Spectrophotometric Determination of Arsenic in Water Samples by Thiophene-2-Carboxaldehyde Thiosemicarbazone impregnated with alumina
Md. Faridur Rahman1*, Aniruddha Chakraborty2 and Tanmoy Das1
1Department of Chemistry, University of Burdwan, Burdwan-713104, India
2Union Christian Training College, Murshidabad, WB, India.
Corresponding Authors E-mail: faridur1979@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/310471
Article Received on :
Article Accepted on :
Article Published : 05 Nov 2015
Thiophene-2-carboxaldehyde thiosemicarbazone has been successfully applied as an analytical reagent for micro level solid phase spectrophotometric determination of As (III) at pH 3.5. Here it can be seen that arsenic reacts with the new chelating ligand in acidic medium to form the As (III)-tctsc complex. The complex showed maximum absorbance at λ max 281 nm for As (III) ions. The Beer’s law range was 0.1-1.0 mg/L. Job’s method and Mole-ratio method showed that As (III)-ligand ratio in the complex is 1:2. The Molar absorptivity and Sandell’s sensitivity were 5.96 x 104 L mol-1 cm-1 and 0.01μg/cm2 respectively for As (III) ions. The proposed method has been successfully applied to the determination of arsenic in drinking water samples of Kaliachak, in the district of Malda, West Bengal, India.
KEYWORDS:Arsenic-tctsc Complex; Drinking Water Samples; Solid Phase Spectrophotometric (SPS) Determination
Download this article as:| Copy the following to cite this article: Rahman Md. F, Chakraborty A, Das T. Spectrophotometric Determination of Arsenic in Water Samples by Thiophene-2-Carboxaldehyde Thiosemicarbazone impregnated with alumina. Orient J Chem 2015;31(4). |
| Copy the following to cite this URL: Rahman Md. F, Chakraborty A, Das T. Spectrophotometric Determination of Arsenic in Water Samples by Thiophene-2-Carboxaldehyde Thiosemicarbazone impregnated with alumina. Orient J Chem 2015;31(4). Available from: http://www.orientjchem.org/?p=12213 |
Introduction
The natural occurrence of arsenic as in the form of Arsenopyrite (FeAsS), Orpiment (As2S), Realgar (AsS), Arsenolite (AsS4O6), Arsenic pentaoxide (As2O5) and it is also a significant component in minerals like energite, clomeykite, arthurite, chalcophyllite, conihalcite and pronstite. It is used in industry as wood preservatives and in paints, dyes, metals, soaps, insecticides and semi-conductors. Arsenic is a toxic metalloid, but it does not present in its elemental state. It is placed Gr.V and Pr.4 of the periodic table. The metallic form of arsenic with zero valency does not exerts adverse effects. In natural water, soluble arsenic occurs in the form of arsenate (As-V) and arsenite (As-III) in two different state of which arsenite (As-III) is toxic in natural environment. The source of arsenic in ground water is mostly due to the leaching of geological materials, minerals precipitation, and dissolution of unstable arsenic minerals, pesticides and fertilizers Apart from the above sources, it is also released into the environment through burning fossil fuels, cement manufacturing and mining activities1-6.
Some authors have suggested that the reduction dissolution of Fe-oxyhydroxide (FeOOH) is the dominant mechanism for the contamination of arsenic into ground water. Probable path for the arsenic contamination is due to the reduction of FeOOH and that the process is driven by the breakdown of natural organic matter present in the strata. Due to the toxicity nature of arsenic, World Health Organisation (WHO) has been allowed 0.05 mg/L of arsenic in drinking water. More than this, its presence causes various health problems like cardiovascular problem, gastrointestinal, haematological effects, dermal effects etc. Apart from this, elevated arsenic incidence is observed as pockets. Arsenius skin lesion like thickening of skin, white and black spots etc were noticed among the people of various countries in Asia and South
America. Long term ingestion of arsenic with drinking water causes hyper-pigmentation, melanosis, gangrene in limbs, hyper-keratosis on the palms of the hands and soles of the feet, black foot disease, and bone marrow depression. Arsenic is also toxic to the liver and it causes cirrhosis.
