesis of 1,3,5- trisubstituted pyrazoline nucleus containing compounds and screening for antimicrobial activity, Med. Chem. Res., 2012., 21: 3376–3381
- Abdullah Sulaiman Al-Ayed, Synthesis, spectroscopy and electrochemistry of new Int J Pharm Bio Sci 2013 Apr; 4(2): (P) 727 – 733 This article can be downloaded from www.ijpbs.net P – 733 3-(5-aryl-4,5-dihydro-1H-pyrazol-3-yl)-4- hydroxy-2H-chromene-2-one as a novel class of potential antibacterial and antioxidant derivatives, Inter. J. Org. Chem., 1: 87-96, (2011)
- Seham Hassan Y, Synthesis and biological activity of some new pyrazoline and pyrimidine derivatives, J. Braz. Chem. Soc., 22(7): 1286-1298, (2011)
- Wang, X. H., Wang, X. K., Liang, Y. J., Shi, Z., Zhang, J., Chen, L. M., Fu, L. W., 2010. Chin J Cancer. A cell based screen for anti-cancer activity of pyrazolone derivatives. 29,980-987.
- Kumar Siva K, Rajasekharan A. Synthesis and Characterisation, in vitro antioxidants activity of Nmannich base of pyrazolone derivatives. Int. journal of research of pharmacy and chemistry. 2012; 2(2): 327-337.
- Parmar N, Shashikant T, Rikin P, Barad H, Jajda H, Thakkar V. Synthesis, antimicrobial and antioxidant activities of some 5-pyrazolone based schiff bases. J Saudi Chemical society, 2012
- Soni JP, Sen DJ and Modh KM. SAR studies of synthesized pyrazolone derivatives of imidazol, benzimidazole and benzotriazole moiety for anti-inflammatory activity. Journal of applied pharmaceutical science. 2011; 1(04): 115-120
- Kumar Siva K, Rajasekharan A. Synthesis and Characterisation, in vitro antioxidants activity of Nmannich base of pyrazolone derivatives. Int. journal of research of pharmacy and chemistry. 2012; 2(2): 327-337.
- Devnath HP, Islam MR. Synthesis of some pyrazolone derivatives from ciprofloxacin and study of their cytotoxicity. Bangladesh J Pharmacol. 2010; 5: 30-33.
- Khan Rahat, Uddin Imam, Alam M, Sultan D. Synthesis and cytotoxic activity of pyrazolone derivatives. Journal of the Bangladesh pharmacological society. 2008; 3: 27-35.
- Shweta S. Bule , M. R. Kumbhare, P. R. Dighe Synthesis and in-vitro Biological Evaluation of a Novel Series of 4-(Substituted)-5-Methyl-2-Phenyl-1, 2-Dihydro-3H-Pyrazol-3-One as Antioxidant. J. Chem. Bio. Phy. Sci. Sec. B; 2013, Vol.3, No.3; 1996-2005. E- ISSN: 2249 –1929
- Anees A Siddiqui, Md. Azizur Rahman, Md. Shaharyar, Ravinesh Mishra, Synthesis And Anticonvulsant Activity Of Some Substituted 3,5-Diphenyl-2-Pyrazoline-1-Carboxamide Derivatives.Chemical Sciences Journal, 2010: CSJ-8 (1-10).
- Shaik Abdul Rahaman et al /Int.J.PharmTech Res.2013,5(1).
- G.Mariyappan,B.P Saha,LSutharson,A Halder,Synthesis and bioactivity evaluation of pyrazolone derivatives. Indian Journal of Chemistry 2010:49B(1671-1674).
- Shweta S. Bule*, M. R. Kumbhare, P.R. Dighe, Synthesis and in-vitro Biological Evaluation of aNovel Series of 4-(Substituted)-5-Methyl-2-Phenyl-1,2-Dihydro-3H-Pyrazol-3-One as Antioxidant J. Chem. Bio. Phy. Sci. Sec. B; 2013, Vol.3, No.3; 1996-2005.
- Meyer BN, Ferrigni NR, Putnam JE, Jacobsen LB, Nichols DE, McLaughlin JL.Brine shrimp: a convenient general bioassay for active plant constituents.plant med 1982 May;45(5):31-4.
- N.V Kavitha*, Kalpana Divekar, Brahmani Priyadarshini , S. Gajanan. M. Manjunath, Synthesis and antimicrobial activities of some new pyrazole derivatives :Der Pharma Chemica, 2011, 3 (4):55-62
- Nusrat Binta Ahasan and Md. Rabiul Islam, Cytotoxicity study of pyrazole derivatives: Bangladesh J Pharmacol 2007; 2: 81-87

This work is licensed under a Creative Commons Attribution 4.0 International License.









