(Acridine)(tetrahydroborato)zinc Complex [Zn(BH4)2(acr)]: A New Stable and Efficient Reducing Agent
Sina Mohammadi and Davood Setamdideh*
Department of Chemistry, Mahabad Branch, Islamic Azad University, Mahabad, Iran. corresponding Author E-mail: davood.setamdideh@gmail.com
DOI : http://dx.doi.org/10.13005/ojc/310470
Article Received on :
Article Accepted on :
Article Published : 22 Oct 2015
(Acridine)(tetrahydroborato)zinc complex[Zn(BH4)2(acr)] has been prepared by complexation of oneequimolar amounts of zinc tetrahydroborate and one equimolar amounts of acridine at room temperatureas gray stable reducing agents. Also, [Zn(BH4)2(acr)]has been used for reduce of different carbonyl compounds such as aldehydes, ketones, α, β-unsaturated carbonyl compounds, acyloins and a-diketones to their corresponding alcohols in excellent yields (85-95%). The reduction reactions have been carried outwithin 30-120 min by using of 0.5-1 equivalents of [Zn(BH4)2(acr)] in CH3CN at room temperature or under reflux conditions.
KEYWORDS:Zn(BH4)2; Acridine; Reduction; Carbonyl Compounds
Download this article as:| Copy the following to cite this article: Mohammadi S, Setamdideh D. (Acridine)(tetrahydroborato)zinc Complex [Zn(BH4)2(acr)]: A New Stable and Efficient Reducing Agent. Orient J Chem 2015;31(4). |
| Copy the following to cite this URL: Mohammadi S, Setamdideh D. (Acridine)(tetrahydroborato)zinc Complex [Zn(BH4)2(acr)]: A New Stable and Efficient Reducing Agent. Orient J Chem 2015;31(4). Available from: http://www.orientjchem.org/?p=12104 |
Introduction
Zn(BH4)2is unique because of a) the coordination ability of Zn2+, b) it’ssolubility in aprotic solventssuch as THF, Et2O and DME, c) an efficient chemo-, regio- and stereoselective reducing agent.So,it’s using and application is interesting in organic synthesis1-2.
Several Combination reducing systems of Zn(BH4)2 such as Zn(BH4)2/TMEDA3a, Zn(BH4)2/Me3SiCl3b, Zn(BH4)2/TFA/DME3c, Zn(BH4)2/H2O3d, Zn(BH4)2/Al2O33e, Zn(BH4)2/C 3f, Zn(BH4)2/2NaCl3g, Zn(BH4)2/U.S.3h, and Zn(BH4)2/ZrCl43i are interesting and have been used for different reduction purposes.However, zinc tetrahydroborate has been used less than regular reducing agents in laboratory, probably because of a) non-availability as a commercial reagent b) being freshly prepared. So, Zn(BH4)2, has been modified as stable complexes such as [Zn(BH4)2(dabco)]4,[Zn(BH4)2(pyz)]n5, [Zn(BH4)2(PPh3)] &[Zn(BH4)2(PPh3)2]6, [Zn(BH4)2(bpy)]7, [Zn(BH4)2(py)]8, [Zn(BH4)2XP4]9, [Zn(BH4)2(nmi)]10a, [Zn(BH4)2(nic)]10b and [Zn(BH4)2(caf)]10c.
In continuation of our interest for preparation of new modified tetrahydroborates, we have prepareda new stable ligand-zinc tetrahydroboratei.e. (acridine)(tetrahydroborato)zinc complex; [Zn(BH4)2(acr)]. Also, in this context,we have investigated the ability of [Zn(BH4)2(acr)] for the reduction of carbonyl compounds such as aldehydes, ketones,acyloins, a-diketones to their corresponding alcohols.
Results and Discussions
We examined the reduction of benzaldehyde as a model reaction. Among the tested different solvents benzaldehyde reduction was better in CH3CN. Our experiments showed that using 0.5 molar equivalents of [Zn(BH4)2(acr)] in CH3CN (3 mL) is the best conditions. Then, [Zn(BH4)2(acr)] has been used for reduce of different aldehydes under optimized reaction conditions (Table 1, entries 1-9). All reduction reactions were completed within 30-60 min by 0.5 molar equivalents of [Zn(BH4)2(acr)] in excellent yields of products(92-95%).