From the environmental point of view, the removal of arsenic from natural water, process solution and effluents is an important task to the concern people. Many reported methods have been conventionally employed for the determination and removal of arsenic from natural water samples. Some examples are polarographic speciation, atomic absorption spectrometry (AAS), hydride generatively coupled plasma atomic emission spectrometry (HG-ICP-AES), and capillary electrophoresis inductively coupled plasma mass spectroscopy (E-ICP-MS), electrothermal atomic absorption spectrometry (ETAAS), cathodic stripping voltammetry, anodic stripping voltammetry, neutron activation analysis, ionselective electrodes and energy dispersive X-ray fluorescence spectrometry7-12.
America. Long term ingestion of arsenic with drinking water causes hyper-pigmentation, melanosis, gangrene in limbs, hyper-keratosis on the palms of the hands and soles of the feet, black foot disease, and bone marrow depression. Arsenic is also toxic to the liver and it causes cirrhosis.
From the environmental point of view, the removal of arsenic from natural water, process solution and effluents is an important task to the concern people. Many reported methods have been conventionally employed for the determination and removal of arsenic from natural water samples. Some examples are polarographic speciation, atomic absorption spectrometry (AAS), hydride generatively coupled plasma atomic emission spectrometry (HG-ICP-AES), and capillary electrophoresis inductively coupled plasma mass spectroscopy (E-ICP-MS), electrothermal atomic absorption spectrometry (ETAAS), cathodic stripping voltammetry, anodic stripping voltammetry, neutron activation analysis, ionselective electrodes and energy dispersive X-ray fluorescence spectrometry7-12.
Materials and Methods
Instruments
A Shimadzu spectrophotometer (UV-1800) was used for recording all absorbance measurements. The pH measurements were carried out by a labtronic pH meter equipped with a combined glass-calomel electrode. A mechanical shaker (BOD-incubator, YONA Mfg., Indian Instruments Manufacturing Co., Kol-12) was used throughout the investigation.
Hexamethylene ammonium hexaethyldithiocarbamate, ammonium pyrolinedithiocarbamate (APDC), sodium diethyldithiocarbamate (NaDDC), silver diethylthiocarbamate, azure-B and naphthalene-methyltrioctyl ammonium chloride etc. are chelating agents which are used for the extractive determination of arsenic18-24. Among the above chelating agents sulphur donor atom containing chelating agents have great affinity for the complexation with arsenic and this encourage us to make sulphur containing another chelating agent for the extraction of arsenic from natural water.
In the present work, a new chelating agent i.e. thiophene-2-carboxaldehyde thiosemicarbazone has been found good application for the removal of arsenic from drinking water.
In the present investigation, the chelating agent is incorporated with alumina as matrix for the micro level determination of arsenic from DMF medium. The new method based on solid phase spectrophotometric (SPS) for the determination of arsenic complexed with thiophene-2-carboxaldehyde thiosemicarbazone.
Different optimum parameters are optimized such as pH, effects of temperature, concentration of metal ions, effects of diverse ions and elution studies etc by this method. It is simple, sensitive, economical and reliable spectrophotometric method than other reported methods for arsenic determination.
Reagents and Solutions
All the reagents used were of analytical grade from E. Merck, Germany and other metal salts from BDH, India were also of analytical grade. Thiosemicarbazide and thiophene-2-carboxaldehyde (Sigma and Aldrich) were used for the complexation. A standard stock of 0.01 M solution of arsenic (III) was prepared by dissolving appropriate amount of pure grade chemicals in double distilled deionised water. The working solutions were prepared by diluting appropriate volumes of stock solution in distilled water. For analytical application of the proposed method drinking water samples were collected in five different tube wells of Kaliachak situated in the district of Malda, West Bengal, India which is arsenic polluted area. Plastic bottles of 200 ml capacity were used for sample collection which were cleaned by soap and followed by washing in conc. HNO3. Before collection of sample, 0.5 ml of conc. H2SO4 solution was added in each plastic bottle because it preserves sample solutions for ~ 100 days.
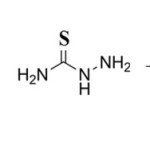 |
Scheme 1 Click here to View scheme |
Preparation of sorbent Matrix
Air dried of 1.0 g thiophene-2-carboxaldehyde thiosemicarbazone was impregnated with 3.0 g of alumina at the ratio of 1:2 in DMF in the presence of sodium carbonate. The mixture solution was stirred for 1 hour at room temperature. The resulting homogeneous composition was left in an oven at 600 c for one day. Finally, the sorbent matrix was ground into powder and preserved for further used.
Analytical procedure
For the batch method of extraction 300 mg of sorbents matrix were taken in each 100 ml flask with 20 ml of 0.1 mg/L of As (III) and 1 ml of DMF was added in each sets and the pH of the solutions mixture was adjusted to 3.5 by using dil. HCl and dil. NaOH solutions. The mixture of the sorbent matrix and As (III) solutions were shaken by a mechanical shaker at about 400-500 rpm at room temperature for 30 minutes. A reagent blank solution was prepared in the same procedure as mixture solution in absence of arsenic and the absorbance of the sample solution was measured against the blank at λ max 281 nm. The amount of As (III) absorbed on the sorbent materials was calculated by standard calibration curve. The standard solutions of arsenic were prepared in the range of 0.1-1.0 mg/L.
Results and Discussions
The impregnated reagent was used for the spectrophotometric method for determination of As (III). The As (III) ion forms coloured complex with the chelating agent which is stable. The quantitative extraction of As (III) was carried out at pH 3.5 and all the absorbance measurements were measured at λ max 281 nm [Fig.1].
The formula of the complex was ascertained by Job’s and molar ratio’s method as 1:2. Molar absorptivity was 5.96 x 104 L mol-1 cm-1. The analytical parameters of the solid phase spectrophotometric determination of As (III) are given in [Table 1].
Effect of pH on sorption of As (III)
For the quantitative recovery of As (III) by solid phase spectrophotometric (SPS) method pH is a key factor. The effect of pH on the absorbance of As (III)-complex was investigated. This result showed that the absorbance maximum was at pH 3.5. The quantitative extraction (> 95 %) found at pH 3.5. Hence, pH 3.5 was considered for the further studies. The results are shown in [Fig.2].
Influence of temperature
The influence of temperature on the absorbance of As (III)-tctsc was investigated in the temperature range of 20-600 C at pH 3.5. The maximum and stable absorbance was found at 350 C which predicting the optimum temperature for the complexation. Hence, for the extraction of As (III) by tctsc chelating agent the temperature 350 was chosen for further investigation which was shown in [Fig.3].
Effect of reagent (tctsc) concentration
The effect of the reagent concentration impregnated with alumina was investigated in the range of 0.1-1.0 mg. The results showed that extraction of As (III) increases on the addition of the reagent. It was found that quantitative extraction (> 90 %) at 0.5 mg. But beyond this range of the reagent concentration does not influence on the extraction of As (III).
Effect of As (III) ion concentration
At a fixed amount of tctsc concentration (0.5) the percentage extraction of As (III) was studied in the range of 0.1-1.0 mg/L at pH 3.5. It can be seen [Table 2] that over 80 % of As (III) was extracted in the above series of solutions range.
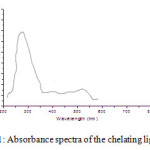 |
Figure 1: Absorbance spectra of the chelating ligand. Click here to View figure |
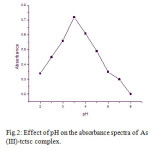 |
Figure 2: Effect of pH on the absorbance spectra of As (III)-tctsc complex. Click here to View figure |
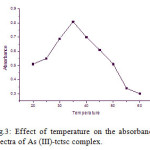 |
Figure 3: Effect of temperature on the absorbance spectra of As (III)-tctsc complex. Click here to View figure |
The Stoichiometry of As (III)-tctsc complex
The stoichiometry of As-tctsc complex was determined by Mole-Ratio method and Job’s method of continuous variation at fixed absorbance λ max 281 nm.