Our next attempt was the reduction of ketones. We optimized the reaction conditions with acetophenone as model compound. The reduction of ketones require a higher molar amounts of [Zn(BH4)2(acr)] because the reactivity of ketones is lower than aldehydes. The reduction reactions were carried out with 1 molar equivalents of [Zn(BH4)2(acr)] at reflux conditions in CH3CN. All reductions were completed within 80-120 min with high to excellent yields of products (85-93%) as shown in Table 1 (entries 10-17).
![Table 1. Reduction of a Variety of Carbonyl Compounds such as Aldehydes (entries 1-9), Ketones (entries 10-14), -diketones (15-16), Acyloins (entriy 17) and α, β-unsaturated carbonyl Compounds (entries 18-20) to their Corresponding Alcohols with [Zn(BH4)2(acr)] as Reducing Agent in CH3CN.](http://www.orientjchem.org/wp-content/uploads/2015/10/Vol31_No4_Acri_Dav_T1-150x150.jpg) |
Table 1: Reduction of a Variety of Carbonyl Compounds such as Aldehydes (entries 1-9), Ketones (entries 10-14), a-diketones (15-16), Acyloins (entriy 17) and α, β-unsaturated carbonyl Compounds (entries 18-20) to their Corresponding Alcohols with [Zn(BH4)2(acr)] as Reducing Agent in CH3CN. Click here to View table |
We also investigated the potential of the 1,2-reduction of α,β-unsaturated aldehydes and ketones with [Zn(BH4)2(acr)]. The reduction of cinnamaldehyde with 0.5 molar equivalents of the [Zn(BH4)2(acr)] exclusivity afforded the 1,2-reduction product after 40 min at room temperature in CH3CN. In this reaction, cinnamyl alcohol was obtained in 95% yield (Table 1, entry 18). Under this protocol, reduction of conjugated ketones such as benzylidenacetone (Table 1, entry 19) and chalcone (Table 1, entry 20) were achieved efficiently with 1 molar equivalents of [Zn(BH4)2(acr)] at reflux conditions in CH3CN in excellent yields (95-96%). The efficiency of [Zn(BH4)2(acr)] has been compared with other reported reducing systems (Table 2). In all cases[Zn(BH4)2(acr)] has a good potential for the reduction of organic carbonyl compounds.
![Table 2. Comparison of the Reduction of Aldehydes and Ketones by [Zn(BH4)2(acr)] in CH3CN with other Reported Reducing Agents.](http://www.orientjchem.org/wp-content/uploads/2015/10/Vol31_No4_Acri_Dav_T2-150x150.jpg) |
Table 2: Comparison of the Reduction of Aldehydes and Ketones by [Zn(BH4)2(acr)] in CH3CN with other Reported Reducing Agents. Click here to View table |
Experimental
All substrates and reagents were purchased from commercially sources with the best quality and used without further purification. IR and 1H NMR spectra were recorded on PerkinElmer FT-IR RXI and 300 MHz Bruker spectrometers, respectively. The products were characterized by their 1H NMR or IR spectra and comparison with authentic samples (melting or boiling points). Organic layers were dried over anhydrous sodium sulfate. All yields referred to isolated pure products. 1H NMR &TLC was applied for the purity determination of substrates, products and reaction monitoring over silica gel 60 F254 aluminum sheet.
Preparation of (Acridine)(tetrahydroborato)zinc Complex;[Zn(BH4)2(acr)]:
An ethereal solution of Zn(BH4)2 (0.16 M, 250 mL) was prepared from ZnCl4(5.452 g, 0.04 mol) and NaBH4 (3.177 g, 0.084 mol)according to an available procedure in the literature10. Then, acridine (7.17 g, 0.04 mol) in ether (50 mL) was added dropwise to the ethereal solution of Zn(BH4)2 and stirred for 30 min. Evaporation of the solvent under vacuum at room temperature gave [Zn(BH4)2(acr)] as a withe powder in a quantitative yield (10.08 g, 92%). Found: Zn: 23.2 %, B: 7.3 %. Calculated for C13H17B2NZn, Zn: 23.84 %, B: 7.88%. Scheme 1.