(a) Mole-Ratio method.
The extraction of arsenic was carried out in the range of 0.1-1.0 mg of the reagent concentration. The nature of the curve was straight line from the origin and then it becomes horizontal for further addition of the reagent which predicting the nature of the complex is 1:2. The curve is shown in [Fig.4].
(b) Job’s method of continuous variation.
For a fixed of the reagent concentration (0.5 mg/L) with the different concentration of As (III) ions was studied. The nature of the curve obtained by this method predicting the nature of the complex is 1:2. This is shown in [Fig.5]. The molar absorptivity is 5.96 x 104 L mol-1 cm-1.
Effect of foreign ions
As evident from [Table 3], in the solid phase spectrophotometric determination of arsenic, the presence of macro amounts of foreign ions of first transition series did not interfere in this sorption process. Only phosphate and vanadate interfere to some extent. So, the determination of arsenic was possible from the binary mixture of some transition, alkali and alkaline earth metal ions.
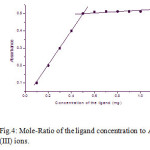 |
Figure 4: Mole-Ratio of the ligand concentration to As (III) ions. Click here to View figure |
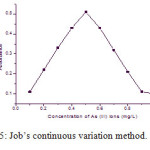 |
Figure 5: Job’s continuous variation method. Click here to View figure |
Table 1: Physical and Analytical Characteristic of As (III)-tctsc Complex.
|
Parameters |
Value |
|
pH |
3.5 |
|
Temperature |
350C |
|
Molar absorbance |
λ max 281 nm |
|
Limit of Detection (μg/ml) |
0.1-1.0 |
|
Beer’s law range |
0.1-1.0 mg/L |
|
Molar absorptivity |
5.96 x 104 L mol-1 cm-1 |
|
Stability Constant (β) |
8.01×107 |
|
Sandell’s Sensitivity |
0.01 μg/cm2 |
|
Compositions (Metal:Ligand) |
1:2 |
Effects of eluting agents on Desorption of As (III)
For quantitative recovery of As (III), selection of a suitable eluating agent is one of the important factors. But absorption of As (III) took place at low pH helps us to select acidic eluating agent like HCl, HNO3, oxalic acid and CH3COOH. Finally, it can be seen from [Table 4] that quantitative recovery of As (III) was found by using 1M HCl. The quantitative recovery was investigated by using 5 ml, 10 ml, 15 ml and 20 ml of 1M HCl for As (III).
Analytical Application
The application of the proposed method at optimum condition was applied for the determination of arsenic in drinking water samples which were collected in five different tube wells of Kaliachak situated in the district of Malda, West Bengal, India which is an arsenic polluted area. The drinking water samples were filtered through 0.45 μm pore size Millipore membrane filter and then shaked with sorbent matrix after adjusting the pH 3.5. The results summarised in [Table 5] and EDTA was used to remove the interferences. The results obtained by the proposed method for the determination of arsenic were about >90 %. So, the proposed method is very selective for separation of arsenic in different water samples.
Table2: Effect of As (III) ions concentration on the percentage extraction at pH 3.5
|
Used |
Extracted |
% E |
% RSD |
|
0.1 |
0.08 |
81 |
0.11 |
|
0.2 |
0.16 |
80 |
0.05 |
|
0.3 |
0.23 |
79 |
0.09 |
|
0.4 |
0.312 |
78 |
0.05 |
|
0.5 |
0.41 |
82 |
0.11 |
|
0.6 |
0.477 |
79.5 |
0.08 |
|
0.7 |
0.546 |
81 |
0.03 |
|
0.8 |
0.628 |
78.5 |
0.12 |
|
0.9 |
0.712 |
79.2 |
0.07 |
|
1.0 |
0.8 |
80 |
0.1 |
Table 3: Separation of 0.1 mg/L of As (III) ion from binary mixtures in a sample volume of 50 ml
|
Ions |
Tolerance |
% Recovery of |
Ions |
Tolerance |
% Recovery of |
|
Na+ |
5 |
101 |
Cl– |
100 |
98.7 |
|
K+ |
5 |
98.1 |
Br– |
100 |
99.4 |
|
Cd2+ |
5 |
99.5 |
SiO32- |
100 |
99.1 |
|
Ca2+ |
100 |
98.3 |
VO3– |
5 |
91.2 |
|
Mg2+ |
100 |
100 |
CH3COO– |
50 |
99.5 |
|
Al3+ |
50 |
102 |
CO32- |
50 |
97.5 |
|
Cr3+ |
50 |
99.2 |
SO42- |
50 |
97.8 |
|
Co2+ |
50 |
99.98 |
SO32- |
50 |
99.2 |
|
Fe3+ |
50 |
98.1 |
PO43+ |
5 |
92.4 |
|
Ni2+ |
50 |
97.5 |
NO3– |
500 |
98.7 |
|
Zn2+ |
5 |
97 |
NO2– |
500 |
100 |
|
Bi3+ |
50 |
98.6 |
CN– |
500 |
101 |
|
Cu2+ |
5 |
97.2 |
– |
– |
– |
Table 4: Desorption of studies of As (III) by different eluents.
|
Eluents |
% Recovery |
% RSD |
|
|
HCl |
1 mol L-1 |
93 .5 |
± 0.5 |
|
HCl |
2 mol L-1 |
83.2 |
± 0.7 |
|
HCl |
3 mol L-1 |
85.6 |
± 0.7 |
|
HNO3 |
1 mol L-1 |
87 |
± 0.8 |
|
HNO3 |
2 mol L-1 |
86.5 |
± 0.4 |
|
HNO3 |
2 mol L-1 |
85.2 |
± 0.2 |
|
CH3COOH |
1 mol L-1 |
65 |
± 0.6 |
|
C2H2O4 |
1 mol L-1 |
66 |
± 0.4 |
Table 5: Determination of Arsenic in drinking water samples by the proposed method.
|
Samples |
As (III) added |
As (III) found |
% Recovery |
|
|
Tube Wells |
Sample 1 |
0.00 |
3.46 |
— |
|
5.00 |
8.51 |
101 |
||
|
Sample 2 |
0.00 |
3.12 |
— |
|
|
5.00 |
8.21 |
101 |
||
|
Sample 3 |
0.00 |
3.7 |
— |
|
|
5.00 |
8.4 |
94 |
||
|
Sample 4 |
0.00 |
2.17 |
— |
|
|
5.00 |
7.12 |
99 |
||
|
Sample 5 |
0.00 |
2.37 |
— |
|
|
5.00 |
7.38 |
100 |
||
|
Tap Water |
Sample 1 |
0.00 |
3.01 |
— |
|
5.00 |
7.89 |
97.6 |
||
|
Sample 2 |
0.00 |
2.18 |
— |
|
|
5.00 |
7.01 |
96.6 |
||
|
Sample 3 |
0.00 |
1.76 |
— |
|
|
5.00 |
6.8 |
100 |
||
|
Sample 4 |
0.00 |
3.42 |
— |
|
|
5.00 |
8.12 |
94 |
||
|
Sample 5 |
0.00 |
3.17 |
— |
|
|
5.00 |
8.13 |
99.2 |
||
Proposed Structure of As (III)-tctsc Complex
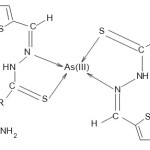 |
Scheme 1 Click here to View scheme |
Conclusion
The proposed method is a selective, accurate and sensitive for the determination of arsenic based on complexation due to the presence of soft base sulphur donor sites. The suggested method could be used for rapid determination of As (III) in drinking water samples. The wide applicability and simplicity of the proposed method make it different from other existing methods. It is evident in the proposed method that thiophene-2-carboxaldehyde thiosemicarbazone (tctsc) is most sensitive for the spectrophotometric determination of arsenic.