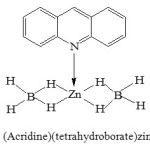 |
Scheme 1: (Acridine)(tetrahydroborate)zinc complex |
Reduction of Acetophenone to 1-phenylethanol with [Zn(BH4)2(acr)], A Typical Procedure:
In a round-bottomed flask (10 mL), equipped with a magnetic stirrer, a solution of acetophenone (0.120 g, 1mmol) in CH3CN (3 mL) was prepared. The complex reducing agent (0.274 g, 1mmol) was then added as a solid and the mixture was stirred at reflux conditions. TLC monitored the progress of the reaction (eluent; CCl4/Et2O : 5/2). After completion of the reaction in 90 min, a solution of 5% HCl (5 mL) was added to the reaction mixture and stirred for 10 min. The mixture was extracted with CH2Cl2 (3 × 10 mL) and dried over the anhydrous sodium sulfate. Evaporation of the solvent and short column chromatography of the resulting crude material over silica gel by eluent of CCl4/Et2O : 5/2 afforded the pure liquid benzyl alcohol (0.113 g, 93% yield).
Conclusion
In this context, we have shown that [Zn(BH4)2(acr)] reduces a variety of carbonyl compounds to their corresponding alcohols in high to excellent yields. Reduction reactions were carried out with 0.5-1 molar equivalents of [Zn(BH4)2(acr)] at room temperature and reflux conditions in CH3CN without any other additive. In addition, regioselectivity of this system was also investigated with exclusive 1,2-reduction of conjugated carbonyl compounds to their corresponding allylic alcohols in high to excellent yields. Reduction of acyloins and α-diketones by this reducing system also produced the corresponding vicinal diols.
Acknowledgments
The authors gratefully acknowledge financial assistance by the research council of the Islamic Azad University branch of Mahabad.
References
- Narasimhan, S.; Balakumar, R. Aldrichim.Acta. 1998,31, 19-26.
- (a) Ranu, B. C. Synlett 1993, 885-892. b) Ranu, B. C.; Chakraborty, R.Tetrahedron Lett. 1990, 31, 7663-7664. (c) Sarkar, D. C.; Das, A. R.;Ranu, B. C.J. Org. Chem. 1990, 55, 5799-5801.
- (a) Kotsuki, H.; Ushio, Y.; Yoshimura, N.; Ochi, M. Bull. Chem. Soc. Jpn. 1988,61, 2684-2686.(b) Kotsuki, H.; Ushio, Y.; Yoshimura, N.; Ochi, M. J. Org. Chem. 1987, 52, 2594-2596. (c) Ranu, B. C.; Das, A. R. J. Chem. Soc. Perkin Trans. 1 1992, 1561-1562. (d) Setamdideh, D.; Khezri, B.; Rahmatollahzadeh, M.; Aliporamjad, A. Asian J. Chem.2012, 8, 3591-3596. (e) Setamdideh, D.; Khezri, B.; Rahmatollahzadeh, M.; J. Serb. Chem. Soc. 2013, 78, 1-13. (f) Setamdideh, D.; Rahmatollahzadeh, M. J. Mex.Chem. Soc. 2012, 56, 169-175. (g) Setamdideh. D.; Khaledi, L. S. Afr. J. Chem.2013, 66, 150–157.(h) Fanari, S.;Setamdideh. D.; Orient. J. Chem., 2014, 30, 695-697. (i) Rasol, F.;Setamdideh. D.; Orient. J. Chem., 2013, 29, 497-499.
- Firouzabadi, H.;Adibi, M.;Zeynizadeh, B. Synth. Commun.1988, 28,1257-1273.
- Tamami, B.;Lakouraj, M. M.Synth. Commun.1995, 25, 3089-3096.
- Firouzabadi, H.; Adibi, M. Phosphorus Sulfur Silicon Relat. Elem. 1998, 142, 125-147.
- Zeynizadeh, B.Bull. Chem. Soc. Jpn. 2003,76, 317-326.
- (a) Zeynizadeh, B.;Faraji, F.Bull. Korean Chem. Soc.2003, 24, 453-459. (b) Zeynizadeh, B.;Zahmatkesh, K.J. Chin. Chem. Soc.2003, 50, 267-271. (c) Zeynizadeh, B.;Zahmatkesh, K.J. Chin. Chem. Soc.2004, 51, 801-806. (d) Zeynizadeh, B.;Zahmatkesh, K.J. Chin. Chem. Soc.2005, 52, 109-112.
- Firouzabadi, H.; Tamami, B.; Goudarzian, N.Synth. Commun.1991,21, 2275-2285.
- (a) Zeynizadeh, B.; Setamdideh, D. Asian J. Chem. 2009, 21, 3603-3610. (b) Setamdideh, D.;Rafig, M.E-J. Chem.2012, 9, 2338-2345. c) Abdollahpour, F.; Setamdideh, D. Orient. J. Chem. 2015, 31, in press.

This work is licensed under a Creative Commons Attribution 4.0 International License.