Acknowledgement
The authors are thankful to the Department of Chemistry of the University of Burdwan, Burdwan, India and Union Christian Training College, Berhampore, Murshidabad, West Bengal, India for providing the necessary laboratory facilities.
References
- Behpour, Mohsen; Ghoreishi, S.M., and Salehi, Sakineh, Acta Chim. Solv. 2005, 52, 323-327.
- Kumar, R. Kishore; Venkatesarlu, S; Reddy K. Vijaya, Kumar; Tulasammam, P.; Govinda, V.; Jyothi, N.V.V.; and Venkateswarlu, P., J. Chem. Pharm. Res.; 2011; 3(3), 626-634.
- Singh, Seema; Harjit, Jeena; Kataria, H.C.; and Amlathe, Sulbah, IOSR Journal of Applied Chemistry (IOSR-JAC), 2014, 7(4),.57-64.
- Rahman, M.F.; Chakraborty, A., and Das, T., IOSR Journal of Applied Chemistry (IOSR-JAC), 2015, 1,.14-20.
- Rahman, Md. Faridur; Chakraborty, Aniruddha; and Das, Tanmoy, Material Sc. Res. India, 2015, 12(2).
- Rehman, F.; and Mairaj, Samya, Orient. J.Chem., 2012, 28(2),. 881-885.
- Karayunlu, Seda; Ay, Umit, J. of Anal. Chem., 2010, 65,.244-248.
- Rahman, M.F.; and Chakraborty, Aniruddha, European Academic Research, 2014, II (3),.4195-4203.
- Rustamov, N.Kh; Abbasova, G.G., American J. Anal.Chem., 2014, 5, 275-280.
- Tavallali, H. and Kiani, S., Int. Journal of ChemTech Res., 2012, 4(3),.1182-1186.
- Moghimi, Ali, Australian J. of Basic and App. Sc., 2012, 6(3), 349-355.
- Rahman, M.F.; Chakraborty,A and Das, T, IOSR Journal of Applied Chemistry (IOSR JAC), 2014,.7, 10, .1,.54-69.
- Rahman, M. F.; Chakraborty, A., Bandyopadhyay, S. N.; Das, T., Golden Research Thoughts, 2014, 3, .9,.1-6.
- Elangovan,D. & Chalakh, M.L., Arsenic pollution in West Bengal.
- Purkait, Barendra and Roy, Amit Kr., A Report of Geological Survey of India.
- De, Anil K, Environmental Chemistry, Seventh Edition, New Age Int. Pub., 2010, 248.
- Mukherjee, Dulal C; Chowdhury, Sumit B; Paul, Ajoy and Das, Paulomi, J. Indian Chem. Soc., 2012,89,.9-18.
- Mehdi, Hosaini & Nasser, Dalali, Indian J. Chem. Tech., 2012, 19,.337-341.
- Pawar, R B; Padgaonkar; S B & Sawant, A D, Indian J. Chem. Tech., 2001, 8,.200-203.
- Dalali, Nasser; Farhangi, Leila & Hosseini, Mehdi, Indian Journal of Chemical Technology, 2011, 18,. 183-187.
- Bosque-Sendra, Juan M.; Valencia, C.; Boudra, Said, Fresenius J Anal Chem, 1998, 360, 31-37.
- Valencia, M.C.; Nicolas, E. Arana; Capitan-Vallvey, L.F., Talanta, 1999, 49, 915-921.
- Jayprakash Chauhan S; Pandya Ajit V, Int. Journal of Eng. Sc. Inven., 2013, 2(2)
- Siddappa, K.;Sultana, Mayana Nabiya, Int. J. Res. Chem. Environ.,2014, 3(2) 78-84.

This work is licensed under a Creative Commons Attribution 4.0 International License.









